A 34-years-old woman, gravida 2, para 2, underwent Essure® (Bayer) hysteroscopic sterilization in 2009. The patient had dyslipidemia and venous insufficiency of the lower limbs. In childhood she had surgical history of ventriculoperitoneal shunt due to hydrocephalus. Her menstrual cycles were regular. She reported a normal and uneventful gynecological follow up.
After eleven years, at 45-years-old, she started abnormal uterine bleeding, intermenstrual and heavy menstrual, with no other relevant symptoms. Gynecological examination showed a cervical ectropion; bimanual exam was painless and pelvic masses were not found. Human Papilloma Virus testing was negative for high-risk genotypes.
Transvaginal ultrasound was performed and revealed the presence of a hyperechogenic structure, with a coiled shape, into the endometrial cavity. There was a similar structure in the right uterine horn, but it was absent in the left side (Figure 1). These findings suggested intrauterine migration of the left Essure® microinsert.
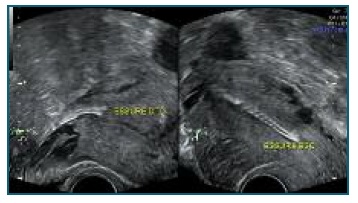
Figure 1 Transvaginal ultrasound - right Essure® well positioned (transverse plane of the uterus); Left Essure® in intracavitary position (longitudinal plane of the uterus).
Office hysteroscopy was undertaken to remove the intracavitary microinsert component. The procedure was successful despite the limited visualization caused by uterine bleeding (Figure 2), with minimal patient discomfort.
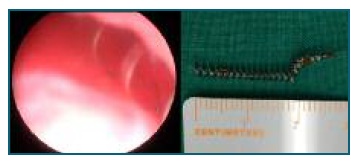
Figure 2 Hysteroscopy - Left Essure® in intracavitary position (limited visualization due to uterine hemorrhage). Observe spiral shape.
At the 3-month follow-up she was asymptomatic and reported regular menstrual cycles.
Hysteroscopic sterilization with Essure® was performed in Portugal from 2002 to 2017. The system consisted of double nitinol coils, a component of polyethylene terephthalate and stainless steel which were placed in each fallopian tube, by hysteroscopy1. The device promoted a local inflammatory response culminating in the development of fibrosis and tubal occlusion through foreign body reaction2. Essure® was an effective sterilization method with the advantage of being easily inserted in an office setting, not requiring general anesthesia neither abdominal incisions. It was specially counselled in women with significant comorbidities when other contraceptive methods were contraindicated1.
Over the years of Essure® use several serious long-term complications have been described, such as, development of nickel allergy (in susceptible individuals due to chronic antigen exposure) (3, migration to the abdominal cavity (after perforation), migration to the uterine cavity and malposition or expulsion2. Abdominal pain, pelvic inflammatory disease and abnormal uterine bleeding due to malposition and chronic inflammation are other reported adverse events3. Moreover, these occurences are almost always indications for removal of the device4. Depending on complications and associated injuries removal of Essure® can be performed by hysteroscopy or laparoscopy5. If the first route is feasible, there is still controversy in removing simultaneously the well positioned component of the device. When laparoscopy is an option, two types of procedures are suggested, salpingostomy with removal of the devices or salpingectomy and cornuectomy. In case of failure of less invasive procedures, hysterectomy with bilateral salpingectomy, could be an option4.
With this report, the authors intent to draw attention to long-term complications of a device still carried by a significant number of women and show the importance of knowing the ultrasound imaging in its normal and abnormal placement.
For the clinical case report patient informed consent was obtained.














