Introduction
Aortic dissection (AD) is an uncommon condition, with a reported incidence of 3-5 cases per 100 000 people per year.(1) It is considered one of the most severe acute aortic syndromes, with a high morbidity and mortality rate, especially in the acute setting.(1)
The most recent Society for Vascular Surgery guidelines have updated the classic Stanford classification for aortic dissections. Currently, they are classified as “type A” if the proximal tear occurs in the ascending aorta and “type B” if the proximal tear occurs distally to the innominate artery.(2) The latter are responsible for about 33% of these cases.(3) Another important classification is regarding the timing of presentation. Currently, ADs are classified as “hyperacute” if presenting in the first 24 hours after onset, “acute” if they present in the first 2 weeks, “subacute” between 2 weeks and 3 months, followed by “chronic” after this time.(4)
Type A aortic dissections are usually considered surgical emergencies (recommendation class I, evidence level B)(5), since a conservative treatment has historically been associated with unacceptable high complication and mortality rates, estimated as 1% for every hour in the first 48h after onset.(5)
In the case of TBAD, treatment depends on the clinical presentation and usually includes a conservative approach with medical therapy and monitoring in the case of “uncomplicated” TBAD. However, even in uncomplicated dissection, the natural history is alarming: 25-50% of them tend to develop late aortic related complications (rupture, aneurysmal degeneration and mal perfusion syndromes) with a dissection-related mortality of 38.2% at 4 years.(6)
When these dissections enter the chronic phase, the most relevant clinical problem is aneurysmal degeneration, which can involve the thoracic or abdominal aorta separately, but usually affects the whole thoraco-abdominal aorta starting at the proximal entry tear zone (commonly in the descending thoracic aorta just distally to the left subclavian artery) and ending at the aortic bifurcation or iliac arteries. It is estimated that 20-40% of patients with a chronic TBAD will eventually develop an aneurysm requiring an aortic repair, and overall, 25% of all descending and thoraco-abdominal aortic aneurysms originate following dissections.(7)
Aneurysm repair is recommended when a thoraco-abdominal aortic aneurysm (TAAA) has a diameter 60mm or if aneurysm growth occurs ate a rate 1cm/year.(8-10) Post dissection TAAA (PD-TAAA) are challenging, not only due the extension of aortic involvement, but also because one has two deal with two lumens, with the following varying origins of the visceral arteries, and with proximal and distal intimal tears, which might lead persistent false lumen perfusion.
Currently, various surgical techniques exist to deal with these PD-TAAA, namely open, endovascular or hybrid repair.(2,10)
Open repair is still considered by some to be the gold standard in these aneurysms, however, due to the high related morbidity and mortality, these should be performed in high-volume referral centers10 and in patients with acceptable surgical risk. Endovascular repair is technically challenging but with a lower short-term morbidity/mortality, however, long-term durability is still unknown.(10)
The aim of the study was to analyze the results with open and endovascular repair in patients with PD-TAAA treated at a tertiary university hospital.
Methods
A single-center retrospective cohort of prospectively collected data was performed. We included all consecutive patients with PD-TAAA who were submitted to repair, either open or endovascular, at Centro Hospitalar Universitário Lisboa Norte (CHULN) between January 2013 and May 2020. The timing of the study was chosen in order to include the experience of the department after the initial “learning curve”, regarding open and endovascular repair.
We analyzed the baseline data regarding age, sex, American Society for Anesthesiologists score (ASA), aneurysm diameter, cardiovascular risk factors [hypertension (defined as repeated blood pressure >140/90 or receiving anti-hypertensive), dyslipidemia (defined as elevation in LDL/total cholesterol or triglyceride levels or receiving lipid lowering medications), chronic kidney disease (defined as glomerular filtration rate <60), smoking and diabetes mellitus] and connective tissue disease diagnosis. We analyzed all the data regarding the procedure and pre and pos-operative CT-angiography (CTA) scans.
We divided our cohort according to the type of repair: Group 1 included patients submitted to endovascular repair and Group 2 included patients treated with an open repair and compared outcomes and characteristics between them.
