Introduction
Despite many advances in science in recent years, tuberculosis remains one of the leading causes of deaths worldwide, especially in developing countries [1]. Although pulmonary tuberculosis is the most common site of tuberculosis infection, abdominal tuberculosis is becoming more frequent, which includes involvement of the peritoneum, abdominal visceral organs, lymph nodes, and gastrointestinal tuberculosis [2]. In gastrointestinal tuberculosis, infection of the stomach and rectum is sparsely reported in the literature and has major limitations in its diagnosis [3]. We report 2 cases in immuno-competent and immunosuppressed patients who pre-sented gastric, duodenal, and rectal ulcers due to Mycobacterium tuberculosis infection.
Case Report
Case1
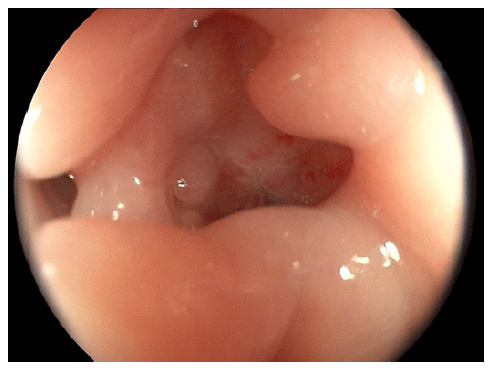
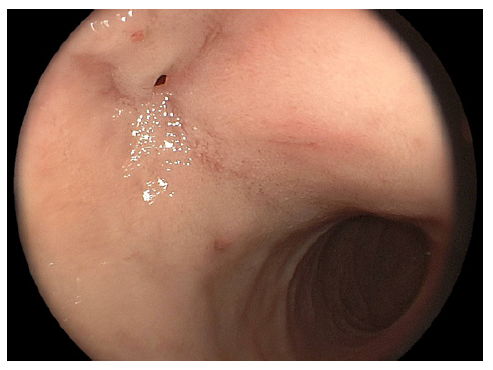
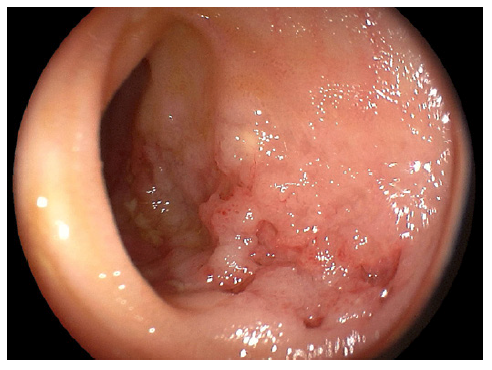
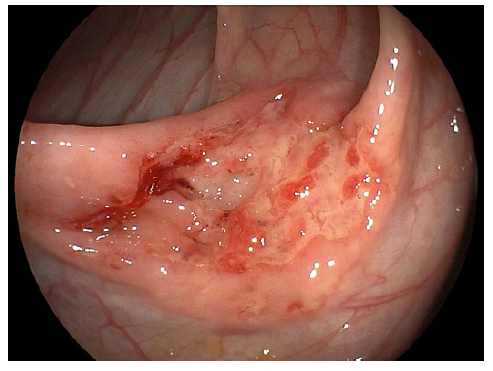
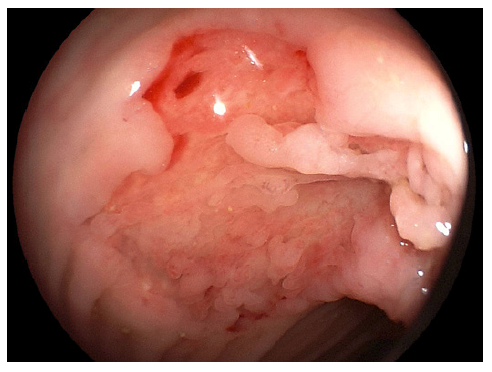
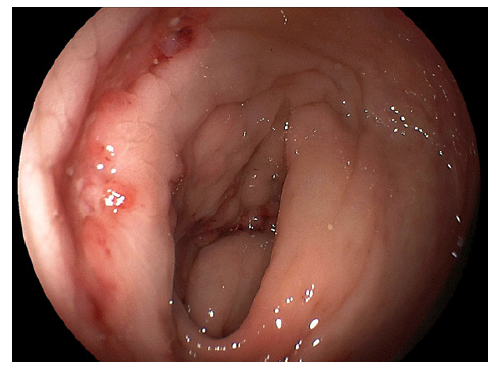
We present the case of a 17-year-old male who presented to the emergency department with a 2-year history of cough, chronic abdominal pain, intermittent nausea and vomiting, non-bloody chronic diarrhea, and progressive weight loss up to 40 kg, equivalent to 45% of his body weight. On admission, vital signs were stable. On examination, the abdomen was tender to palpation with diffuse mild pain. No rebound was identified. Laboratory findings demonstrated hemoglobin of 8.7 g/dL (N = 13.2-16.6), mean corpuscular volume of 73 (N = 80-100), mean corpuscular hemoglobin of 22 (N = 27-32), and severe hypoalbuminemia of 2.3 g/dL (N ≥3.5). Biochemical and liver profiles were unremarkable. Human immunodeficiency virus and human T-cell lymphotropic virus 1 and 2 tests were negative. Functional stool test including presence of mucus, blood, polymorphonuclear leukocytes, lactoferrin, fat strains, and serial parasite analysis revealed no significant abnormalities; however, the plain chest X-ray showed apical infiltrates in both hemithorax (shown in Fig. 1). In the abdominal ultrasound, mild hepatomegaly was found without ascites. Endoscopic exams were mandatory for the patient. In the greater curvature of the gastric body, an irregular circumferential ulcer was