INTRODUCTION
In the last three decades, soybean (Glycine max) has become one of the most produced and consumed grains worldwide, generating employment and income for thousands of people, with the United States as the largest world producer, followed by Brazil. Due to its adaptation and high productivity in different soil and climatic conditions, in the world G. max is cultivated in about 125 million ha with a production of 353 million tons in 2020 (FAOStat, 2022; Karges et al., 2022). In addition, due to the high global demand for food, soy is considered one of the main sources of plant protein. The highlight of these countries as the world's largest producers is related to access to various production technologies that allow increased productivity (Puchalsky et al., 2018).
However, in addition to machinery and products for soil preparation, disease and pest control in the crop, it is necessary to use quality seeds for sowing, as it is essential for good production. Soybean seeds are usually infested by fungi, among which can be highlighted the genera Aspergillus, Colletotrichum, Penicillium, Cladosporium, Fusarium, Chaetomium, Curvularia, Alternaria, Engyodontium, Guehomyces, Nigrospora, Diaphorte e Phoma (Yang et al., 2018; Escamilla et al., 2019; Oliveira et al., 2020).
Among the treatments performed on the seeds before sowing is the use of commercial fungicides, aiming to reduce the population of phytopathogenic fungi present in the seeds, which may reduce and compromise the establishment of the planting population in the field. The excessive use of these products has promoted the manifestation of resistant fungal populations, in addition to harmful effects to the environment, people and the seeds themselves and inhibition of the production of secondary compounds, requiring the search for efficient alternative treatments that do not have unhealthy effects and are easy access (Arasu et al., 2019; Cai et al., 2019). In addition to synthetic fungicides, the control of fungi in soybean seeds can be done through extracts of higher plant parts and even with sodium bicarbonate (Afolabi and Kareem, 2018; Krishnamurthy et al., 2008).
Thus, one can highlight the essential oils, volatile chemical compounds with low molecular weight, derived from the secondary metabolism of plants with antifungal activities (Arasu et al., 2019; Cai et al., 2019). The higher or lower biological activity of essential oils depends on their chemical constituents, such as limonene, citral, carvacrol, caryophyllene, elemene, eugenol, cineol, and furanodiene (Marin et al., 2018; Cai et al., 2019).
Among the species of plants producing essential oil are those of the genus Eucalyptus, which in studies have been shown to be effective in fungal control (Clavijo-Romero et al., 2019; Sabo and Knezevic, 2019). The essential oils of aromatic species, such as the genus Eucalyptus, are shown to be effective in the control and development of fungi that affect soybean seeds, such as Fusarium and Colletotrichum (España et al., 2017; Gakuubi et al., 2017). The antifungal activity of essential oils is also associated with the presence of terpenoid and phenolic compounds, which have a lipophilic character that makes the cell membrane of the fungus permeable, causing leakage of its contents (Knaak and Fiuza, 2010). The composition of essential oils depends on the plant genotype, the edaphoclimatic conditions where the plants are found, plant age, and extraction site, among other factors (Miranda et al., 2016).
In addition to efficiency in phytosanitary control, it is necessary that essential oils maintain the physiological quality of seeds, as some studies report the presence of compounds such as monoterpenes and terpenoids at high concentrations can harm seeds (Alipour et al., 2019). Thus, the objective of this study was to evaluate the efficiency of essential oils extracted from the leaves of the hybrids Eucalyptus tereticornis x Eucalyptus urophylla and Eucalyptus grandis x Eucalyptus urophylla on the sanitary and physiological quality of soybean seeds.
METHODOLOGY
Location, period of research development and chromatographic analysis of essential oils
The study was conducted at the Plant Science Laboratory of the Federal University of Piauí, Campus Professora Cinobelina Elvas (UFPI/CPCE) (09°04'28 "S and 44°21'31" W), from December 2018 to January 2019. Essential oils were provided by the organic chemistry laboratory of UFPI/CPCE, extracted from the leaves of the hybrids E. tereticornis x E. urophylla (HEtu) and E. grandis x E. urophylla (HEgu) located at the Experimental Farm of Alborada do Gurguéia, belonging to UFPI/CPCE, located in the municipality of Alvorada do Gurguéia (08°25'26 "S 43°46'37" W).
