Serviços Personalizados
Journal
Artigo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Acessos
Acessos
Links relacionados
-
 Similares em
SciELO
Similares em
SciELO
Compartilhar
Portugaliae Electrochimica Acta
versão impressa ISSN 0872-1904
Port. Electrochim. Acta vol.33 no.3 Coimbra maio 2015
https://doi.org/10.4152/pea.201503153
Kinetic Investigation of Quaternary Ni-Fe-P-C Composite Coating as a New Catalyst for Hydrogen Evolution Reaction
Ali R. Madramaa,* , Abolfazl F. Zonouzb and Hamid R. Pouretedalb
a Faculty of Chemistry and Chemical Engineering, Malek-Ashtar University of Technology, Tehran 15875-1774, Iran
b Department of Applied Chemistry, Malek-Ashtar University of Technology, Isfahan 83145-115, Iran
Abstract
A new quaternary Ni-Fe-P-C composite coating was deposited on copper substrates by an electrodeposition process. The morphology, structure and composition of the prepared electrode were characterized using scanning electron microscopy (SEM), X- ray diffraction (XRD) pattern, and energy dispersive X-ray spectroscopy (EDX). The activity of the composite coating toward hydrogen evolution reaction (HER) was evaluated on the basis of the steady-state polarization Tafel curves and electrochemical impedance spectroscopy (EIS) in 1 M NaOH at temperature of 298 K. The experimental results revealed that the HER activity of the Ni-Fe-P-C coatings was enhanced compared with both ternary coatings of Ni-P-C and Ni-Fe-C. The source of its activity was found from its high surface area and its intrinsic properties. It was sufficient to assume the Volmer-Heyrovsky mechanism with the Heyrovsky reaction as the rate- determining step to explain the experimental data. Besides the high electrocatalytic activity, excellent chemical and electrochemical stability were observed for Ni-Fe-P-C composite coating toward the HER in 1 M NaOH at 298 K.
Keywords: hydrogen evolution reaction, quaternary Ni-Fe-P-C coatings, composite coatings, electrochemical activity, electrocatalyst.
Introduction
Hydrogen is the cleanest and ideal fuel and it is considered as an ideal energy carrier that can be an alternative to fossil fuels [1]. Hydrogen evolution reaction (HER) is one of the most intensively studied reactions in electrochemistry because it occurs through a limited number of reaction steps with only one reaction intermediate involved [2]. One major aspect of the research undertaken is to improve catalytic electrode materials in terms of their activity, efficiency and mechanical and corrosion stability [3]. Noble metals such as platinum and ruthenium are the most active materials, but these metals are very expensive [4]. Although non-platinum active metals such as Fe, Ni or Co are considerably cheaper, they suffer from corrosion and passivation under the HER conditions and low electrocatalytic activity; therefore, great efforts are being made in order to improve electrocatalytic stability and activity of these materials, usually performed by alloying them and modifying their synthesis routes [5,6]. In recent years, many efforts have been made to develop new materials with high electrocatalytic activity and low-cost [7-10]. In the previous works [11,12] we reported the electrocatalytic activities of Ni-Fe-C and Ni-P-C composite electrodes toward the HER, where the activities were improved mainly by increasing the intrinsic activity induced by carbon embedded to Ni-Fe and Ni-P matrix, respectively. The current study was planned to combine interesting properties of Ni-P-C composite (i.e., excellent physical stability and moderate electrocatalytic activity) with those of iron in Ni-Fe-C composite (i.e., excellent activity) to fabricate a new type of catalyst (Ni-Fe-P-C) and examine its activity for the HER in alkaline solution by steady-state polarization Tafel curves, and electrochemical impedance spectroscopy (EIS).
Experimental
Electrode preparation
All chemicals were of analytical reagent grade and were used as received and also the all solutions were prepared by double distilled water. Electrodeposition of the quaternary Ni-Fe-P-C composite coatings was carried out under constant current in a double wall glass cell containing a volume of 300 mL of depositing electrolyte, a copper rod sealed in a heat shrinkable tube exposing a 0.229 cm2 surface area as substrate, and a platinum plate (purity of 99.99% and surface area >25 cm2) as counter electrode. Before electrodeposition, the copper substrate was polished with sandpaper (P 600, Siawats), washed with water, immersed in a 1:1 HNO3 solution, washed with water, and immediately introduced into the cell for electrodeposition. The simple electrodeposition method of Ni-Fe-P-C was performed in a three-step method. Composition of the bath and electrodeposition conditions of each step are given in Table 1.
