Introduction
Dryocosmus kuriphilus Yasumatsu (Hymenoptera: Cynipidae, Cinipini), also known as chestnut gall wasp, is original from mainland China and has spread throughout all the main chestnut growing regions, being today one of the main pests that affect chestnut trees (Brussino et al., 2002; Rieske, 2007; EFSA, 2010).
This insect’s life cycle starts in early summer, with the adult’s emergence, which, given their thelytoky nature do not require mating, and are able to immediately start oviposition. The oviposition occurs exclusively inside the chestnut tree’s buds, which makes this parasitism obligatory for this insect to complete its life cycle (Everatt, 2015; Santos et al., 2017). The adults have a short life span of approximately 10 days, during which they can lay as many as 100 eggs in groups of 3 to 5 per bud (EPPO, 2005). The eggs take about 30 to 40 days to hatch, but the larvae will stay dormant and inconspicuous during winter, in the buds of chestnut trees. In the beginning of the following spring, when the chestnut tree starts its vegetative activity, they wake up and start inducing the gall formation, of which they will feed and stay inside the gall for approximately 30 days. After that, pupation starts, which will last until the beginning of summer, when the insects emerge (Cooper & Rieske, 2010; Santos et al., 2017).
D. kuriphilus can cause 50% to 70% loss of tree’s chestnut and chestnut lumber yield and can reach a maximum of 80% in cases of extreme infestation (EPPO, 2005; Gehring et al., 2018; Matošević et al., 2016). As such, controlling D. kuriphilus populations is a serious challenge for chestnut growers around the world. The ecology of this insect makes the use of conventional pest control methods, like chemical pesticides useless, as it spends the majority of its life cycle (egg, larval and pupa phase) protected inside the buds and gall. Therefore, biological solutions must be found, the most promising ones involving the use of D. kuriphilus natural enemies, the parasitoid chalcid wasps. While various chalcid wasps found around the world proved capable of parasitizing D. kuriphilus galls, with various levels of success depending on region and species, their combined effort is usually not enough to contain the gall wasp’s infestations (Moriya et al., 1989). The exception is mainland China, where D. kuriphilus populations are usually kept low enough to allow for chestnut tree growth by the local parasitoid communities. Amongst them, Torymus sinensis Kamijo has shown the highest potential to be successfully used as a biocontrol agent for the chestnut gall wasp.
Torymus sinensis Kamijo is a parasitoid wasp from the Torymidae family (Hymenoptera: Chalcidoidea), specialized in parasitizing gall wasps, specially D. kuriphilus. T. sinensis is univoltine, with one generation per year, and haplodiploid, giving birth to haploid males from unfertilized eggs and diploid females from fertilized ones (Quacchia et al., 2014a). Females lay their eggs in early spring, inside the larval chambers of recently formed D. kuriphilus galls, usually one egg per host larva. If multiple eggs are laid in the same chamber, only one of them will grow to adulthood due to cannibalism among the hatched larvae (Ferracini et al., 2015). The larvae hatch quickly from the eggs and immediately start to feed from their host. At spring’s end, they have already consumed the host, and entered a dormant state until late winter, when pupation starts. They finally emerge in early spring, synchronous with the sprouting of chestnut trees and the formation of D. kuriphilus galls (Ferracini et al., 2015; Gibbs et al., 2011; Matošević et al., 2016).
T. sinensis is capable of prolonged diapause, meaning that a small percentage of individuals (< 5%) stay dormant for one year. This strategy is rarely found in oak gall parasitoids, presumably because most of them are polyphagous and have a vast array of host species, which makes oviposition a low risk, and representing a high success effort (Stone et al., 2002). In contrast, T. sinensis has only one host, and if the availability of D. kuriphilus galls drops for one year and hinders the oviposition success of an entire generation, a small cohort of diapausing individuals will provide a demographic back up for the next year, increasing the likeness of these parasitoids surviving temporary extinctions of the host population. As D. kuriphilus populations fluctuate in its natural habitats in mainland China, it is common to observe heavy infestations for 2 to 3 years, followed by up to 10 years of small populations with mild infestation (Quacchia et al., 2014a; Stone et al., 2002). This cyclical fluctuation and the narrow host range are probably the major factors for the development of prolonged diapausing in T. sinensis (Quacchia et al., 2014a).