identified with a diameter of 10 mm, elevated margins and a base with high-density fibrin (shown in Fig. 2). In the anterior wall of the duodenal bulb, a 0.5-mm orifice was present from which a scarce whitish discharge was obtained (shown in Fig. 3). In the colonoscopy evaluation, multiple irregular circumferential ulcers were seen, which involved the terminal ileum (shown in Fig. 4), ileocecal valve, cecum, colon (shown in Fig. 5, 6), and rectum (shown in Fig. 7). Biopsies obtained from the upper and lower lesions were positive to M. tuberculosis in the GeneXpert (MTB/RIF) assay, and no resistance to Rifampicin was detected. Histopathology revealed chronic granulomatous inflammation with multinucleated giant cells. First-line antituberculosis drugs were initiated with good clinical improvement. Positive culture results were obtained after 2 months. Informed consent was obtained from the patient for case and image publication.
Case 2
A 43-year-old male with a 2-month diagnosis of acquired immune deficiency syndrome without treatment (viral load of 280,000 copies/mL and a CD4 count of 67 cells/mm3), presented with 6 months of intermittent fever, diffuse abdominal pain, progressive weight loss up to 10 kg (27% of his body weight), nausea, and vomiting. On examination, the patient was febrile, pale, and emaciated. Hemoglobin of 9.4 g/dL (N = 13.2-16.6), mean corpuscular volume of 86 (N = 80-100), mean corpuscular hemoglobin of 29.2 (N = 27-32), and a non-pathogenic intestinal parasite, Entameba coli, in the parasitology exam were identified. The chest X-ray and sputum analysis were negative for tuberculosis. Abdominal computed tomography revealed retroperitoneal lymphadenopathy, splenomegaly, and a fluid collection in the psoas muscle of 2 cm with no need for drainage. Due to gastrointestinal symptoms such as nausea and vomiting, we decided to perform an upper gastrointestinal endoscopy. A deep irregular ulcer of 8 mm with a fibrin base was seen in the gastric fundus (shown in Fig. 8). Acid-fast bacilli staining in the biopsy was positive for M. tuberculosis. With the administration of first-line antituberculosis drugs, the patient had an optimal clinical recovery. Informed consent was obtained from the patient for case and image publication.
