Case Presentation
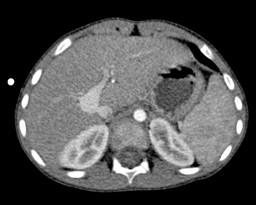
A 4-year-old male child presented was referred to our department with a history of an incidental finding reported on an abdominal ultrasound which was performed during the diagnostic workup of an interauricular communication. The patient was asymptomatic. For further characterization of the aforementioned finding, a contrast-enhanced computed tomography (CE-CT) with multiplanar and maximum intensity projection reformation (MIP) was performed, followed by digital subtraction angiography (DSA).(Fig. 1)
Discussion
Congenital extrahepatic portosystemic shunt (CEPS), also known as Abernethy malformation, refers to an abnormal congenital communication between the portal and systemic venous systems in which the intestinal and splenic venous blood bypasses the liver.1,2 It is a rare condition - less than 300 cases reported in the literature - and is usually associated with other congenital anomalies, most frequently genitourinary malformations, cardiac abnormalities and musculoskeletal defects. (Fig. 2)2,3,4,5
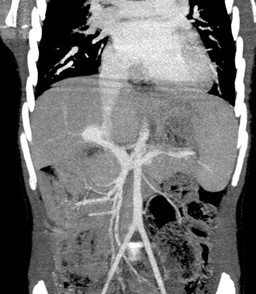
Figure 2: Maximum intensity projection (MIP) coronal CT reconstruction. The extrahepatic portal anatomy and the abovementioned abnormal portosystemic shunt are visible.
The above-mentioned alterations in the splanchnic circulation produce hemodynamic and physiological changes that explain the clinical spectrum of this condition.6,7,8,9,10,11,12 Hepatic encephalopathy (caused by the neurotoxicity from ammonia and other digestive toxins) is the main complication. Other complications include hepatopulmonary syndrome and pulmonary arterial hypertension (due to non-metabolized intestinal vasoactive mediators that induce vasodilation or vasoconstriction in the pulmonary vascular bed) and nodular hepatic lesions (in the setting of developmental, functional and regenerative changes in the liver that result from the deviation of hepatotrophic substances and increased arterial hepatic influx). There are also completely asymptomatic forms, which are detected incidentally or demonstrate increased sensitivity to hyperammonemia.(Figs. 3, 4, 5)
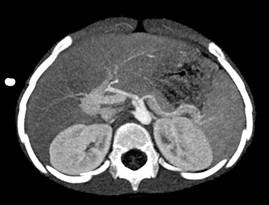
Figure 3: Maximum intensity projection (MIP) axial CT reconstruction. The abnormal portosystemic shunt is depicted as well as several hypoplastic but patent intrahepatic portal venous branches.
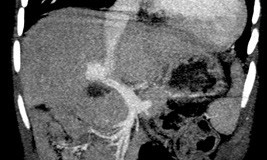
Figure 4: Maximum intensity projection (MIP) coronal CT reconstruction. The portosystemic shunt is demonstrated in the coronal plane.
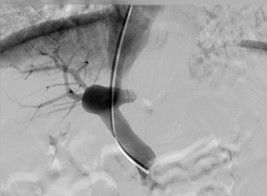
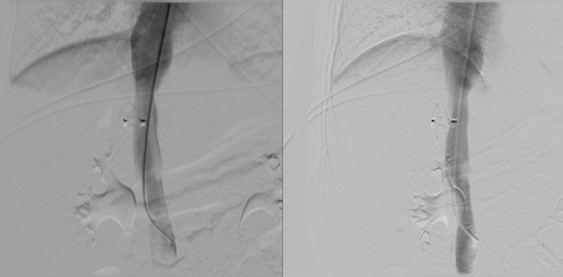
Figure 5: Direct portography via the abovementioned portosystemic shunt and through internal jugular vein access. Note the tip of the catheter at the proximal portion of the portal vein and the portosystemic shunt at its distal portion.
Liver nodules have been previous described in as much as 40% of patients with Abernethy malformation, with the majority being focal nodular hyperplasia and regenerative nodular hyperplasia (70%) while hepatocellular carcinoma and adenomas have a lower proportion (20% and 10%, respectively).6,11,12,13 However, a recent international observational study (the largest cohort of CEPS patients until now and the only one in which most patients reached adulthood at the end of follow-up)14 has shown higher overall incidence of liver nodules (65%) and a higher proportion of hepatocellular carcinoma and adenomas. The authors attribute these worrisome results to the older age of the patients included in the study and to the better imaging accuracy in recent years.
Congenital extrahepatic portosystemic shunt is anatomically classified in two types:1,2
In type 1 CEPS there is an end-to-end portocaval shunt with complete absence of intrahepatic portal venous branches, which results in a complete bypass of the splanchic blood. These shunts are further classified in type 1a (separate drainage of the superior mesenteric and splenic veins into the inferior vena cava) and type 1b (superior mesenteric and splenic veins form a common trunk which drains into the inferior vena cava).
In type 2 CEPS there is a side-to-side portocaval shunt with hypoplastic but patent intrahepatic portal venous branches, which produces a partial bypass of the splanchic blood.
This anatomical classification has classical therapeutic implications: type 1 CEPS are initially managed with follow-up and conservative treatment, with liver transplant as the only curative treatment; type 2 CEPS can be treated with shunt closure, either surgical or endovascular. It is worth noting that multiple reports of cases classified as type 1 CEPS at CT or MRI were shown to actually have intrahepatic portal vein flow when further studied by angiography with a shunt occlusion test.13,15,16,17,18,19 This is explained by the presence of hypoplastic intrahepatic branches that are not depicted in cross-sectional techniques such as CT or MRI and reiterates the importance of the shunt occlusion test in the diagnostic workup of these patients, particularly when evaluating possible therapeutic options (previously diagnosed type 1 CEPS may become eligible for shunt closure).
In this case endovascular shunt closure was perfomed with an Amplatzer™ Septal Occluder (waist diameter: 6 mm; discs diameter: 14 mm and 16 mm. The post-treatment angiography revealed complete exclusion of the shunt (Fig. 6).