Services on Demand
Journal
Article
Indicators
-
 Cited by SciELO
Cited by SciELO -
 Access statistics
Access statistics
Related links
-
 Similars in
SciELO
Similars in
SciELO
Share
GE-Portuguese Journal of Gastroenterology
Print version ISSN 2341-4545
GE Port J Gastroenterol vol.27 no.4 Lisboa Aug. 2020
https://doi.org/10.1159/000505033
IMAGES IN GASTROENTEROLOGY AND HEPATOLOGY
Colonic Mantle Cell Lymphoma with Multiple Lymphomatous Polyposis
Linfoma de células do manto do colon com polipose linfomatosa múltipla
Rama Nassria, Ammar Nassrib, Ahmad Alkhasawnehc, Bruno de Souza Ribeirob, Ron Scheyb
aDepartment of Medicine, Alfaisal University, Riyadh, Kingdom of Saudi Arabia; bDivision of Gastroenterology, Department of Medicine, University of Florida at Jacksonville, Jacksonville, FL, USA; cDepartment of Pathology, University of Florida at Jacksonville, Jacksonville, FL, USA
* Corresponding author.
Keywords: Cancer, Colon, Mantle cell lymphoma
Palavras-Chave: Cancro, Colon, Linfoma do manto
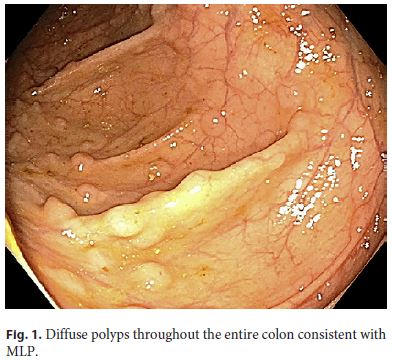
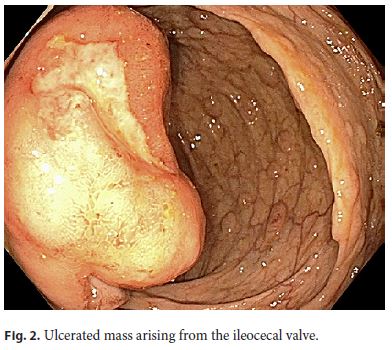
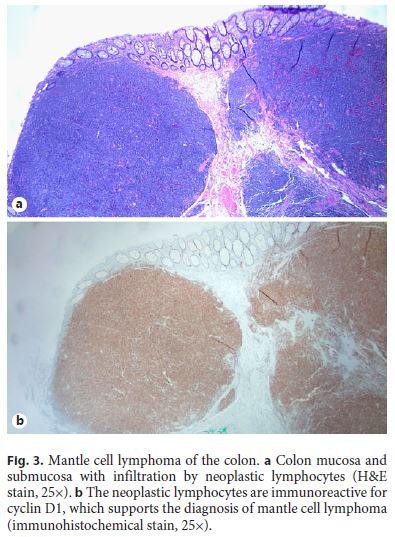
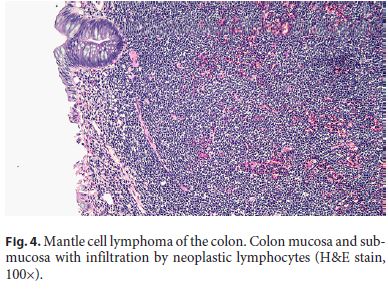
A 64-year-old white male with a past medical history of coronary artery disease, hypertension, and diabetes presented for a screening colonoscopy. He had no complaints of hematochezia, nor of any fevers, fatigue, night sweats, or chills. A colonoscopy was performed and revealed diffuse, innumerable sessile sub-centimeter polyps throughout the colon (Fig. 1), as well as a fungating, ulcerated mass arising from the ileocecal valve (Fig. 2). The mass was biopsied and representative samples of the polyps were resected. Pathology revealed mantle cell lymphoma, with the neoplastic lymphocytes staining for CD20 and cyclin D1, but negative for CD3 (Fig. 3, 4). Computed tomography revealed extensive mesenteric and retroperitoneal lymphadenopathy, as well as prominent carinal and hilar nodes. Flow cytometry showed monoclonal CD5- positive B cells, and a bone marrow biopsy revealed 40– 50% involvement of the bone marrow by mantle cell lymphoma. A PET scan was performed, and revealed extensive FDG-avid retroperitoneal, mesenteric, iliac, inguinal, and bilateral axillary lymphadenopathy as well as an FDGavid cecal mass. The patient was diagnosed with stage IV mantle cell lymphoma, had a Port-a-Cath placed, and was initiated on rituximab and bendamustine given his comorbidities and poor functional status.