(i) First, a layer of Ni was deposited from bath A, (ii) then Ni-P was coated on Ni from bath B and finally (iii) Ni-Fe-P-C deposited from bath D. The electrodeposition steps were performed in 30 min for each bath and at temperature of 298 K.
Electrode measurements
A two-compartment Pyrex® glass cell, consisting of a large surface area Pt plate electrode as the counter electrode and an external Hg/HgO/1 M NaOH electrode as the reference electrode was used for electrochemical measurements. The details of the electrochemical cell have been described previously [11]. All measurements were performed in purified 1 M NaOH degassed with argon (99.99%). The steady-state polarization Tafel curves and EIS measurements were carried out using a Parstat 2273 Potentiostat/Galvanostat. The details of the measurements have been described previously [11]. The Tafel plots were recorded galvanostatically (45 s after a constant current application) at cathodic current densities range from j=-100 mA cm-2 to j= -1 μA cm-2. At the steady state, the electrode potentials were recorded for iR drop, determined by the EIS method at various electrode potentials. The EIS measurements were performed at the steady-state [11,12]. Surface morphology and chemical composition of the alloys were investigated by scanning electron microscopy (SEM) and energy dispersive X-ray diffraction microanalysis (EDX) (Philips-XL-30). Crystal structure of the alloys was determined by X-ray diffraction (XRD) analysis (Philips-X'Pert-MPD) using Cu Kα wavelength of 1.5418 Å.
Results and discussion
Composition and surface morphology of the coatings
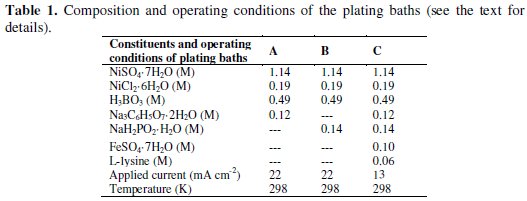
The SEM micrograph and a typical EDX spectrum obtained on the surface of the as-deposited Ni-Fe-P-C coating surface are shown in Figs. 1 and 2, respectively.

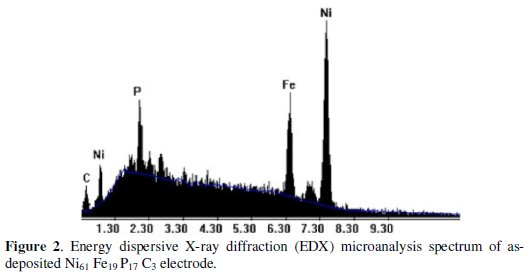
The SEM micrographs of the investigated quaternary coating (Fig. 1) displayed globular island-structure. This structure is probably accountable for high electrochemical activity and large real surface area of the quaternary Ni-Fe-P-C coating. Furthermore, the quaternary coating of Ni-Fe-P-C showed good adherence to the lower layers (Ni-P layer deposited from bath B in Table 1) and good physical stability and was hard and difficult to remove with a sandpaper. The experiment was not successful if the second layer in the coating (bath B in Table 1) was changed with Ni-Fe. The EDX spectrum shows that the chemical composition of the top layer of the coatings prepared under optimized conditions (Table 1) was Ni61Fe19P17C3. The diffraction pattern (Fig. 3) of the Ni-Fe-P-C coating shows an amorphous structure with a main broad peak centered at 44.5 Å.
Kinetic studies
Steady-state polarization curves
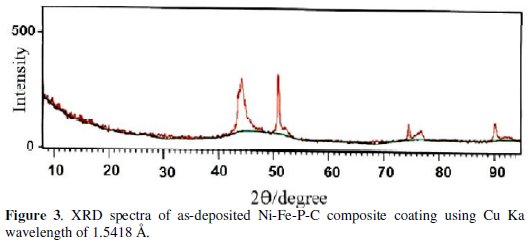
The apparent activity of the electrode was studied by steady-state polarization Tafel curves for the HER in 1 M NaOH on Ni-Fe-P-C composite coating at 298 K in argon atmosphere. The iR-corrected Tafel curves for Ni-Fe and Ni-Fe-C, Ni-P and Ni-P-C, and Ni-Fe-P-C are displayed in Fig. 4 and the kinetic parameters of the HER determined from linear part of the Tafel curves, i.e. exchange current density (j0), Tafel slopes (b), and overpotential at j=250 mA cm-2 (η250) are presented in Table 2.