At CHULN, decision to select patients for each surgical approach occurs regarding some baseline decision factors. Open repair is offered to patients who are young, have connective tissue disease, have an aortic or graft infection, as bailout for endovascular repair or if endovascular repair is not feasible due to anatomic reasons. In the remainder of cases, currently, endovascular repair with custom-made fenestrated/branched grafts is performed.
All patients were operated under general anesthesia. Endovascular repairs were performed in an angio suite, using the Philips Azurion system with fusion imaging. Open repairs were performed in a regular operating room with a multidisciplinary team, which includes dedicated anesthesiologists, cardiac and vascular surgeons, perfusionists, dedicated nursed and a neurophysiologist. In all cases, preventive protocol for spinal cord ischemia was implemented, including placement of a lumbar cerebrovascular fluid (CSF) drain with a target medullary pressure 10mmHg; target hemoglobin level 10mg/dL; peripheral O2 saturation >95%; mean arterial pressure >90mmHg and medullary perfusion pressure 80mmHg.
In patients submitted to open repair, intercostal artery revascularization was defined according to patency of these arteries on CTA assessment and guided by the variation in limb motor evoked sensitive and motor potentials measured intra-operatively.
In-hospital mortality was defined as the primary outcome. Secondary outcomes were divided as perioperative and follow-up outcomes. Perioperative secondary outcomes were acute kidney injury [defined increase in creatinine of e 0.3mg/dl or 1.5 x baseline value or oliguria (urine output <0.5mg/dl/h)], hospital infection (defined as any infection diagnosed by the attending clinician), medullary ischemia, length-of-stay in intensive care unit and length-of-stay in the hospital. Follow-up secondary outcomes were re-intervention rate endoleaks, visceral vessel patency and follow-up mortality.
Statistical analysis was performed using SPSS software, version 26 (Statistical Package for the Social Sciences, Inc., Chicago, IL), for Mac. Continuous variables are presented as mean (standard deviation-SD) if normally distributed and median (interquartile range - IQR) in not. Category variables are presented as frequency (percentage).
Mann-Whitney test was used when comparing continuous variables and Fisher’s exact test to compare categorical variables. All analyses were considered statistically significant if a two-tailed p-value < 0.05 was observed.
All patients provided informed consent before treatment and Institutional Review Board approval was waived for this study.
Results
Demographic data and aneurysm characterization (table I and II):
During the study, 21 patients were operated (15 men and 6 women), 8 were submitted to endovascular repair (Group 1) and 13 were submitted to open repair (Group 2). Mean age was 56 (SD: 15) and was significantly lower in patients in Group 2 [group 1: 68 (SD:11), group 2: 48 (SD:12), p=0.004]. Three patients in group 2 had connective tissue disease: 1 patient with Loyes-Dietz syndrome and 2 patients has Marfan syndrome.
Patients in group 1 had a higher baseline surgical risk [group 1: all ASA 3; group 2: mean 2 (DP:0.4), p<0,001]. There was no significant difference in cardiovascular risk factors (hypertension, dyslipidemia, chronic kidney disease or diabetes).
According to the Crawford classification modified by Safi11,12 for TAAA, 3 type I aneurysms, 14 type II, 3 type III and 1 type V aneurysms were treated (table II).
Surgical details:
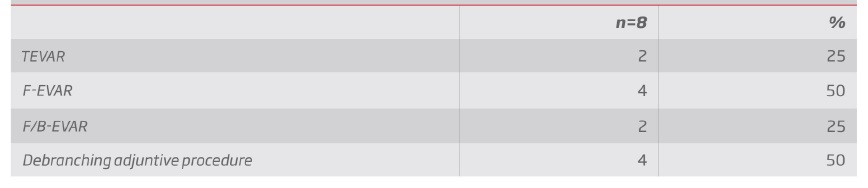
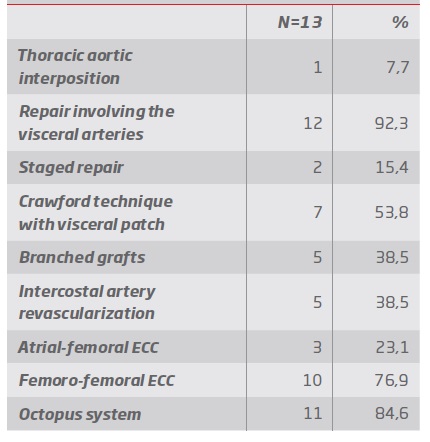
Group 1 - Endovascular repair (table III and IV): In group 1, 2 patients a TEVAR was performed, 4 were submitted to fenestrated repair with a custom-made graft and in 2 a custom-made graft with a combination of fenestrations and branched were used (see example in figure 1). In 4 cases, supra-aortic trunk debranching was performed in order to achieve an adequate landing zone (3 of zone 2 of Ishimaru, and 1 of zone 1).