Recent releases of T. sinensis in affected countries such as Japan, USA, and several European countries, proved to be effective in keeping D. kuriphilus populations below the economic loss threshold (< 30% branch infestation) after periods of 6 to 18 years, depending on the region (Matošević et al., 2016; Moriya et al., 1989; Murakami et al., 2001; Quacchia et al., 2008; Quacchia et al., 2014b; Rieske, 2007). Data from 2010 showed that the time required to achieve this threshold can be reduced with successive T. sinensis releases, and by increasing its life cycle synchronization with that of D. kuriphilus. This can be achieved by keeping the T. sinensis individuals at lower temperatures when necessary, which also extends its longevity (Quacchia et al., 2008; EFSA, 2010). Recognition of T. sinensis prolonged diapause is important for its use as biocontrol. While traditional management of chestnut orchards usually involves the removal and destruction of pruned shoots and fallen leaves in winter, this might be delayed for two years to allow parasitoid individuals having an extended diapause to complete their development (Quacchia et al., 2014a).
To better understand the ecology of the parasitoid T. sinensis and to assess its potential of in the control of the chestnut gall wasp, this work aims to study T. sinensis ability to establish a population, it’s dispersion ability and the effect of the release density on the populations of D. kuriphilus in the Portuguese region of Minho.
1. Materials and methods
Three orchards were selected to conduct the biological treatment in the Minho region, with chestnut trees seriously infected by the wasp (presence of galls in 51-80% of shoots), according to the “National Plan of Action for the Control of the insect Dryocosmus kuriphilus Yasumatsu Chestnut Gall Wasp”. The chestnut trees present in each site were identified with the help of their respective owners and the guide for chestnut varieties from the Instituto Nacional dos Recursos Biológicos (INRB) (Costa et al., 2008).
To evaluate the effect of the number of T. sinensis released in the parasitism rates and population establishment, different quantities of insects were released in each of the sites. The release of T. sinensis was divided in release units, each composed by a total of 120 females and 70 males, divided between 10 Falcon tubes. Each Falcon tube also contained a honey drop to provide the insects sustenance.
Three release units were applied in Vale do Neiva (T1), two release units in Cossourado (T2) and one in Fragoso (T3) (table 1).
The three release sites were regularly monitored to assess the phenological state of the chestnut trees, and establish when the phenological state “D” (when the leaves sprout and starts developing) was going to be present, as recommended by the “National Plan of Action for the Control of the insect Dryocosmus kuriphilus Yasumatsu Chestnut Gall Wasp” to be the most effective time to perform the releases, as to increase the chances of successful oviposition.
The releases were made on the 26th April 2018, when the chestnut trees were displaying the phenological state “D”.
The galls formed in the spring of 2018, which were potentially parasitized by the released T. sinensis were collected in January 2019. To evaluate the dispersion capacity of T. sinensis, galls were also collected from sites at approximately 200m distance from the original release sites. These secondary sites were comprised by a smaller group of chestnut trees (≈15) and each had a direct line of sight to their counterpart site, with trees from different species doting the way. In all the sites, all the chestnut trees present showed signs of D. kuriphilus infestation.
For each site and respective 200m site, 200 galls were collected and kept in labelled ziplock bags. The galls were then cleaned of leaves, branches and insects, and stored in emergence boxes. Two boxes per site were used, each one stored with 100 galls. The emergence boxes were stored at room temperature, out of direct sunlight. The adult emerging insects present in the Falcon tubes were regularly collected and stored in the laboratory, in Eppendorf tubes with 70% ethanol. The insects were then identified to species or genus level with a stereo microscope. After identification, the insects were quantified and again maintained in Eppendorf tubes with 70% ethanol and stored according to genus for future observation. The boxes were opened in July 2019 for gall cleaning and inspection, and to collect emergent insects which weren’t able to reach the Falcon tubes. All collected data was recorded for later statistical analysis.
