Introduction
Myeloproliferative neoplasms (MPN) are characterized by clonal proliferation of hematopoietic stem cells, with overproduction of functional granulocytes, erythrocytes, and/or platelets. Classic MPN include polycythemia vera (PV), essential thrombocythemia (ET), primary myelofibrosis (PMF), and chronic myeloid leukemia. One major complication is the occurrence of venous and arterial thrombosis. There is a strong association between MPN and splanchnic vein thrombosis (SVT) [1].
SVT is a rare disorder that encompasses thrombosis formation in the portal, hepatic, splenic, or superior mesenteric venous systems. In a large multicenter study, the estimated incidence rate for portal vein thrombosis was only 0.7 cases per 100,000 patients per year [2]. In a recent meta-analysis, the prevalence of MPN in SVT was approximately 30% [3]. Given this strong association, screening for MPN is recommended when SVT is diagnosed [3].
Here, we report a rare case of a young male who presented with portal-splenic-superior mesenteric vein thrombosis that was found to be secondary to PMF in the prefibrotic stage. There is a limited number of cases of MPN presenting as SVT with such extension, especially at early stages. We also performed a literature review on this topic.
Case Presentation
A 31-year-old male with no significant previous medical history presented to the emergency department with a 3-week history of epigastric pain that radiated to the back and was aggravated after meals. He denied experiencing nausea, vomiting, diarrhea, gastrointestinal bleeding, weight loss, chills, or any other symptoms. Physical examination revealed epigastric tenderness, but without signs of peritoneal inflammation. His general state of health was good. He was afebrile and hemodynamically stable. Laboratory evaluation revealed mild thrombocytosis (a platelet count of 420,000 × 103/μL) associated with elevated levels of alanine aminotransferase (144 IU/mL), aspartate aminotransferase (93 IU/mL), lactate dehydrogenase (609 IU/mL), and C-reactive protein (47.2 mg/L). Hemoglobin, leukocyte count, urea, creatinine, bilirubin, alkaline phosphatase, γ-glutamyl transferase, amylase, lipase, and coagulation studies were all normal. Contrast-enhanced abdominal computerized tomography (CT) revealed extensive SVT involving the portal, splenic, and superior mesenteric veins (Fig. 1). Splenomegaly was also evident. No signs of chronic liver disease or suspicious liver nodules were identified.
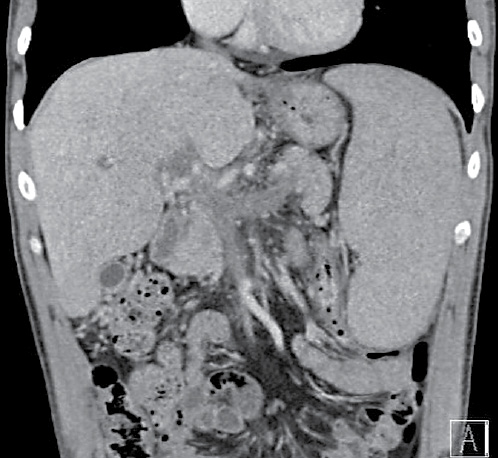
Fig. 1 Contrast-enhanced abdominal computerized tomography image demonstrating extensive splanchnic vein thrombosis involving the portal, splenic, and superior mesenteric veins. The thrombus extends through the splenic vein, almost reaching the splenic hilum, and involving the entire portal vein by extending into its left and right intrahepatic branches. Splenomegaly is also evident, with an enlarged spleen measuring 17.5 cm. No signs of chronic liver disease or suspicious liver nodules were identified.
He was admitted and started anticoagulation with warfarin with a target international normalized ratio (INR) of 2.0-3.0. The patient’s medical records revealed elevated platelet counts for at least 6 years, with a peak of approximately 673,000 × 103/μL 3 years previously. No significant changes in other blood cells were found. The possibility of a MPN was considered, and so analysis for the JAK2 V617F mutation and a bone marrow biopsy were performed. Lupus anticoagulant, anti-cardiolipin and anti-B2 glycoprotein antibodies were normal. α-Fetoprotein was also normal. Esophagogastroduodenoscopy revealed large esophageal (Fig. 2a) and gastric (Fig. 2b) varices. Primary prophylaxis for variceal bleeding with propranolol was started. INR was maintained inside the target range; after 21 days, he was discharged home anticoagulated with warfarin.
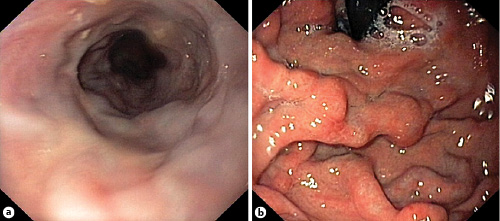
Fig. 2 Upper digestive endoscopy demonstrating signs of portal hypertension. a Large esophageal varices were identified at distal esophagus. b Endoscopic examination of gastric fundus revealed the presence of varices.