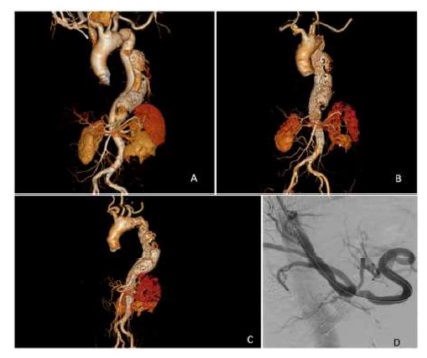
Figure 1 ACT-angiography 3D reconstruction of a type III thoraco-abdominal aortic aneurysm; BPost-operative CT-angiography 3D reconstruction of a type III thoraco-abdominal aortic aneurysm - following endovascular repair with a custom-made endograft (celiac trunk branch and fenestrations for the superior mesenteric artery and renal arteries); CLeft lateral view; DIntra-operative angiography showing celiac trunk catheterization through the false lumen through the aortic flap ostium fenestration.
Table I Patients demographic data. ASA: American Society of Anesthesiologists score; Md (IQR): median (interquartile range); X(SD): mean (standard deviation); NS: Non Significant
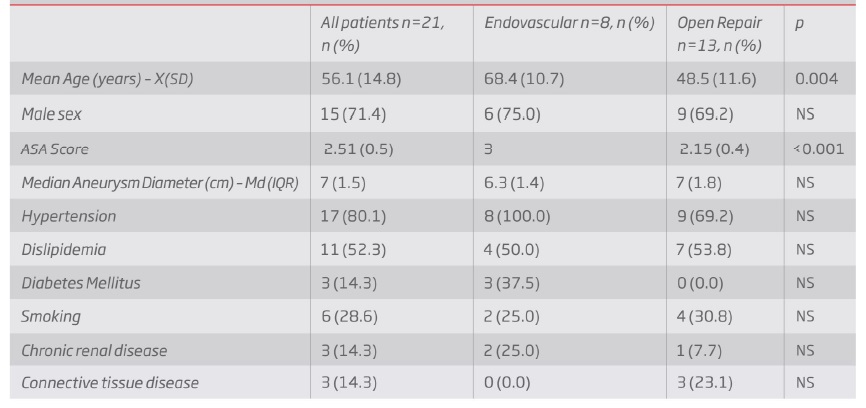
Table II Types of thoraco-abdominal aortic aneurysms according to the Safi modified Crawford Classification. TAAA - Thoraco-Abdominal Aortic Aneurysm
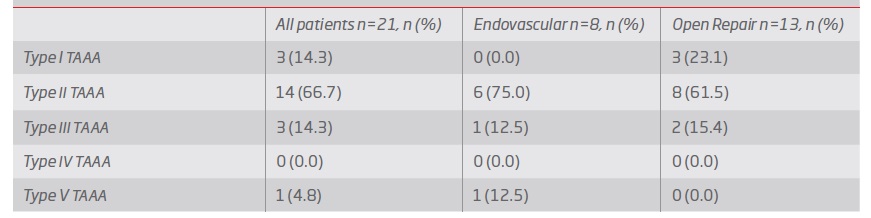
Table III Surgical details of patients submitted to endovascular repair. CSF: cerebrovascular fluid; TAAA: thoraco-abdominal aortic aneurysm; B/FEVAR: Branched and Fenestrated endovascular aortic repair; F: female; FEVAR: Fenestrated endovascular aortic repair; M: male; N: no; TEVAR: reparação endovascular de aneurisma da aorta torácica; Y:yes
Group 2 - Open repair (table V and VI): In group 2, one patient was submitted to thoracic aortic interposition with a Dacron graft (in order to repair the area that was most dilated) and in the other 12 patients, repair involved the visceral aorta. Repair was staged in 2 cases attending to spinal cord ischemia risk. Seven patients were repaired using the Crawford technique with a visceral patch (figure 2) and in 5 (including all patients with connective tissue disease) branched grafts were used. In 3 patients left heart atrio-femoral extracorporeal bypass with centrifuge pump was used, in the remainder femoro-femoral minimized extra-corporeal circulation (MECC) was used. The octopus system for visceral artery perfusion were used in 11 patients (table IV).