Assessment of the parasitism rates due to Torymus sinensis
To quantify the mortality rates caused by the action of T. sinensis, about 80 extra galls were collected along with the previous collections and kept separately in labelled ziplock bags. The galls were then brought to the laboratory and stored in closed glass jars to avoid the escape of emerging insects and mold growth. To assess the parasitism rates due to T. sinensis, 50 galls per sample and per site were dissected under a stereo microscope, and each larvae or adult of T. sinensis found was counted. Also, around 50 galls were taken from the emergence boxes to compliment the previous samples. The parasitism rates were calculated by relating the number of T. sinensis specimens found with the total number of chambers in the sample, as follows:
Parasitized chambers without T. sinensis were also counted to assess the natural parasitism rates (due to the native parasitoid species) in these sites. For each site, the dissected galls were given a number and grouped in groups of 10 galls, to create replicate values. Due to the different amounts of T. sinensis released in the different sites, another graph was made to remove this variable, where the average rates were divided by the number of releases made: T1/3, T2/2 and T3/1. All collected data was recorded for later statistical analysis.
Assessment of the parasitism rates due to T. sinensis on the second year after release (2019)
To quantify the mortality rates caused by the action of T. sinensis on the second year after release, new collection were made in September of 2019. About 80 galls were collected on the 30th of September and kept in labelled ziplock bags. The galls were then brought to the laboratory and stored in closed glass jars to avoid the escape of emerging insects and mold growth. To assess the parasitism rates due to T. sinensis, 50 galls per site were dissected under a stereo microscope, and each chamber containing T. sinensis was counted. Due to the date of collections, all the T. sinensis found were still in their larval state. The number of chambers containing T. sinensis was divided by the total number of chambers in the sample to obtain the parasitism rates, using the same methods as with the previous year samples. Parasitized chambers without T. sinensis were also counted to assess the natural parasitism rates in these sites.
The statistical analysis of the collected data was carried out with the help of the software R (R Core Team 2018, version 1.1.442) and Graphpad (Graphpad Prism version 6.01). The data samples were subjected to the D’Agostino-Pearson and Shapiro-Wilk normality tests and transformed to y= y when the normality requisites weren’t verified. The data was then subjected to analyses of variance, such as the parametric ANOVA, the Dunn’s and the Sidak’s multiple comparison tests and t-test.
2. Results
For each site, two samples of 50 galls each were dissected (50 from the original collections and 50 from the emergence boxes), for a total of 100 galls per site. In the site T1, a total of 301 gall chambers were observed, 117 of which were classified as parasitized and 12 T. sinensis individuals were found. In site T1200m 330 gall chambers were analysed, 125 of which were parasitized and 11 T. sinensis individuals were found. A total of 367 gall chambers were analysed in site T2, 140 of which were classified as parasitized and 13 T. sinensis individuals were found. In site T2200m 358 gall chambers were analysed, 132 of which were parasitized and 15 T. sinensis individuals were found. In the site T3, 463 gall chambers were analysed, 166 of which were classified as parasitized and 6 T. sinensis individuals were found. Finally, in site T3200m, 285 gall chambers were analysed, 108 of which were classified as parasitized, and only one T. sinensis specimen was found (Table 2).
The percentage of T. sinensis found was similar in sites T1 (3.89%), T1200m (3.52%), T2 (3.71%) and T2200m (4.19%), with only the sites T3 (1.3%) and T3200m (0.26%) showing the lowest parasitism rates. When divided by the number of release units applied to each site, the parasitism rates found were the highest in sites T2 (1.85%) and T2200m (2.10%), followed by sites T1 (1.30%), T3 (1.30%) and T1200m (1.20%), and with the lowest rate still sowing in site T3200m (0.26%).