Discussion
Gastrointestinal tuberculosis is the most frequent site of affection in abdominal tuberculosis and can be a diagnostic challenge as it can mimic other illnesses as inflammatory bowel diseases, neoplasms, ischemic ulcers, and other infections [3, 4]. The most common affected sites are the ileum, cecum, and proximal colon. Gastric tuberculosis is an uncommon site of infection due to gastric acid that acts as a protective barrier against microorganisms; nevertheless, a high bacterial load can produce direct injury leading to infection. Other routes of contamination are hematogenous, retrograde lymphatic spread, and direct contact with affected adyacent organs [3, 5]. Symptoms are usually unspecific, ranging from non-specific abdominal pain, dyspepsia, weight loss, and anorexia [3]. Gastric tuberculosis can manifest endoscopically as multiple or single irregular ulcers, fistula formations, and hypertrophic-stenosing lesions, which can produce gastric outlet obstruction [6]. Ulcers are usually located in the lesser curve of the prepyloric region and antrum; however, fundic ulcers have also been described in the literature [7]. Our patients presented ulcers in the gastric corpus and fundus. These lesions were probably related to retrograde lymphatic spread that, although uncommon, has been reported before [3]. Duodenal tuberculosis should be suspected in areas with high endemicity of tuberculosis and can share some symptoms of gastric tuberculosis. In endoscopy or radiological exams, luminal narrowing, mucosal ulcerations, or extrinsic compressions might be seen. In some cases, patients require surgery due to stenosis of the duodenum, and the diagnosis is made postoperatively [8, 9]. Colonic tuberculosis involvement is being more frequently reported in the last years. It mainly affects the ascending colon and cecum, and symptoms can be unspecific as abdominal pain or present with hematochezia or diarrhea. In colonoscopy, lesions can present as ulcers, nodules, polypoid lesions, or luminal narrowing. These lesions usually respond satisfactorily to antituberculosis treatment [10]. On the other hand, rectal tuberculosis is rare and usually associated with immunosuppressive states [11]. Patients can present symptoms such as chronic diarrhea, rectal bleeding, tenesmus, and perineal fistulas, which can mimic an inflammatory bowel disease or cancer [12]. Our patient presented a linear ulcer in the rectum; however, nodular, stenotic, and hypertrophic le-sions have been reported by authors before [11, 13].
Diagnosis is usually complicated due to the difficulty in getting an adequate tissue sample and the diagnostic test used. Different methods for diagnosis of gastrointestinal tuberculosis include acid-fast bacilli (AFB) staining, culture, GeneXpert MTB/RIF, and DNA-PCR. AFB is usually performed with the Ziehl-Neelsen method, is cost-effective, and has a specificity of 100%. However, the main disadvantage is its low sensitivity of around 31% [14]. Culture is the gold standard test; although it has a specificity of 100%, its sensitivity of only 9.3% can limit an accurate diagnosis [15]. GeneXpert MTB/RIF is a real-time PCR-based test that can also detect resistance to Rifampicin. Studies have shown high sensitivity values in biopsy samples ranging from 81.6 up to 95.7% and specificity from 78.9 to 100% [14, 16]. This test is considered one of the best for the diagnosis of gastrointestinal tuberculosis infection. Finally, DNA-PCR is also very effective with sensitivities up to 65% and specificity of 100% [14].
First-line treatment consists of isoniazid, rifampicin, ethambutol, and pyrazinamide. Administration of intravenous medication can be given in special situations as oral intolerance or adverse reactions [14]. Treatment length of 6 months has been suggested by most guide-lines; however, some studies have suggested longer treatment might have better outcomes. In a systematic review published in 2016, no benefits were found in 9-month regimens versus the standard 6-month regimen. Until now, new recommendations cannot be made [17].
Gastrointestinal tuberculosis can manifest as a primary infection or in association with pulmonary tuberculosis in disseminated states. The coexistence between gastrointestinal and pulmonary tuberculosis ranges from 15 to 25%. The rest are reported to be isolated gastrointestinal tuberculosis; however, this number might be under-estimated due to difficulties in diagnosing pulmonary tuberculosis. Treatment of pulmonary infection is adequate for gastrointestinal tuberculosis, and patients usually have a good prognosis [18, 19].
Gastroduodenal and colorectal tuberculosis, although rare, should be considered as a diagnosis in immunosuppressed and immunocompetent patients. An adequate tissue sample, which should be obtained from the edge and base of the ulcers, and appropriate diagnostic tests are essential for the diagnosis [20].