The oil of the HEtu was extracted at 9:00 am and the HEgu oil was extracted at 03:00 pm. Collection at different times of the day provides greater oil concentration and probability of finding different chemical compounds (Figueiredo et al., 2008). The choice of hybrids for essential oil extraction was due to the fact that there are few studies using Eucalyptus hybrid species to combat phytopathogenic agents, since the planting of hybrids compared to single species is greater in Brazilian territory (Assis and Mafia, 2007). In addition, the presence of major compounds already known in the species used, such as 1,8-cineole, β-pipene, α-pinene and p-cymene (cymol) are highly effective in controlling phytopathogenic fungi (Marin et al., 2018; Cai et al., 2019; Knaak and Fiuza, 2010).
The oils were extracted from the leaves, by the hydrodistillation technique, using Clevenger apparatus (Castro, 2008). Samples of 200.0 g of leaves were used with 500 mL of distilled water in a round-bottomed flask with a capacity of 1.0 L (Chaves et al., 2018). The hydrodistillation process lasted approximately two hours, from the boiling point of the water. After extraction, the oils were separated from the water by simple filtration using anhydrous sodium sulfate (Na2SO4). Then, stored in a refrigerator at -14 °C for further analysis of the chemical constituents.
The chromatographic analyzes were performed at the Central Laboratory of Analytical Chemistry of the Federal University of West Bahia in a gas chromatography apparatus with mass detector, model GCMS-2010, brand Shimadzu. The conditions programmed in the device were: DB-SMS model fused silica capillary column with 30 m long x 25 µm film thickness. The carrier gas used in this process was Helium with a constant flow of 1.0 mL.min-1. In the separation process, injection was used in split mode 1:20 with injector temperature 220 ºC at 3 ºC.min-1 and then 240-300 ºC at 3 ºC.min-1. For detection, the ion source at 300 ºC and the electronic impact technique at 70 eV were applied. Essential oils were filtered and diluted in methanol and analyzes were performed in triplicate using 10 µL of oil.
The identification of constituents was performed using the GCMS Postrum Analysis 4.42 software, by patterns in the spectral libraries (Wiley, NIST) and visual comparisons with mass spectra of substances found in the literature (Adams, 2007; Souza, 2009). The relative abundance (%) of the constituents was obtained from the area that each peak represented in the sample, and the compounds were organized according to their retention time.
Health test
Conducted in the laboratory at an average temperature of 25 °C and relative humidity ± 60%, in a completely randomized design (CRD), 2x5 factorial scheme, consisting of two essential oils extracted from the leaves of the hybrids HEtu and HEgu, with five concentrations (0, 0.5, 0.75, 1.0 and 2.0 µL.mL -1), totaling 10 treatments, with eight replicates of 25 seeds each, totaling 200 seeds per treatment according to the manual of Rules for Seed Analysis (RSA) (MAPA, 2009). The concentration 0 µL.mL-1 was used as a control, where the seeds were treated only with autoclaved distilled water (ADW). The seeds used were cultivar FTR 4179 IPRO, granted by a local company.
For greater effectiveness of the oils, they were dissolved in acetone (2 mL of acetone/µL of oil). A volume of 40 mL of each treatment was placed in 100 mL beakers, and the seeds were then allowed to emerge for five minutes. After treatment, the seeds were placed in gerbox boxes previously sterilized with alcohol 70 % containing three sheets of autoclaved germitest paper and moistened with ADW, with each gerbox functioning as a replicate. The volume in milliliters of ADW used to moisten the papers was obtained by multiplying its mass by 2.5. Then, the gerbox boxes were placed in BOD incubator (Biochemical Oxygen Demand), tcnal model-TE-341/240L, at 20 °C (optimal temperature for fungal growth) for seven days. After this period, seed health was evaluated, and the fungi were identified at the genus level according to their morphological characteristics observed under an optical microscope and identification key. The fungi were recorded only for the presence or absence in the seeds, counting the number of infested seeds.
Physiological quality of seeds
Developed according to the RAS, following the methodology in germitest paper rolls (MAPA, 2009). We used the CRD, 2 x 4 + 1 factorial scheme, consisting of two essential oils, previously described, with four concentrations (0.5; 0.75; 1.0 and 2.0 µL.mL -1), plus an additional control treated with ADW alone, with four replicates of 50 seeds per treatment, in germitest paper rolls moistened with ADW, at a ratio of 2.5 times the dry paper weight, stored in BOD at 25°C. After eight days, the following evaluations were performed:
-Germination percentage (GP): calculated by the formula GP = (N/A) x 100, where N = number of seeds germinated at the end of the test; A = number of seeds placed to germinate.
