Introduction
Vitamin D deficiency is a worldwide epidemic in all age groups, including in countries with apparently sufficient sunlight exposure1. The Institute Of Medicine (IOM) defined vitamin D deficiency levels below 20 ng/ml2. According to this definition, it is estimated in Europe a prevalence of 40.4%3, regardless of age and ethnic groups, and latitude of study populations.
Major changes in maternal calcium homeostasis and bone metabolism occur in pregnancy to satisfy the fetus´ needs4. Vitamin D is an important regulator of calcium homeostasis. During pregnancy, vitamin D level starts to increase through the first trimester and continue until delivery, probably due to enhanced renal synthesis and increased extra-renal synthesis in the placenta4. For this increment is essential to maintain proper 25-dihydroxyvitamin D levels available, a precursor of vitamin D active form5.
Maternal vitamin D deficiency in pregnancy has been associated with adverse pregnancy outcomes. Recent systematic reviews and meta-analysis suggested that low vitamin D levels during pregnancy can increase the risk of preeclampsia (PE)6,7, gestational diabetes mellitus (GDM)8-10, cesarean section11,12, preterm birth (PTB)13,14 and low birthweight (LBW)15. However, some limitations of these studies deserve attention. First, the majority of them had a retrospective design. Second, only a single measurement of vitamin D level was performed in these investigations and at different moments of pregnancy. These studies included heterogeneous population with different sun exposure, ethnic groups, a wide variation concerning dose, type, and duration of Vitamin D supplementation. Also, in a greater number of investigations, Vitamin D supplementation was only initiated after 20 weeks, and at this time of pregnancy, few improvements will be expected. Finally, some important confounders (as body mass index (BMI) and maternal age) were not considered by the majority of authors.
Due to a lack of strong evidence, there is no consensus regarding the role of vitamin D status on pregnancy outcomes. Recommendations adopted among internationally recognized societies vary. Currently, the World Health Organization (WHO) does not recommend vitamin D supplementation during pregnancy as part of routine antenatal care16. The organization only recommends this supplementation for pregnant women with documented vitamin D deficiency, which matches the American Congress of Obstetricians and Gynecologists guidelines17. The Royal College of Obstetricians and Gynaecologists suggest 400 IU/day for all pregnant women18.
The effect of low vitamin D levels during pregnancy is still not understood and well-designed prospective studies are still needed. The purpose of this research was to determine the association between first trimester vitamin D status and pregnancy outcomes.
Methods and materials
A prospective observational study was performed including women with a singleton gestation who underwent first-trimester screening for aneuploidy at a Portuguese perinatal center, between April and June 2018. This study was conducted with the approval of the Ethics Committee. All participants signed written informed consent. Women who were less than 18 years old, were non-caucasian, who had renal disease or who were supplemented with vitamin D during pregnancy were excluded. Likewise, records with incomplete outcome data were not included. Any pregnancies with lethal congenital anomalies were excluded. Information on maternal sociodemographics, medical and obstetrical history, gestational age at inclusion, pregnancy course and delivery outcome were collected. Serum plasma 25-OH vitamin D concentrations were analysed between 11 weeks 0 days and 13 weeks 6 days of gestation using Chemiluminescent Microparticle Immunoassay. The lower limit of detection and lower limit of quantification achieved was 3.1 ng/mL and 8.0 ng/mL, respectively. Vitamin D deficiency was defined at levels lower than 20ng/ml. In a primary analysis, two groups (vitamin D <20ng/ml and ≥20ng/ml) were evaluated, as well as their association with adverse pregnancy outcomes.
The evaluated outcomes included gestational hypertension, PE, GDM, PTB, and LBW. Gestational hypertension was defined as new onset of hypertension (systolic blood pressure ≥140 mmHg and/or diastolic blood pressure ≥90 mmHg) at ≥20 weeks of gestation, in the absence of proteinuria or new signs of end-organ dysfunction. The blood pressure readings were documented in two occasions, at least, through a four hours interspersed period. PE was defined as new onset of hypertension and either proteinuria or end-organ dysfunction after 20 weeks of gestation. Criteria for diagnosis were: Systolic blood pressure ≥140 mmHg or diastolic blood pressure 90 mmHg, and proteinuria ≥0.3 grams in a 24-hour urine specimen or protein:creatinine ratio ≥0.3, or signs of end-organ dysfunction (platelet count <100.000/microliter, serum creatinine >1.1 mg/dL or doubling of the serum creatinine, elevated serum transaminases to twice normal concentration). GDM was diagnosed if fasting glucose level ≥92 mg/dL and <126mg/dL at first trimester or at least one value of plasma glucose concentration ≥92, 180 and 153 mg/dl (for fasting, one-hour and 2-hour post glucose load glucose values respectively), after performing a 75g oral glucose tolerance test at 24-28 weeks of gestation. PTB was defined as a delivery occurring before 37 weeks of pregnancy. LBW was defined as a birth weight of less than 2500 g liveborn, regardless of gestational age.