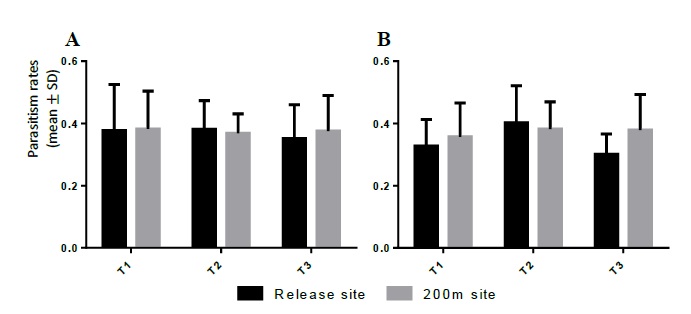
The raw data sets of the parasitism rates (A) and of those rates divided by the respective release effort (B) were analysed by regular ANOVA (A: F = 3.372, P = 0.010; B: F = 2.172, P = 0.071) and Tukey’s multiple comparison test, with significant differences found, in both analysis, between the 200m distance T2 site and the 200m distance T3 site (a: MD = 0.017; b: MD = 0.018) (figure 1).
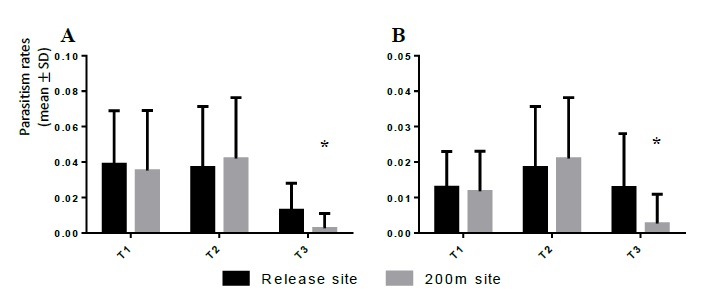
Figure 1 Parasitism rate (average ± standard deviation) of Torymus sinensis in the release sites and at 200m distance. (A) Average parasitism rates; (B) the same rates divided by release effort (T1/3; T2/2; T3/1).
For the analysis of the parasitism rates in the second year after the releases (2019), 50 galls were dissected for each site. In site T1, 131 gall chambers were analysed, 46 of which were classified as parasitized and no T. sinensis individuals were found. In site T1200m 113 gall chambers were analysed, 42 of which were parasitized and also no T. sinensis individuals were found. Site T2 had a total of 105 gall chambers analysed, 42 of which were classified as parasitized and one T. sinensis was found. In site T2200m 127 gall chambers were analysed, 50 of which were parasitized and 16 T. sinensis individuals were found. In the site T3, 110 gall chambers were analysed, 33 of which were classified as parasitized and 3 T. sinensis individuals were found. Finally, in site T3200m, 77 gall chambers were analysed, 29 of which were classified as parasitized, and 2 T. sinensis specimens were found (Table 2).
One year after the releases, the parasitism rates decreased in sites T1 (0%), T1200m (0%) and T2 (0.8%) but increased in sites T2200m (13%) T3 (2.49%) and T3200m (2.54%). The largest change occurred in site T2200m, where the parasitism rate increased from 4.19% to 13%, a change of almost 10%. When related with the number of releases, the site T2200m still showed by far the highest parasitism rates.
The parasitism rates from the second generation of galls collected in the release sites was calculated and analysed using the same methods from the previous year. The raw data sets of the parasitism rates (figure 2A) and of those rates divided by the respective release effort (figure 2B) were analysed by regular ANOVA (a: F = 16.11, P < 0.001; b: F = 7.495, P < 0.001). In the data set of figure 2A, the Tukey’s multiple comparison test found significant differences between the 200m distance T2 site and all the other sites (Tukey’s MDs: vs T1: -0.130; vs T1 200m: -0.130; vs T2: -0.122; vs T3: -0.105; vs T3 200m: 0.105). In the data set of figure 2B, the Tukey’s multiple comparison test showed significant differences between the 200m distance T2 site and all the other sites except for the 200m distance T3 site (Tuckey’s MDs: vs T1: -0.065; vs T1 200m: -0.065; vs T2: -0.061; vs T3: -0.040).