Mantle cell lymphomas involving the gastrointestinal tract are rare, accounting for 4–9% of all gastrointestinal B-cell lymphomas [1]. A modified Ann Arbor classification is used for anatomic description of disease extent [2], although functional status and risk stratification using scores, such as the Mantle Cell International Prognostic Index (MIPI), also play a large role in treatment. Multiple lymphomatous polyposis (MLP) is an uncommon type of primary gastrointestinal lymphoma, presenting as multiple lymphomatous polyps throughout the gastrointestinal tract. Although MLP is most commonly seen with mantle cell lymphoma, it has also been reported in marginal B-cell lymphomas, follicular lymphomas, and mucosa-associated lymphoid tissue (MALT) lymphomas, which makes adequate endoscopic sampling and immunohistochemistry mandatory. There may be a lymphoid mass in the terminal ileum, which may be the initial focus of disease in 35–48% of cases, although a rectal mass is sometimes described (6%) [1]. Presenting complaints are most often abdominal pain (65%), followed by diarrhea (32%) and hematochezia (22%). Primary gastrointestinal tract mantle cell lymphoma has a poor prognosis, with a median survival of 3–4 years [3]. Although traditional treatment regimens are comprised of R-CHOP [3], recent studies have suggested a role for bendamustine and rituximab [4], particularly if the patient’s functional status precludes a more aggressive chemotherapy regimen.
References
1 Ruskoné-Fourmestraux A, Audouin J. Primary gastrointestinal tract mantle cell lymphoma as multiple lymphomatous polyposis. Best Pract Res Clin Gastroenterol. 2010 Feb;24(1):35–42.
2 Cheson BD, Fisher RI, Barrington SF, Cavalli F, Schwartz LH, Zucca E, et al.; Alliance, Australasian Leukaemia and Lymphoma Group; Eastern Cooperative Oncology Group; European Mantle Cell Lymphoma Consortium; Italian Lymphoma Foundation; European Organisation for Research; Treatment of Cancer/Dutch Hemato-Oncology Group; Grupo Español de Médula Ósea; German High-Grade Lymphoma Study Group; German Hodgkin’s Study Group; Japanese Lymphorra Study Group; Lymphoma Study Association; NCIC Clinical Trials Group; Nordic Lymphoma Study Group; Southwest Oncology Group; United Kingdom National Cancer Research Institute. Recommendations for initial evaluation, staging, and response assessment of Hodgkin and non-Hodgkin lymphoma: the Lugano classification. J Clin Oncol. 2014 Sep;32(27):3059–68.
3 Lenz G, Dreyling M, Hoster E, Wörmann B, Dührsen U, Metzner B, et al. Immunochemotherapy with rituximab and cyclophosphamide, doxorubicin, vincristine, and prednisone significantly improves response and time to treatment failure, but not long-term outcome in patients with previously untreated mantle cell lymphoma: results of a prospective randomized trial of the German Low Grade Lymphoma Study Group (GLSG). J Clin Oncol. 2005 Mar;23(9):1984–92.
4 Flinn IW, van der Jagt R, Kahl B, Wood P, Hawkins T, MacDonald D, et al. First-Line Treatment of Patients With Indolent Non-Hodgkin Lymphoma or Mantle-Cell Lymphoma With Bendamustine Plus Rituximab Versus R-CHOP or R-CVP: Results of the BRIGHT 5-Year Follow-Up Study. J Clin Oncol. 2019 Apr;37(12):984–91.
Statement of Ethics
Consent from the patient was obtained for publication. This paper was conducted ethically in accordance with the Declaration of Helsinki.
Disclosure Statement
The authors have no conflicts of interest to declare.
Funding Sources
The authors have no funding sources to declare.
* Corresponding author.
Ammar Nassri, MD
University of Florida at Jacksonville
4555 Emerson Street, Building 1, Suite 300
Jacksonville, FL 32207 (USA)
E-Mail ammar.nassri@jax.ufl.edu
Received: October 11, 2019; Accepted after revision: November 26, 2019
Author Contributions
All authors made substantial contribution to the conception of the work and the acquisition of data for the work. R.N. drafted the work, contributed to the literature review, and revised it. A.N. drafted the work, contributed to the literature review, performed the endoscopies, and obtained the images. A.A. contributed to the literature review, revised the work, and interpreted the pathological slides. B.S.R. drafted and revised the work and performed the endoscopy. R.S. drafted and revised the work and performed the endoscopy. All authors critically revised the manuscript, approved the final version to be published, and agree to be accountable for all aspects of the work.