The patient was subsequently evaluated at a hematology consultation. Remarkably, the presence of the JAK2 V617F mutation was established. The bone marrow biopsy revealed hypercellularity, with increased proliferation of myeloid and megakaryocytic cell lines and an increased myeloid/erythroid cell ratio (Fig. 3a). There were no maturation abnormalities involving myeloid or erythroid cells. In contrast, atypical megakaryocytes with hyperlobulated hyperchromatic “cloud-like” nuclei were found (Fig. 3b). The reticulin fibers network was focally enhanced, without significant reticulin fibrosis. he was negative for the BCR/ABL1 mutation. These findings were consistent with PMF in a prefibrotic stage. Cytoreductive therapy with weekly subcutaneous pegylated interferon was started.
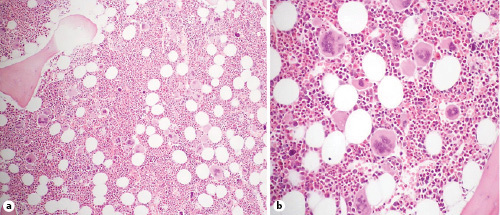
Fig. 3 Histopathological examination of the bone marrow. a Hypercellular bone marrow, with an increased number of myeloid cells and megakaryocytes. H&E. ×100. b Clusters of atypical megakaryocytes with hyperlobulated “cloud-like” nuclei. H&E. ×200.
Approximately 1 month after discharge, he was readmitted for abdominal pain. Abdominal CT still revealed portal-splenic-mesenteric venous thrombosis. Given the stability of the thrombus size despite anticoagulation and therapeutic INR, thrombolysis or shunt placement by interventional radiology were considered; however, the clot burden precluded these approaches. Despite an improvement in abdominal pain, repeat CT after 10 days revealed a stable thrombus size. He was discharged home maintaining warfarin and weekly pegylated interferon.
After these events, the patient evolved favorably with progressive resolution of his symptoms. He maintained anticoagulation and 5 months later, Doppler ultrasound revealed complete dissolution of the thrombus. After 14 months, the patient remains asymptomatic and maintains cytoreductive therapy for his hematological disease and anticoagulation with warfarin for the prophylaxis of recurrent thrombosis.
Discussion
MPN are the most common underlying prothrombotic disorder found in patients with SVT, in the absence of local factors such as liver cirrhosis or nearby malignancy. Risk factors for MPN-associated SVT include young age, female gender, concomitant hypercoagulable disorders, and the JAK2 V617F mutation. The pathophysiology of SVT is thought to derive from local interactions between atypical blood cells and aberrant splanchnic endothelial cells that increase the likelihood of thrombus formation. SVT may represent the first manifestation of MPN (particularly when the JAK2 V617F mutation is present) and, importantly, it can present with few overt MPN disease characteristics, possibly because it occurs during an early stage or because characteristic erythrocytosis or thrombocytosis may be masked by hypersplenism and hemodilution secondary to portal hypertension [1].
In a retrospective study involving 181 patients with MPN-associated SVT, the thrombus involved the portal vein in 60.3% of patients, the hepatic vein in 17.1%, the splenic vein in 13%, and the mesenteric vein in 10% [4]. Another retrospective study involving 84 patients demonstrated that MPN-associated SVT involved the portal vein in 69% of the cases, and that the thrombus extended to the mesenteric and/or splenic vein in < 20% of these patients [5]. Although the thrombus extension is usually limited to one of these venous systems, SVT involving more than one of the main splanchnic veins has rarely been described as the onset manifestation of MPN, including ET [6-10], PV [10, 11], and PMF [12] cases (summarized in Table 1). In some of these patients, the thrombus extended throughout the portal, splenic, and superior mesenteric venous systems [6-8, 10], similar to in our patient. This is a critical clinical situation that may result in severe complications. Abdominal pain resulting from increased venous pressure is almost universal and can be incapacitating. Other serious potential complications include variceal bleeding, ascites or hepatic encephalopathy resulting from portal hypertension [9, 13], splenic infarction secondary to splenic vein obstruction [6], and intestinal ischemia secondary to mesenteric vein obstruction [14].
Table 1 Summary of previous cases of splanchnic vein thrombosis involving >1 of the main splanchnic veins in association with myeloproliferative neoplasms
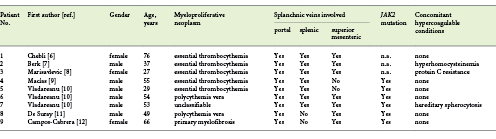
It is also interesting that, although some patients have concomitant hypercoagulable conditions like hyperhomocysteinemia [7] or resistance to activated C protein [8] which can contribute to the large thrombus size, in other cases, the single prothrombotic risk factor was the presence of the JAK2 V617F mutation [9-12], supporting an important role for this mutation in the development of extensive SVT. In one of these cases, SVT involving the portal and superior mesenteric veins was reported as the first manifestation of overt PMF [12]. Here, we also report extensive SVT in the form of PMF, but a larger thrombus size (portal, splenic, and superior mesenteric veins), albeit it was detected at an earlier stage of the disease (i.e., the prefibrotic stage). Notably, the patient also did not present with any concomitant hypercoagulable condition besides the JAK2 V617F mutation, which therefore appears to be an important prothrombotic risk factor that can, per se, induce the formation of a large thrombus.