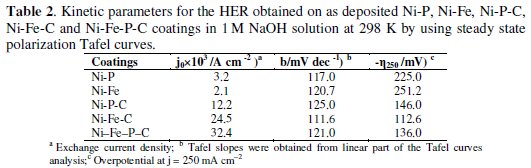
These parameters were calculated taking into account the geometric surface area of the investigated coatings. The Tafel curves for the quaternary coating showed only one Tafel slope in the whole range of the studied potentials (Fig. 4), that is similar to Ni-P based coatings. In terms of j0, Table 2 shows that Ni-Fe-P-C coating was more active catalyst toward the HER than other ternary coatings at the HER equilibrium potential. The value of j0 for the Ni-Fe-P-C (32.4×103 /A cm-2) was higher than the Ni-P-C (12.2×103 /A cm-2) and Ni-Fe-C (24.5×103 /A cm-2) coatings. Furthermore, at far from equilibrium, i.e., high current densities, a comparison of η250 values for the quaternary Ni-Fe-P-C coating (-η250=136.0 mV) with the ternary coatings of Ni-P-C (-η250=146.0 mV) and Ni-Fe-C (-η250=112.6 mV) indicates that the quaternary coating had higher activity than Ni-P-C, but lower activity than Ni-Fe-C coating toward the HER. Although, it was predictable from previous work [11] that the higher content of iron in the composite coatings could enhance its intrinsic activity toward the HER, we observed a decrease in stability of the quaternary coatings if the iron value was larger than the values showed in Table 1 (bath B). However, the steady-state polarization studies, alone, are not enough to explain the kinetics of HER. Thus, more precise information was obtained by the EIS, and analyzed in conjunction with Tafel curve data to find the source of increased activities.
Electrochemical impedance spectroscopy (EIS)
In the present work, the EIS measurements were used to investigate the interfacial behavior and activities of the Ni-Fe-P-C composite electrode toward the HER in a wide range of frequency and potential. The values of electrode potentials at which the EIS measurements are taken were chosen from the linear part of the potential region obtained by d.c. polarization measurements. A typical impedance diagram for the quaternary Ni-Fe-P-C coating at various overpotentials in 1 M NaOH is presented in Fig. 5.
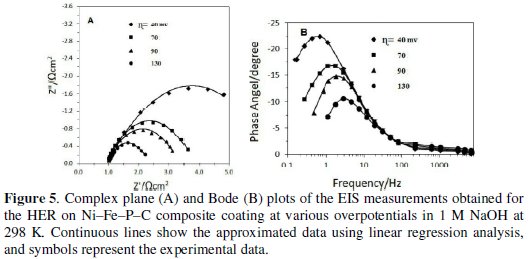
It can see that the Niquist diagram (Fig. 5 A) looks like only one slightly distorted capacitive semicircle in the entire range of the studied overpotentials. After testing the models related to the HER [13,14], considering the fit criteria such as ''number of model parameters'' and ''Chi square values'' [15], the CPE model was found to be enough for approximations of the EIS data of the investigating electrode. Approximation of the experimental EIS data with one-CPE model permitted estimation of the solution resistance (Rs), capacitive parameter (T) related to the average double layer capacitance (Cdl), charge transfer resistance (Rct) and dispersion parameter related to the CPE model (φ). Additionally, determination of the exact number of active sites per unit of surface area is a difficult task [16]; however, a comparison between values of 20 μFcm-2 assumed for the double layer capacitance of a smooth electrode [17] and the Cdl of objective electrode is a suitable method to estimate surface roughness (Rf):
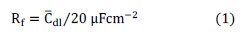
The value of 4908 was obtained for Rf of the Ni-Fe-P-C by approximation of the EIS data (Table 3, column 4).
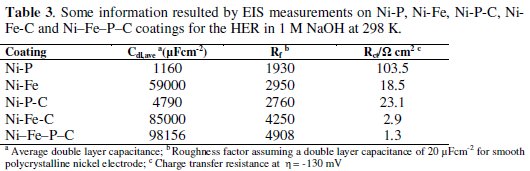
This high real surface area confirmed the SEM micrograph of the investigated coating (Fig. 1) and is one of the high activity sources of Ni-Fe-P-C toward the HER. The increasing factor of the Rf from the quaternary Ni-Fe-P-C coating to Ni-P-C and Ni-Fe-C was 78% and 16%, respectively. Also, the decreasing factor of the Rct from Ni-Fe-P-C to Ni-P-C and Ni-Fe-C was 94% and 55%, respectively. Therefore, the EIS studies revealed that Ni-Fe-P-C coating was more active catalyst for the HER than ternary coatings of Ni-P-C and Ni-Fe-C that were reported previously [11,12].