-Percentage of abnormal seedlings (PAS): PAS = (W/A) x 100, where P = number of abnormal seedlings and A = number of seeds placed to germinate. Abnormal seedlings were considered those that did not have the potential to continue their development and give rise to normal plants, even growing under favorable conditions, analyzing the following characteristics: Damage, deformations, deterioration, both of the shoot and the root system (MAPA, 2009).
-Percentage of dead seeds (PDS): PDS = (M/A) x 100, where M = number of dead seeds; A = number of seeds placed to germinate.
The count was performed daily and always at the same time, being considered germinated the seeds whose radicle length was greater than or equal to 2 mm, to calculate the germination speed index (GSI) according to the formula GSI = ∑ (ni/ti), where ni = number of seeds that germinated at time “i”, ti = time after installation of the test; i = 1 ( 8 days according to Maguire (1962).
Statistical analysis
The data were subjected to the Shapiro-Wilk normality test using the statistical program R and then analysis of variance. When significant, the means of the qualitative factors (essential oils) were compared by Tukey’s test at 5% probability, and those of the quantitative factors (concentrations) were subjected to regression analysis using SigmaPlot version 12.0 software.
RESULTS
Chromatographic analysis of essential oils
In the essential oil of HEgu, 97.15% of the constituents were identified, belonging to the classes of monoterpenes (α-pinene, limonene, 1,8-cineol) and terpenoids (p-cymeno [cymol], α-terpineol, α-acetate of terpinyl) (Table 1), with emphasis on the monoterpene 1,8-cineol, which represents 90.47% of the chemical composition of the oil (Table 1).
Table 1 Chemical composition of E. grandis x E. urophylla and E. tereticornis x E. urophylla hybrid essential oils
| Constituents | Content (%) | |
| E. grandis x E. urophylla hybrid essential oil | ||
| α-pinene | 1.03 | |
| p-cymene (cymol) | 1.25 | |
| Limonene | 0.82 | |
| 1,8-Cineol | 90.47 | |
| α-terpineol | 1.39 | |
| α-terpinyl acetate | 2.19 | |
| Total percentage of identified constituents | 97.15% | |
| Monoterpenes | 3 | |
| Terpenoids | 3 | |
| Esoteric | 0 | |
| Total of identified constituents | 6 | |
| E. tereticornis x E. urophylla hybrid essential oil | ||
| α-pinene | 5.66 | |
| β-pipene | 3.33 | |
| α-phellandrene | 3.34 | |
| p-cymene (cymol) | 25.94 | |
| 1,8-Cineol | 14.2 | |
| (( terpineol | 5.74 | |
| Terpinen-4-ol | 6.03 | |
| α-terpineol | 1.82 | |
| Bicycloelemene | 1.06 | |
| α-terpinyl acetate | 2.46 | |
| Bicyclogermacrene | 2.86 | |
| Spathulenol | 2.93 | |
| Total percentage of identified constituents | 75.37% | |
| Monoterpenes | 6 | |
| Terpenoids | 4 | |
| Sesquiterpenes | 2 | |
| Total of identified constituents | 12 | |
In HEtu oil, 75.37% of the constituents were identified, of which six belong to the monoterpene class (α-pinene, β-pipene, α-phellandrene, 1,8-cineol, terpinen-4-ol, α-terpineol), four to terpenoids ((( terpineol, terpinyl α-acetate, spathulenol, p-cymeno [cymol]) and two of the sesquiterpenes (bicycloelemene, bicyclogermacrene) (Table 1), highlighting the terpenoid p-cymeno (cymol) representing 25.94% of the composition (Table 1).
Sanitary quality of seeds
There was an effect of the isolated factors (essential oils and concentrations) and an interaction between them (essential oils x concentrations) for the genera Aspergillus and Fusarium, while for the genus Cladosporium, there was an effect only for the isolated factor concentrations, and for Penicillium, there wasn’t significant effect of the isolated factors or of the interaction between them (Table 2).