Some variables were considered to be confounders for PE, namely, maternal age, parity, pre-pregnancy BMI, and chronic hypertension. Maternal age, pre-pregnancy BMI, weight gain, smoking habits, hypertension, family history of diabetes and history of GDM were considered for GDM. Maternal age, parity, pre-pregnancy BMI, gestational week, extremes of neonatal birthweight and GDM were considered for cesarean section. Gestational hypertension, chronic hypertension, pregestational diabetes, and preeclampsia were considered for LBW.
Statistical analyses were performed using the Statistical Package for the Social Sciences (SPSS Inc. Chicago, IL, version 23). Continuous variables were compared between groups using the T-test and dichotomous variables using the Fisher’s Exact test or the Chi-square test. In the final phase, logistic regression analysis was performed to evaluate possible confounding or adjustment factors. OR and 95%CI were calculated. The goodness of fit was evaluated by the Hosmer-Lemeshow test. Statistical significance was defined as p-value <0.05.
Results
One hundred and eighty five pregnant women were recruited. There were 141 (76,2%) women who exhibited low levels of vitamin D. The means age of patients was 32±5 years with a BMI of 28.2±5.2 Kg/m2 and it was comparable between the two groups (Table I). There was no statistical significance regarding the presence of medical comorbidities such as hypertension. GDM was found to be higher in pregnant women with vitamin D deficiency (p=0.004). No statistically significant differences were found concerning to pregnancy outcomes between the groups (Table II). There were no statistically significant differences in the mode of delivery and with respect to neonatal outcomes such as gender, birth weight, and Apgar score.
Table I pregnant women characteristics
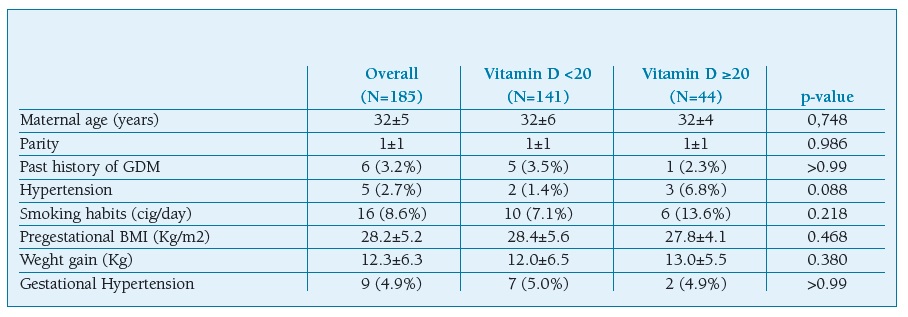
Data were given as frequencies (percent) or median (±standard deviation). BMI, body mass index; Cig, cigarettes; GDM, gestational diabetes mellitus.
Table II maternal and neonatal outcomes
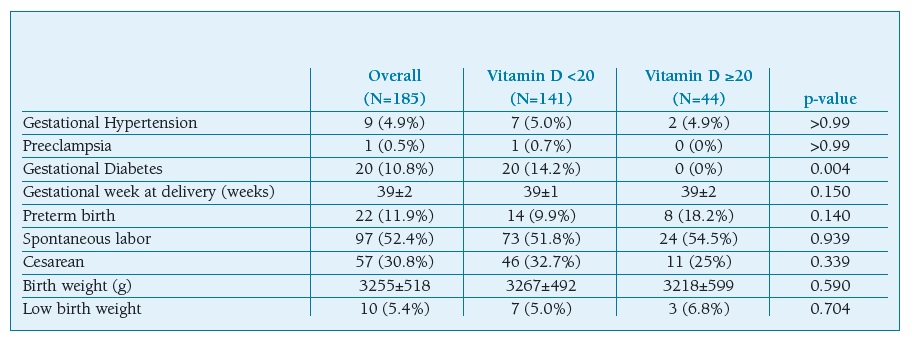
Data were given as frequencies (percent) or median (±standard deviation)
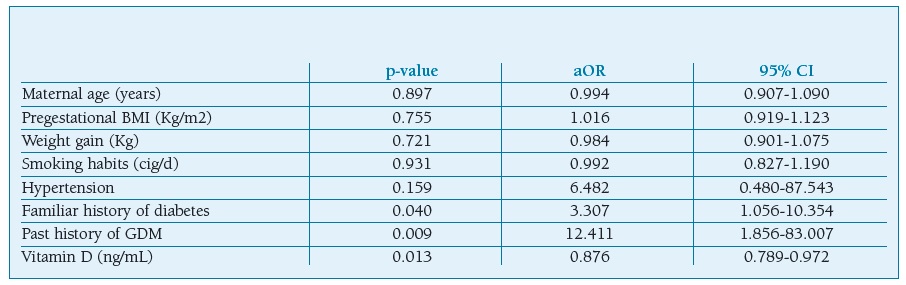
Multiple logistic regression analysis showed that decreased Vitamin D level in the first trimester of gestation is associated with a higher risk of GDM (OR 0.876, CI 0.789-0.972; p=0.013), after adjusting for maternal age, pre-pregnancy BMI, weight gain, smoking habits, hypertension, familiar history of diabetes and history of GDM (Table III).