Rate constants
The determination of the HER rate constants for the studied materials is very import, because it helps us to find some valuable kinetic information such as: HER mechanism and rate determining step, intrinsic activity (synergetic effect) of new catalysts for the HER and precise determination of the HER activity sources. Different methods as galvanostatic step, open circuit potential decay, and Tafel-impedance have been already reported to be effective for evaluation of the rate constants of the HER in alkaline solutions [18]. The kinetic parameters for the quaternary coating were extracted by simultaneous fitting of the Tafel and EIS data (i.e., Tafel-impedance method) to corresponding kinetic equations using the NLS method [19]. The details of the method for determining the HER rate constant have been described previously [11]. The mechanism of HER in alkaline solution involves the formation of an adsorbed hydrogen atom intermediate, MHads(Volmer reaction):
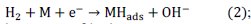
the electrochemical desorption of hydrogen into solution (Heyrovsky reaction):
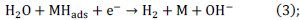
and/or a chemical desorption by the combination of two atoms (Tafel reaction):
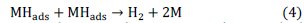
where M represents the electrode materials and MHads the hydrogen adsorbed on coating surface.
A linear variation was observed for log (1/Rct) as a function of η for the Ni-Fe-P-C electrode (Fig. 6).
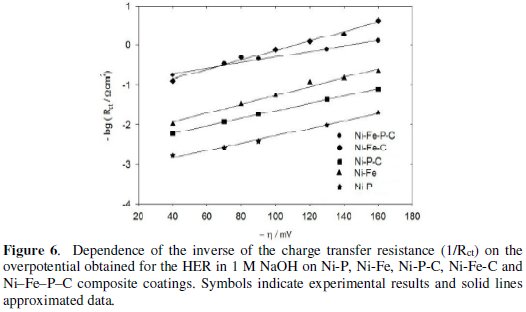
The obtained value 128 mVdec-1, for slope of Ni-Fe-P-C coating at 298 K was in good agreement with those obtained from Tafel curve (Fig. 4 and Table 2) and predicts the Volmer-Heyrovsk'y reaction mechanism [19] for the HER on the investigated quaternary coating.
The determined rate constant (Heyrovsky reaction) value k2 was smaller than k1 rate constant (Volmer reaction) for the Ni-Fe-P-C coating, and hence the Heyrovsky reaction was detected as the rate-determining step. Similar results were previously observed for the ternary coatings of Ni-P-C and Ni-Fe-C, [11,12]. The values of k1, k2, average apparent rate constants (kav = (k1-1 + k2-1)-1) intrinsic activity (kintr=kav/Rf), real exchange current densities (j0/Rf) and symmetry factors (β1 and β2) [4] for quaternary and ternary coatings are presented in Table 4.

The value of kav for the quaternary Ni-Fe-P-C coating ((1.4±0.3)×10-7 mol cm-2s-1) is almost two orders of magnitude larger than those for both ternary Ni-P-C ((5.2±0.3)×10-9 mol cm-2 s-1 and Ni-Fe-C ((4.5±0.5)×10-9 mol cm-2 s-1) coatings. Furthermore, higher intrinsic activity was also observed on the quaternary Ni-Fe-P-C ((2.9±0.3) × 10-11 mol cm-2s-1) as compared with ternary Ni-Fe-C ((1.1±0.5)×10-12 mol cm-2s-1) and somewhat Ni-P-C ((2.2±0.3)×10-11 mol cm-2s-1) coatings. Additionally, the EIS studies showed higher value of Rf for quaternary Ni-Fe-P-C coating than both ternary Ni-P-C and Ni-Fe-C coatings by factor of 78% and 16%, respectively. These results revealed that the quaternary coating of Ni-Fe-P-C is a more active electrocatalyst than both Ni-P-C and Ni-Fe-C in alkaline solution and could be used as a high active and inexpensive electrocatalyst for the HER.
Stability of Ni-Fe-P-C electrode
The electrochemical stability of the electrode material is one of the important characteristics that must be established. Thus, the Ni-Fe-P-C coating was tested through long term working experiments by operating at a constant applied overpotential, η=-130mV, at 298 K in 1 M NaOH, and its current response (chronoamperogram) was recorded (Fig. 7).