Table 2 Analysis of variance (mean squares and F test) for fungal genera found in soybean seeds treated with essential oils at different concentrations
| Source of variation | DF | Genera | |||
| Aspergillus | Fusarium | Cladosporium | Penicillium | ||
| Essential oils (EO) | 1 | 189.1125** | 357.0125** | 0.0500ns | 0.1125ns |
| Concentrations (C) | 4 | 47.91875** | 152.45625** | 3.78125** | 0.0625ns |
| EO x C | 4 | 40.08125** | 60.29375** | 0.08125ns | 0.05ns |
| Residue | 70 | 5.5482 | 6.1839 | 0.3321 | 0.0589 |
| CV (%) | 28.13 | 22.06 | 18.23 | 9.80 | |
** Significant at 1%; ns Not significant; CV: coefficient of variation; DF: Degree of freedom.
In the control of the genus Aspergillus, oil of HEgu was more efficient than oil of HEtu at concentrations of 0.5 and 1.0 µL.mL-1, and the values of 0.75 and 2.0 µL.mL-1 did not differ. This result was repeated for the genus Fusarium; however, the control was higher at concentrations of 0.5 and 0.75 µL.mL-1 and showed the same effect at concentrations of 1.0 and 2.0 µL.mL-1 (Table 3).
Table 3 Interaction unfolding of the essential oils of the E. tereticornis x E. urophylla (T x U) and E. grandis x E. urophylla (G x U) hybrids at each concentration level for the genera Aspergillus and Fusarium
| Oils | Concentrations (µL.mL-1) | ||||
| 0 | 0.5 | 0.75 | 1.0 | 2.0 | |
| Aspergillus | |||||
| T x U | 5.25 ± 3.93 a | 8.75 ± 1.75 a | 1.37 ± 0.97 a | 2.12 ± 1.19 a | 0.87 ± 0.65 a |
| G x U | 0.87 ± 0.87 b | 0.75 ± 0.75 b | 0.62 ± 0.47 a | 0.25 ± 0.44 b | 0.50 ± 0.63 a |
| Fusarium | |||||
| T x U | 12.00 ± 4 a | 11.50 ± 1.62 a | 5.50 ± 2.5 a | 3.12 ±1.37 a | 2.62 ± 0.87 a |
| G x U | 7.12 ± 1.62 b | 1.12 ± 0.91 b | 1.50 ± 0.75 b | 1.75 ± 1.37 a | 2.12 ± 1.69 a |
Original means followed by the same letter in the column do not differ by Tukey’s test at 5% probability.
However, there was no significant difference among the concentrations tested in any of the oils for the genus Aspergillus (Figure 1A), whereas for Fusarium, the increase in HEtu oil concentrations favored the reduction, minimizing approximately 78.17% of the population with the maximum concentration (2.0 µL.mL-1). For HEgu oil, the best response was obtained at a concentration of 0.5 µL.mL-1, which reduced approximately 84.27% of the population (Figure 1B).
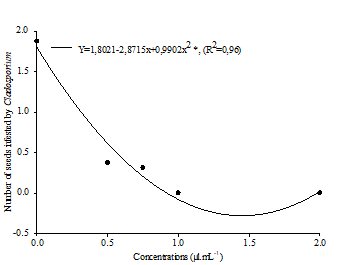


Figure 1 Effect of essential oils from E. tereticornis x E. urophylla (T x U) and E. grandis x E. urophylla (G x U) hybrids at different concentrations about the genera Aspergillus (A), Fusarium (B) and Cladosporium (C) found in soybean seeds. ** and * Means that differ at 1 and 5% probability, respectively, e ns Not significant.
Regarding the genus Cladosporium, there was a significant difference only between concentrations, adjusting to the quadratic polynomial model, totally reducing the population at concentrations of 1.0 and 2.0 µL.mL-1 (Fig. 1C).
Physiological quality of seeds
The behavior of the isolated factors and the interaction between them and the additional control were the same for all physiological variables analyzed (percentage of germination, abnormal seedlings and dead seeds and speed and germination index), with a significant difference only for the isolated factor, essential oils, and interaction of the factors with the additional control (Table 4).
Table 4 Analysis of variance (mean squares and F test) for the physiological variables germination percentage (GP), percentage of abnormal seedlings (PAS), percentage of dead seeds (PDS) and germination speed index (GSI) in seeds. of soybean seedlings treated with essential oils at different concentrations
| Source of variation | DF | GP | PAS | PDS | GSI |
| Essential oils (EO) | 1 | 864.61** | 43.82** | 74.40** | 778.81** |
| Concentrations (C) | 3 | 178.79ns | 2.74ns | 0.11ns | 4.71ns |
| EO x C | 3 | 227.46ns | 2.38ns | 0.41ns | 4.32ns |
| Cont. Ad x (EO x C) | 1 | 92.5*** | 0.85*** | 12.17*** | 66.01*** |
| Treatment | 8 | 1.292.50 | 7.40 | 10.46 | 130.99 |
| Residue | 24 | 155.54 | 1.01 | 0.59 | 4.50 |
| CV (%) | 15.50 | 35.20 | 27.10 | 8.57 |
*** Significant at 0%, ** Significant at 0.1%, ns Not significant, CV: coefficient of variation; DF: Degree of freedom, Cont. Ad: Additional control.