Table IV Endovascular repair. B/FEVAR: Branched and Fenestrated endovascular aortic repair; FEVAR: Fenestrated endovascular aortic repair; TEVAR: reparação endovascular de aneurisma da aorta torácica
All of these patients were operated using limb motor and sensory evoked potentials in order to monitor the development of spinal cord ischemia. In 5 patients intercostal artery revascularization was performed (2 dacron graft bypasses, 2 patches and 1 Safi loop graft).
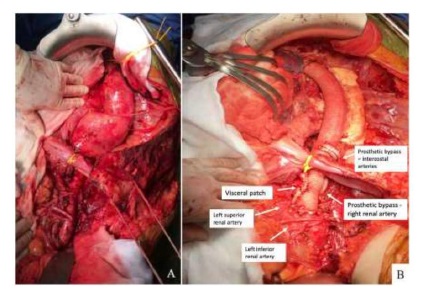
Figure 2 AIntra-operative photograph of a type II thoraco-abdominal aortic aneurysm (TAAA) following thoraco-phreno laparotomy with medial visceral rotation; BIntra-operative photograph of an open TAAA repair using the Crawford technique with a visceral patch (celiac trunk, superior mesenteric artery and left renal artery) with a right renal artery and intercostal bypass.
Comparison between Group 1 and Group 2:
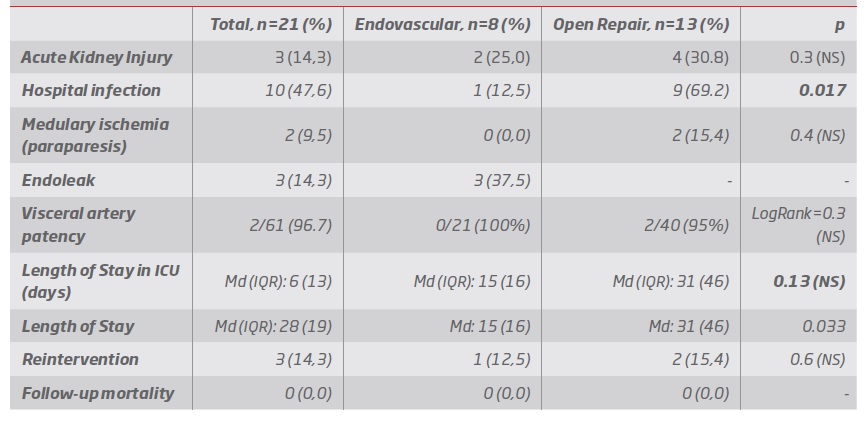
Primary outcome: In-hospital mortality was one e group 1 (12%) and two in group 2 (14%), LogRank=0.9 (table VII)
Secondary outcomes (table VIII):
Regarding perioperative outcomes - Length-of-stay was greater in group 2 [group 1: median 15 days (IQR:16) versus group 2: median 31days (IQR:46), p=0.033], and a tendency was observed towards longer stay in intensive care unit in group 2 [group 1: median 1.5 (IQR:13) versus group 2: median 6 (IQR: 11), p=0.13]. No significant differences were observed regarding spinal cord ischemia (group-1: 0%; group-2: 15%, p= 0.4). All spinal cord ischemia episodes occurring in group 2 (2 patients) were transitory and reverted fully after rehabilitation. In one of these cases we observed intra-operative diminished evoked potentials and in both cases we performed intercostal artery revascularization.