Table 2 Number of gall chambers observed, parasitized chambers and T. sinensis found, and percentage of T. sinensis in the three release sites and their respective 200m site.
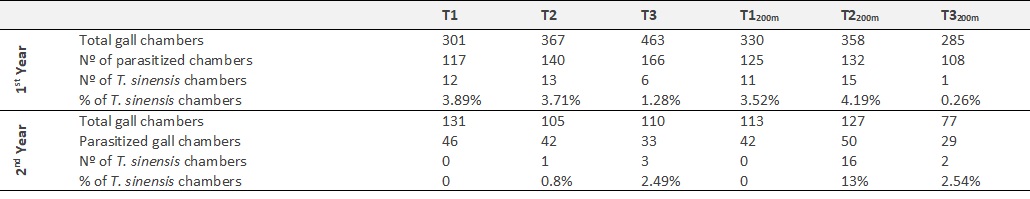
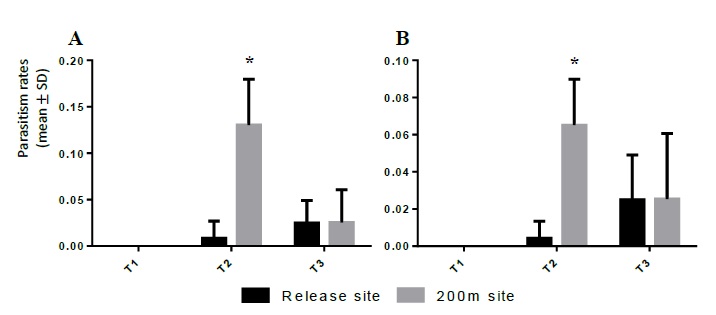
Figure 2 Parasitism rate (average ± standard deviation) of Torymus sinensis in the release sites and at 200m distance, in the second year after the insect’s releases; (A) Average parasitism rates; (B) the same rates divided by release effort (T1/3; T2/2; T3/1).
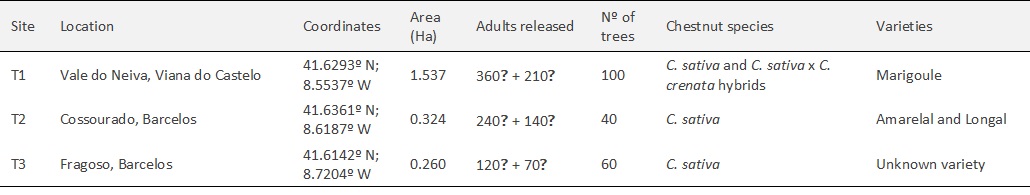
The total parasitism rates, caused by T. sinensis plus the native parasitoid communities, were calculated in the likeness of the previous ones. The chambers containing T. sinensis were classified as parasitized as well. The average parasitism rates varied between the lowest value of 35% in site T3 and the highest one of 38% in site T1200m, in the first year after release (2018) (figure 3A). In the second year after release (2019), the average rates varied between 30% in site T3 and 40% in site T2 (figure 3B).
The raw data sets of the parasitism rates in the first (a) and in the second year (b) after the T. sinensis releases were analysed by regular ANOVA (a: F = 0.109, P = 0.989; b: F = 0.731, P = 0.607) and Tukey’s multiple comparison test, with no significant differences found between the sites.
3. Discussion
In the countries affected by the chestnut gall wasp, D. kuriphilus, the introduced parasitoid T. sinensis has proven multiple times to be an effective method of controlling the pest to manageable levels and so far, it seems to be the best method available to achieve this. In the three sites used for the experimental releases of T. sinensis, galls were collected to assess the efficiency of this parasitoid.