There is little information regarding the variables associated with thrombosis risk in patients with prefibrotic PMF, translating into uncertainty regarding the stratification and management of these patients. Interestingly, in a large retrospective study involving 518 patients with MPN-associated SVT with a reported prevalence of the JAK2 V617F mutation of 90%, the thrombus involved >1 of the main splanchnic veins in approximately 30% of the patients, supporting the notion that JAK2 V617F mutation is per se an important risk factor for thrombosis [15]. However, more than one-third of cases of MPN-associated SVT show at least one coexisting risk factor/hypercoagulable disorder and, more recently, the presence of high-molecular-risk variants has also been associated with worse prognosis, even in patients with a JAK2-positive MPN [16].
Diagnostic criteria for PMF in the prefibrotic stage include: (i) proliferation of atypical megakaryocytes, without significant reticulin fibrosis, accompanied by increased bone marrow cellularity, granulocytic proliferation and, often, decreased erythropoiesis; (ii) not meeting the criteria for other MPN; (iii) the presence of the JAK2, CALR, or MPL mutation. Additionally, at least one minor criterion (anemia, leukocytosis, splenomegaly, or elevated LDH) must be present. Distinction from overt PMF is related to more marked reticulin fibrosis in the latter and from ET because, in this case, megakaryocytes are mature and only very rarely is there minimal reticulin fibrosis [17]. Based on these criteria, this case represents an uncommon clinical scenario where PMF in the prefibrotic stage presented with portal-splenic-superior mesenteric vein thrombosis as the onset manifestation.
It is important to highlight that the survival of patients with MPN-associated SVT is regarded as being influenced primarily by the natural history of the underlying neoplasm, rather than by the recurring thrombotic event and/or the consequences for the splanchnic circulation and liver function. This emphasizes the importance of making a clear distinction between ET and prefibrotic PMF. However, this may be challenging, since PMF in the prefibrotic stage can mimic ET in its presentation and mutation profile; careful morphologic examination is therefore necessary. While the initial presentation of PMF in the prefibrotic stage is often isolated thrombocytosis, thereby mimicking ET, the course of prefibrotic PMF may be highly symptomatic, and the outcome is worse than in ET. Patients with prefibrotic PMF have higher leukocyte and platelet counts, lower hemoglobin, higher lactate dehydrogenase levels, and more frequently display splenomegaly than patients with ET. In the largest series available, involving 661 patients with prefibrotic PMF, the median overall survival was 14.7 years, significantly shorter than the 30.2 years in a parallel cohort of 421 unselected patients with ET [18].
Therapy should be addressed by pursuing 3 main goals: (a) preventing recurrence of thrombosis; (b) man -aging the underlying MPN; and (c) supporting organ dysfunction, particularly of the liver. Management of thrombotic complications associated with MPN may be challenging. Although the risk of SVT is higher than in the general population, these patients are also prone to severe bleeding [19]. In general, patients with MPN-associated SVT should be treated promptly with low-molecular or unfractionated heparin followed by vitamin K antagonists, maintaining an INR in the ther apeutic range of 2.0-3.0. The optimal duration of treatment is unknown, but, in general, life-long anticoagulation with vitamin K antagonists is recommended. However, although this approach has demonstrated a clear benefit in preventing recurrent thrombosis compared to off-treatment, recurrences of SVT or other thromboses may still occur in 15-20% of patients [20]. Although promising, there is still insufficient evidence to support a role for direct-acting oral anticoagulants in this setting [21].
In asymptomatic patients with a low or intermediate risk of disease according to the IPSS/DIPPS/DIPSS classifications, there is no evidence to support the value of disease-modifying therapy and observation alone is recommended. In contrast, cytoreductive treatment is indicated for patients with an intermediate or high risk of disease, and hydroxyurea or ruxolitinib are usually the first-line drugs of choice [22]. Although our patient had a low risk according to IPSS/DIPPS, there are studies demonstrating a good clinical response and safety profile in low-risk PMF patients treated with pegylated interferon, with high rates of splenic response, an improvement in constitutional symptoms, and the resolution of leukocytosis and thrombocytosis, particularly in special populations including young and pregnant patients. In fact, interferon has been recommended as an option for symptomatic low- or intermediate-risk PMF patients by the National Comprehensive Cancer Network. Ultimately, the decision for which cytoreductive agent to use is based on individual factors such as comorbidities, risk status, symptom burden, age, and toxicity profile. In young patients, concern for hydroxyurea long-term effects associated with a known potential for durable responses, and disease modification associated with interferon led expert consensus guidelines to recommend interferon for first-line treatment in patients who are younger [23]. Based on these data, a decision to start cytoreductive therapy with pegylated interferon was taken.
In conclusion, although uncommon, MPN must be considered in the differential diagnosis of SVT, especially in the absence of cirrhosis or nearby malignancy. This case demonstrates that MPN may present with extensive SVT at its onset, even at early stages and in the absence of marked erythrocytosis, thrombocytosis, or concomitant hypercoagulable conditions. The presence of the JAK2 V617T mutation appears to be an important prothrombotic risk factor that can, per se, contribute to the development of large venous thrombosis.