Discussion
In the current study, a low vitamin D level in the first trimester of pregnancy was a predictor of GDM. This result turned out to be a coherent element taking into account some previous reports8,10,19 but different from others20. This non-consensual approach is due to the study designs´ heterogeneity and inadequate consideration of confounding factors. For example, Poel et al. in their systematic review and meta-analysis only included three studies in which maternal age and BMI were considered as confounders10.
Some theories have been suggested for the association between vitamin D and GDM, including repercussions on the pancreas, insulin-responsive tissues, and systemic inflammation21,22. Vitamin D can control the blood glucose levels by binding and activating the vitamin D receptor in the pancreatic beta-cells, regulating the insulin release23,24. The active form of vitamin D directly stimulates the secretion of insulin, once the presence of the vitamin D response element in the insulin gene promoter of pancreatic beta-cells25 and indirectly, regulating the balance between the extracellular and intracellular beta-cell calcium pools25,26. Vitamin D seems to improve insulin sensitivity in peripheral insulin-responsive tissues25. Vitamin D stimulates insulin receptor expression, improving insulin responsiveness for glucose transport, and also has an effect on glucose metabolism through the regulation of calcium flux within cell membranes 25-29. Moreover, low vitamin D status leads to an efficiency reduction in the intestinal calcium absorption, increasing the secretion of parathyroid hormone (PTH). PTH may mediate insulin resistance by reducing glucose uptake by peripheral tissues30. Other mechanisms have been proposed to explain the impact of vitamin D on insulin resistance including the immunoregulatory and anti-inflammatory functions of vitamin D31. Vitamin D might reduce the formation of reactive oxygen species in adipocytes32 through the cellular antioxidants expression controlling, preventing oxidative damage33. Vitamin D also controls vital genes with a potential negative modulator of pro-inflammatory cytokines release34, reducing IL-6, TNF-α, and C-reactive protein35, and prevents transcription of pro-inflammatory factors genes.
Recent systematic reviews and meta-analyses had demonstrated that insufficient serum levels of vitamin D were associated with PE, LBW, PTB and the risk of cesarean section7,8,12,19. In our study there was no association between vitamin D levels and these adverse outcomes. However, due to small sample size, these events should be discussed in further investigations.
A highly prevalence of low vitamin D levels among Portuguese pregnant women in the first trimester was registered in this research. This result is unexpected for a healthy population living in a country with high sun-exposure. There might be some plausible reasons for this happen. The first hypothesis highlights the possibility of a limited sunlight exposure within the recruited women. Vitamin D levels increase in summer and decrease in winter due to the dependency of vitamin D on ultraviolet B radiation. The measurement of vitamin D was performed between April and June, after cold seasons, when people wear more often clothes that cover the entire body. Therefore, the amount of ultraviolet-B radiation reaching the skin was impaired. Probably the cut-off value for vitamin D deficiency should be revised in forthcoming investigations according to the season. Another possible explanation is that a high number of recruited women were overweight and had obesity. The high prevalence of vitamin D deficiency in obese subjects is a well-documented finding, most likely due to the dilution effect of vitamin D in the large adipose mass36.
This study has some strengths that merit consideration. As all included women had a vitamin D measurement in a well-defined interval, early in pregnancy, it is unlikely that vitamin D status in the last trimester of pregnancy influences GDM development. Only Caucasian women were included and since the recruitment occurred in Spring, sun exposure discrepancies were avoided. Variables as maternal age, pregestational BMI and weight gain were considered in the results.
However, several limitations also deserve discussion. A single measurement of Vitamin D in pregnancy is probably not sufficient, since levels can be modified by food consumption, supplement use and sun exposure. Although in this study we excluded women with vitamin D supplementation, we have not questioned on physical activity and dietary intake. Finally, some important markers were not considered as vitamin D receptor activity, calcium, phosphate and PTH levels, insulin-like growth factor I activity and other bone turnover molecules. The authors only proposed to evaluate the significance of vitamin D status on pregnancy outcomes, the impact of supplementation with vitamin D would be addressed in future studies.
Conclusion
A high prevalence of low vitamin D levels among pregnant women in the first trimester was seen in this study. Low vitamin D level was a predictor of GDM. However, there was no significant association between vitamin D status and other maternal and neonatal outcomes. Further investigation is needed to determine whether if vitamin D supplementation would improve pregnancy outcomes when low levels are detected.
Acknowledgements
The authors would like to acknowledge Domingos Ribeiro, Sandra Ferreira, Joaquim Saraiva, Ana Sofia Cardoso, Bárbara Monteiro, Lúcia Lima, Sofia Gavinho and Andreia Gradíssimo for their assistance and support in this research.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
Author contributions
MSG and MLC researched literature and conceived the study. MSG, MLC, VT, JAP and SS were involved in protocol development, gaining ethical approval, patient recruitment and data analysis. MSG and MLC wrote the first draft of the manuscript. All authors reviewed and edited the manuscript and approved the final version of the manuscript.