The parasitoid T. sinensis usually takes a minimum of 3 to 5 years since its first releases to develop a stable population in the sites where it is introduced (Quacchia et al., 2008). Therefore, in the first year after the first releases, it is expected to find small populations and low parasitism rates (Borowiec et al., 2018). In the 3 sites sampled in this study, the parasitism rates in the first year after release were inside the range described in literature for releases of the same size in other European countries, such as France, Italy, Slovenia, Hungary and Croatia. The number of insects released can also influence the population size in a direct way, but only in the first years. The most effective long term method seems to be annual releases of 100 to 200 females (Borowiec et al., 2018; Cascone et al., 2018; Matošević et al., 2016; Quacchia et al., 2008). Despite this relation between the number of insects released and the following populations in the early years, the opposite can also occur, as observed in sites T1 and T1200m. In these sites, despite having shown high T. sinensis’ parasitism rates in the first year and having the highest release effort, relatively with the other sites, no T. sinensis were found in the observed galls. This could mean that the original release was not enough to establish a population, or a lack of synchronism between T. sinensis and D. kuriphilus emergence, which could complicate the establishment of a steady population (Quacchia et al., 2008). This site was the largest considering the area and also the one with the largest chestnut trees, which could also play a significant role in the collections made. Only galls up to 3 meters high were collected, so a smaller proportion of galls were sampled compared to the total in the site. In this way, it is possible that the parasitoid was still present in these sites but was not found in the collected samples. More extensive sampling would be required to properly access the evolution of T. sinensis population in both these sites.
In contrast, the sampling sites T2 and T2200m showed in the first year after release, the largest population of T. sinensis of all sites, with site T2200m showing the biggest growth between the first and second years. Similar results have already been found in France and Italy, where the smallest release produced the biggest population (Borowiec et al., 2014, 2018; Colombari & Battisti, 2016). In this particular case, the smaller size of the chestnut trees and the higher relation between the number of T. sinensis released and trees in site T2, are possible explanations to the high parasitism rates found here. Despite such rapid growth observed in T2200m, it is still well in the range described in France and Italy for a population of T. sinensis two years after the initial release (Borowiec et al., 2018; Gehring et al., 2018; Quacchia et al., 2014b).
In the T3 and T3200m, the area, relative age and size of trees sits between the other four sites, but T3 was the site which received the lowest release effort, which could be the reason for the low parasitism rates found here in the first year. However, in both these sites there was a increase of the parasitism rates observed in the second year. This growth means that under the right conditions, T. sinensis is able to successfully parasitize D. kuriphilus galls and develop a strong second generation capable of migrating at least up to 200m to other chestnut orchards in the span of one year.
When analysing the galls collected in each site for T. sinensis, the native parasitoid chalcid wasps found were also counted, to assess if some displacement had occurred. The parasitism rates due to the native parasitoids communities were between 35% and 38% in the first year, and 30% and 40% in the second one. Although these values show no observable effect in the native parasitoid communities caused by the introduction of T. sinensis, is important to note that its population is still in the early stages of development. According to Quacchia et al., (2014a), T. sinensis females are extremely selective with oviposition, and almost only try to lay their eggs in D. kuriphilus galls. However, one case of mating was registered between T. sinensis and T. beneficus (Yara, 2014; Yara et al., 2007, 2010). This shows that even with T. sinensis high specificity in host and mating choices, there is a risk of changing the ecosystems balance with its introduction.
As T. sinensis proves to be capable of establishing a new population, increase its abundance and expand to nearby orchards in a few years, it is a good candidate for the control of the chestnut gall wasp. However, for the same reasons and the possibility of either hybridization with local species or their displacement, caution must be taken when applying this agent. More information about T. sinensis’ biology and ecology is needed in order to better understand its role in the Portuguese ecosystem and provide the most value in the management of D. kuriphilus.