We observed a 25% rate of acute kidney injury in group 1 and a 31% rate in group 2, p=0.3. In no case was there need for dialysis, and all patients reverted to baseline creatinine values. A higher in-hospital infection rate was observed in group 2 (group 1: 12% (one cervical debranching graft infection); group 2: 69% [9 patients, 4 urinary tract infections, 2 pneumonias, one femoral cross over graft infection, 1 meningitis (secondary to CSF lumbar drain infection) and one case of pyelonephritis and peritonitis], p= 0.017.
Regarding follow-up outcomes - Re-intervention rate was 12% in group 1 (1 patient) and 15% in group 2 (2 patients), p=0.7.
In group 1, there was a need for an early re-intervention in a patient with an endoleak type IA, already submitted to a zone 1 debranching through a retroesophageal tunneling, with the need for proximal aortic wrapping. Unfortunately, this patient ended up developing a prosthetic-esophageal fistula with secondary graft infection (29 days after the index procedure). She was later submitted to a graft removal, coverage of the esophageal defect with sternocleidomastoid muscle and subcutaneous carotid-carotid bypass using great saphenous vein, however, the patient died in the first 24h of this last surgery with multiple organ failure secondary to septic shock.
In group 2, one patient developed surgical femoral wound infection (access zone for MECC) with extension to an old femoro-femoral bypass (performed 5 years prior due to limb malperfusion in the acute setting), with need for femoral graft explantation and bilateral femoral angioplasty using great saphenous vein patch. Another patient was submitted to a re-laparotomy, peritoneal lavage a left ureter repair due to intra-operative ureter iatrogenic injury in the primary repair. This injury had been initially treated with a double J catheter drainage and suturing but later developed urinary leak, pyelonephritis and peritonitis.