Despite the higher efficiency of the oil of HEgu in sanitary quality, HEtu oil was higher for all physiological variables evaluated (Table 5). The oil of HEgu at concentrations of 0.5, 0.75, and 1.0 µL.mL-1 negatively affected germination, and at the concentration of 0.5 µL.mL-1, it promoted a significant increase in the number of abnormal seedlings compared to the control (Table 5). In these last two variables, none of the tested concentrations of the oil of HEtu significantly changed when compared to the control.
The oil of HEgu at all concentrations tested promoted a significant increase and reduction in the percentage of dead seeds (PDS) and germination speed index (GSI), respectively, compared to the control, while the HEtu oil did not significantly influence the PDS at any of the concentrations, however, at concentrations of 0.75 and 1.0 µL.mL-1, the GSI was reduced significantly compared to that of the control (Table 5).
Table 5 Germination percentage (GP), percentage of abnormal seedlings (PAS), percentage of dead seeds (PDS) and germination speed index (GSI) of soybean seeds treated with essential oils of the hybrids E. tereticornis x E. urophylla (T x U) and E. grandis x E. urophylla (G x U)
| Oils | Concentrations (µL.mL-1) | Mean | |||
| 0.5 | 0.75 | 1.0 | 2.0 | ||
| GP (%) | |||||
| T x U | 97.00 ± 2 | 97.00 ± 1 | 98.00 ± 2 | 95.50 ± 1.5 | 96.87 a |
| G x U | 52.00 ± 22*** | 63.00 ± 9.5** | 64.50 ± 10.7** | 76.50 ± 2.5 | 64.00 b |
| Additional control | 92.00 ± 5 | ||||
| PAS (%) | |||||
| T x U | 3.00 ± 2 | 2.00 ± 2 | 1.50 ± 1.5 | 2.50 ± 2.5 | 2.25 b |
| G x U | 27.50 ± 14** | 18.50 ± 4.2 | 17.00 ± 6 | 6.00 ± 4 | 17.25 a |
| Additional control | 7.00 ± 3 | ||||
| PDS (%) | |||||
| T x U | 0.00 ± 0 | 1.00 ± 1 | 0.50 ± 0.7 | 2.00 ± 1 | 0.87 b |
| G x U | 20.50 ± 8*** | 18.50 ± 5*** | 18.50 ± 5*** | 17.50 ± 2*** | 18.75 a |
| Additional control | 1.00 ± 1.5 | ||||
| GSI | |||||
| T x U | 31.17 ± 0.7 | 28.52 ± 1** | 29.33 ± 1.1* | 29.71 ± 1 | 29.68 a |
| G x U | 19.25 ± 3.2*** | 19.04 ± 1.3*** | 19.67 ± 1.9*** | 21.29 ± 0.7*** | 19.81 b |
| Additional control | 33.00 ± 0.7 | ||||
Means followed by the same letter in the column do not differ statistically from each other by Tukey’s test at 5% probability. *, ** and *** Means that differ statistically from the additional control by the Dunnett test at 5, 1 and 0% probability levels, respectively.
DISCUSSION
Essential oils have been used, among other applications, in the control of microorganisms due to their chemical characteristics, which in a way represents an extension of the activity they exert in plants, protecting them against the attack of pathogens such as fungi (Clavijo-Romero et al., 2019; Papoutsis et al., 2019; Sabo and Knezevic, 2019). The chemical composition and the content of each constituent in common among the oils may vary according to the plant genotype, cultivation area, collection conditions, storage and edaphoclimatic conditions (Miranda et al., 2016).
The antifungal properties of essential oils are associated with their composition because they consist of the classes of monoterpenes, terpenoids, benzenes, hemiterpenes, and sesquiterpenes, which give them fungicidal activity (Mikola et al., 2017; Jaime and Ferrer, 2018). Generally, the suppressive action of essential oils on the pathogen occurs due to the rupture of the plasma membrane, granulation of the cytoplasm and the inhibition and/or production of inter- and extracellular enzymes (Knaak and Fiuza, 2010).