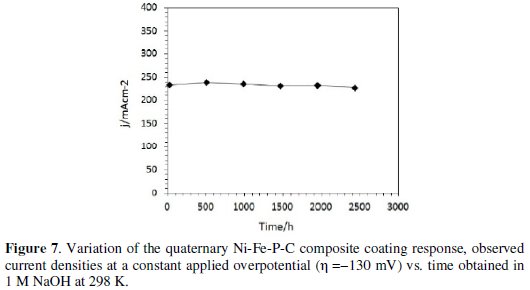
Between consecutive measurements, with an interval of about 20 days, the coating was kept in 1 M NaOH at 298 K. The corresponding current monitored during this time on the electrode, demonstrated no significant changes. This behavior showed that the quaternary coating of Ni-Fe-P-C was electrochemically stable for a relatively long time (104 days) for the HER in alkaline solutions.
Conclusions
The quaternary coating of Ni-Fe-P-C was prepared at low current densities by electrodeposition method from an aqueous solution and studied toward the HER, for the first time. Quantitative kinetic information obtained by the Tafel and EIS measurements revealed that Ni-Fe-P-C coatings have considerably higher electrocatalytic activity for the HER than the previously studied ternary coatings of Ni-P-C and Ni-Fe-C in 1 M NaOH solution. The average apparent rate constants of the assumed mechanism, Volmer-Heyrovsky, were estimated by simultaneous approximation of the Tafel and EIS data. The k2 values were smaller than those obtained for hydrogen adsorption rate constants (k1) for Ni-Fe- P-C electrode. This behavior revealed that Heyrovsky reaction is the rate- determining step for the HER kinetics on this electrode. Its activity is originated from high surface area and from the intrinsic properties of the coating. Also, the Ni-Fe-P-C electrode was physically, chemically, and electrochemically stable for a relatively long time and showed high electrocatalytic activity for the HER.
References
1. Veziroglu T N, Barbir F. Int J Hydrogen Energ. 1992;17:391. [ Links ]
2. Kim H K, Yang D C, Jang I S, et al. Int J Hydrogen Energ. 2009;34:9570. [ Links ]
3. Zeng K, Zhang D. Prog Energ Combust. 2010;36:307. [ Links ]
4. Losiewicz B, Budniok A, Rowinski E, et al. Int J Hydrogen Energ. 2004;29:145. [ Links ]
5. Safizadeh F, Gali E, Houlachi G. Int J Hydrogen Energ. 2015;40:256. [ Links ]
6. Shan Z, Liu Y, Chen Z, et al. Int J Hydrogen Energ. 2008;33:28. [ Links ]
7. Lasia A. In: Vielstich W, Lamm A, Gasteiger HA. editors. Handbook of fuel cells; fundamentals, technology and applications. Part 4. Hydrogen evolution reaction. Vol. 2. Wiley; 2003. p 416. [ Links ]
8. Pierozynski B, Mikolajczyk T, Turemko M, et al. Int J Hydrogen Energ. 2015;40:1795-1799. [ Links ]
9. Du H, Gu S, Liu R, et al. Int J Hydrogen Energ. 2015. In Press. [ Links ]
10. Kichigin V I, Shein A B. Electrochim Acta. 2015;164:260. [ Links ]
11. Shervedani R K, Madram A R. Electrochim Acta. 2007;53:426. [ Links ]
12. Shervedani R K, Madram A R. Int J Hydrogen Energ. 2008;33:2468. [ Links ]
13. Hitz C, Lasia A. J Electroanal Chem. 2002;532:133. [ Links ]
14. Kubisztal J, Budniok A, Lasia A. Int J Hydrogen Energ. 2007;32:1211. [ Links ]
15. Inzelt G, Lang G, J. Electroanal. Chem. 378 (1994) 39.
16. Brug G J, van der Eden A L G, Rehbach M S, et al. J Electroanal Chem. 1984;176:275. [ Links ]
17. Milica P M K, Vladimir M N, Gvozden S T, et al. Int J Hydrogen Energ. 2009;34:703. [ Links ]
18. Trasatti S. In: Gerischer H, Tobias CW, editors. Advances in electrochemical science and engineering. Vol 2. Weinheim. VCH; 1992. p 2. [ Links ]
19. Harrington D A, Conway B E. J Electroanal Chem. 1987;221:1. [ Links ]
Acknowledgements
The authors thank to Department of Applied Chemistry (Malek-Ashtar University of Technology, Isfahan, Iran) for financially support and providing the lab facilities to bring about this work.
*Corresponding author. E-mail address: Ar.madram@gmail.com
Received 9 April 2015; accepted 15 June 2015