Table V Surgical details of patients submitted to open repair. (TAAA: thoraco-abdominal aortic aneurysm; SMA: superior mesenteric artery; CT: celiac trunk; RR: right renal; LR: left renal; IO: intra-ostial; MECC: Minimized Extra Corporeal Circulatory System; N: no; Y: yes; S: sensory; M: motor; CSF: cerebro spinal fluid; F:female; M: male)
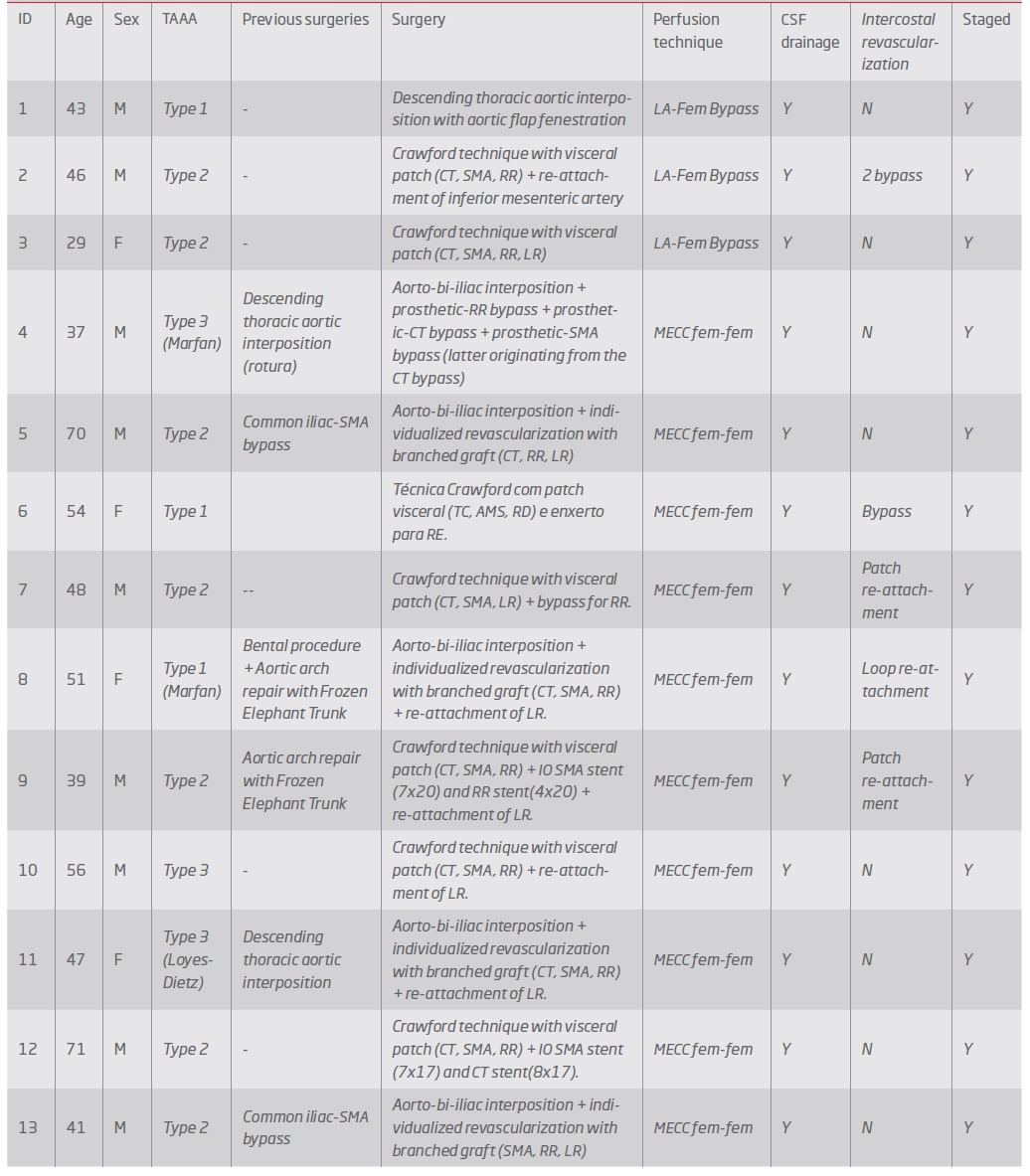
Table VI Open Repair. ECC: Extra corporeal circulation. Tratamento por cirurgia aberta convencional. CEC - Circulação extra-corporal
During follow-up [median 15 months (IQR:55)], no mortality was observed after discharge in both groups.
Target vessel permeability during follow-up was 100% for group 1 (n=21 target vessels) and 95% in group 2 (n=40 target vessels), LogRank=0.3. Two left renal artery bypasses occluded in group 2.
In group 1, late endoleaks rate was 25% [two patients with persistent type 2 (lumbar) endoleaks, excluding the already mentioned early type IA endoleaks). In no case was there aneurysm sac growth and both cases are being monitored conservatively with annual CTA alternated with abdominal aorta ultrasound.
Discussion
The present study included a cohort of 21 patients submitted to either open or endovascular repair of PD-TAAA.
Table VIII Follow-up data, including post-operative complications. ICU: intensive care unit; Md: median; IQR: interquartile range; NS: non-significant
Primary outcome was the difference in in-hospital mortality between both techniques, which was found to be non-significant (LogRank=0.9). It is important to notice, however, that although no difference was found regarding in-hospital mortality, patients submitted to endovascular repair were older (p=0.004) and had a higher ASA score (p<0.001). This difference in mortality might, therefore, be explained by the obvious selection bias in our study, were younger and more fit patients were chosen for open repair, since it is still considered by most as the goldstandard.(10) In addition, attending as this is a single center experience of a rare condition, the small number of patients might explain the lack of significant differences between groups (small number bias).