The inhibitory potential of essential oils depends on their chemical constituents, such as limonene, citral, carvacrol, caryophyllene, elemene, eugenol, 1,8-cineol, and furanodiene (Marin et al., 2018; Cai et al., 2019). The monoterpenes limonene and 1,8-cineol are present in the HEgu oil, while only 1,8-cineol is present in the HEtu oil, explaining the greater efficiency in fungal control of the HEgu oil. The major constituents of essential oils of species of the genus Eucalyptus vary according to the species, with the monoterpene class (1,8-cineol, Citronellal, γ-terpinene, cineol and α-pinene) being the most common (Silva et al., 2006), and the constituents 1,8-cineol and α-pinene were found in both oils.
The difference in concentrations and compounds identified for the two Eucalyptus hybrids used may be related to the edaphoclimatic conditions in the collection region. Since these hybrids are part of experimental plantations, there is no research in the collection region, addressing the influences of climate on the development of secondary compounds of these species.
Most of the constituents identified in the essential oils of this study (mainly 1-8 cineole, α-pinene, p-cymene, β-pinene) are in agreement with those found in the literature and in other pure Eucalyptus species such as: E. globulus, E. camaldulensis, E. saligna, E. deglupta, E. cinerea, E. blakelyi, E. maidenii (Dhakad et al., 2018).
The nonpolar characteristics of the chemical components of the essential oils give them a fungitoxic effect. Due to their nonpolar nature, these substances can interact with the cell membrane of fungi, causing their swelling and, consequently, resulting in the leakage of cellular materials promoting their death. Chemical constituents can also cross the cell membrane of the pathogen, delaying the synthesis of ergosterol, a hormone responsible for the cell vitality of fungi, causing cell death (Goulart et al., 2015). The class of terpenoids and sesquiterpenes has a lipophilic character and makes the cell membrane of the fungus permeable, causing leakage of its contents (Knaak and Fiuza, 2010).
The best results at the highest concentrations (1.0 and 2.0 µL.mL-1) can be explained by the volatilization of the substances present in the oils. When used at lower concentrations, rapid volatilization of their chemical constituents can occur, preventing the synergy of trace elements (present in lower concentrations) with the major elements (Nguyen et al., 2018). The essential oils of aromatic plants are mostly composed of terpenes, which have functions as inhibitors of germination and protection against predators, among others (Pawlowski et al., 2013). In addition to terpenes, monoterpenes are common, and some of their constituents can become toxic at high concentrations (Rahman et al., 2019; Yuan et al., 2019).
Monoterpenes are major components of essential oils of a large number of species and have been reported as effective allelochemicals due to the toxic effect on seed germination (Rahman et al., 2019; Yuan et al., 2019), which explains the negative effects of HEgu oil on germination, as 92.32% of the constituents found in this oil belong to this class, whereas HEtu oil represents only 34.38% (Table 1), which does not harm physiological quality. The monoterpene 1,8-cineol, which represents 90.47% of the constituents of HEgu oil and 14.2% of HEtu, negatively affects germination by decreasing both the mitotic index and cell elongation. In addition, it inhibits starch synthesis, promotes oxidative stress and lipid oxidation and affects mitochondrial respiration (Qiu et al., 2010; Yoshimura et al., 2011).
The allelopathic effect of essential oils may often not occur on germination; however, it can occur on the germination speed index (Rosado et al., 2009), corroborating with the result observed with the use of the oil of HEtu hybrid at concentrations of 0.75 and 1.0 µL.mL-1 (Table 5). The difference between oils even in the zero concentration for the genera Aspergillus and Fusarium may have occurred due to the difficult control of the infesting fungi, despite the asepsis of the site and the gerboxes.
CONCLUSIONS
Despite the greater efficiency in phytosanitary control in soybean seeds, the E. grandis x urophylla hybrid oil negatively affects the physiological quality of the seeds.
The essential oil of the hybrid E. grandis x tereticornis at a concentration of 2.0 µL.mL-1, in addition to being efficient in phytosanitary control, maintains high physiological quality of the seeds and can be used as one of the alternative treatments in seeds of soybean. However, even with positive results, further experiments are needed, using different concentrations of essential oils from the hybrids of this study.