Regarding our secondary outcomes it is important to note that we had 2 patients with temporary spinal cord ischemia (paraparesis) in group 2, which reversed during follow-up and after rehabilitation. This devastating complication, in its permanent form (paraplegia), has been described in 1.1-2.3%(13,14) of patients submitted to open repair, with a lower prevalence described in endovascular repair, which was a similar finding in our study. The application of strict medullary ischemia prevention protocols might have contributed to our results. Protective measures, such as CSF drainage to ensure an adequate medullary perfusion and intra-operative neurophysiologic monitoring with motor and sensory evoked potentials in order to “guide” the surgeon’s decisions, have demonstrated to have a significant impact in prevention of spinal cord ischemia, reducing its incidence in some center from 25.0% to 2.1%.(13)
A complication which was frequent in our study was intra-hospital infection, which was higher in group 2, occurring in 9 out the 13 patients (69%), comparing to group 1 in which only one patient (12%) developed an infection. The higher invasiveness associated with open repair leading to larger wounds, longer time under mechanical ventilation, longer times with urinary catheter, with invasive pressure monitoring with an arterial line and with a central venous catheter, probably contributed to a higher infection rate in group 2. In fact, the majority of these infections were urinary tract infection and respiratory, probably related to these factors. In group 1, the case was an infection of a cervical debranching Dacron graft, which was tunneled posterior do the esophagus. We believe the continuous pulsating trauma of the graft led to the development of the fistula which caused the graft infection and ultimately lead the patient’s death. Since this case, we have refrained from using this tunneling approach, opting currently, for a subcutaneous anterior placement of the grafts.
Higher length-of-stay in the hospital and in the ICU has also been described in other centers when comparing open repair with endovascular repair in these patients.(14)
A target-vessel patency of 100% in group 1 (21/21) and 95% in group 2 (38/40) was found in this study for a median follow-up of 15 months (IQR:55). The absence of occlusion in group 1 might reflect the small number of cases described seeing as in higher volume centers the occlusion rate varies between 0% and 6.5%.(15,16) However, careful and detailed planning using custom-made endografts might also have contributed to our optimal results. Overall, branches were chosen when we anticipated that the target-artery ostium would be more than 1cm away from the main graft, and fenestrations when this distance was estimated to be <1cm.
In group 2, revascularized target-vessel patency rate was 95%, which is in line to has been found in other studies.(17) However, the fact that it occurred in two left renal artery bypasses, has led us to prefer, whenever possible, to re-implant the left renal artery directly in the aortic main graft, rather than to perform a bypass. This has already been described by Kahlberg et al following the experience of the Vascular Surgery department of San Raffael Hospital in Milan.(18)
Only three patients developed the need for re-intervention, 1 in group 1 (12%) and 2 in group 2 (15%), already described above. Studies reporting on endovascular procedures for these patients have described a re-intervention rate of 15-22.5%(16,19), which is slightly superior to what we found. In studies focusing on open repair, re-intervention rate has been described to be between 1-4.4%.(20,21) In endovascular repair, re-interventions are usually related with persistent or new endoleaks or continuous aneurysm growth. In this study, the smaller re-intervention rate might be explained to our conservative approach for type endoleaks with no aneurysm sack enlargement.
This study has some limitations. The small number of cases in our cohort limits the conclusions one may obtain from the study, and the selection bias limits comparative analysis. Given the small number of patients, baseline adjustments for comparison, such as propensity score matching, or regression models were not feasible. However, this selection bias reflects the heterogeneity of the so called “real-world”, with a cohort comprising young and good risk patients, and older and higher risk patients. In fact, for the reasons raised above the internal validity of the comparative analysis of the study is low. However, this analysis and its presentation is useful due to its external validity, which reflects the reality and decision process that occurs in a Vascular Surgery Department.
Other important limitation is the retrospective nature of the study and with the fact that it is a single center experience.
Conclusion
Endovascular repair of PD-TAAAs in this cohort allowed treatment of older patients with higher surgical risk, without an associated increase in mortality. Post-operative complications were more common in patients submitted to open repair, with a significant higher incidence of post-operative infection and longer length-of-stay. The ability to offer both techniques, endovascular and open, in patients with PD-TAAAs, made it possible to offer a patient-tailored approach to a cohort of patients with variable risks and anatomies, resulting in good results in both groups.















