INTRODUCTION
Cryptococcosis is a systemic infection caused by Cryptococcus neo-formans (C. neoformans), an opportunistic encapsulated yeast with worldwide distribution.1,2Infections occur mainly in immunocompromised individuals and symptoms vary according to the integrity of the immune system, ranging from asymptomatic respiratory infection to severe pneumonitis, which can spread to the central nervous system and other tissues, such as skin, prostate or bones.3
The authors report a case of extensive nonspecific cutaneous manifestations of cryptococcosis in a patient with chronic obstructive pulmonary disease, systemic arterial hypertension and dyslipidemia, without the use of immunosuppressants and without evidence suggesting immunosuppression.
CASE REPORT
A 76-year-old male patient was observed in November 12, 2020 at the medical outpatient clinic (AME) in the city of Peruíbe, São Paulo, Brazil, reporting intense pain, edema, erythema and heat, progressing for a week, located on the dorsum of his right hand and all over the left forearm, with some tense blisters. He denied fever (Fig. 1).
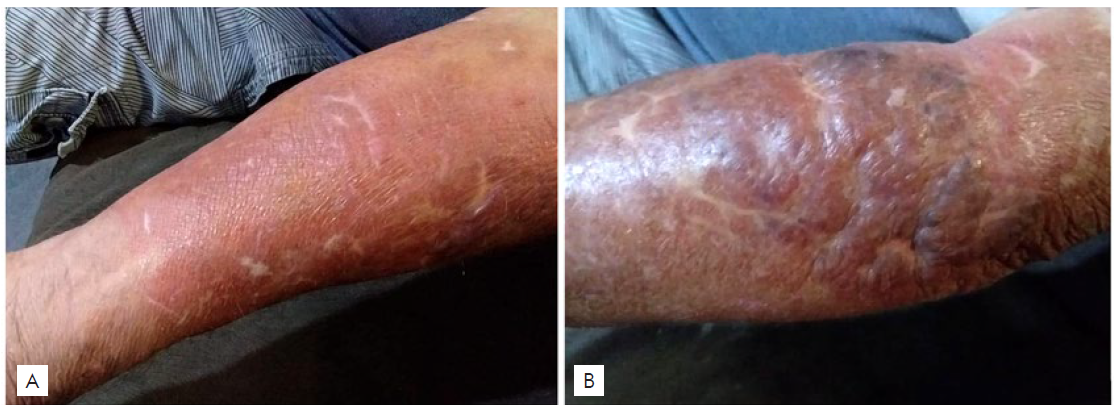
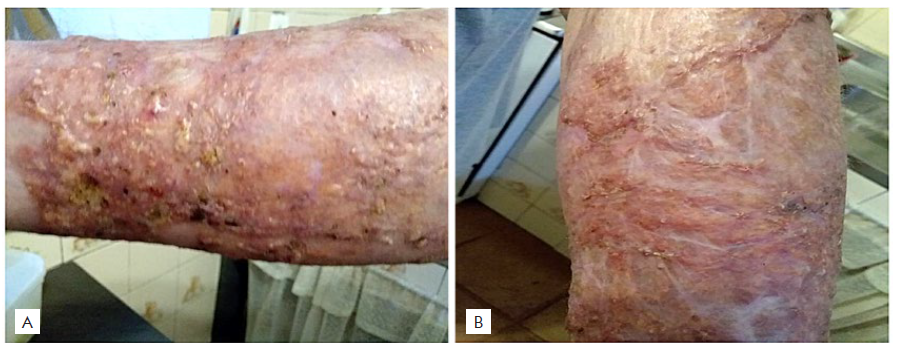
Figure 1 Initial cutaneous lesions: erythematous plaque in the left forearm (A) with a more violaceous and infiltrated areas and a flaccid bullous appearance.
In November 19, the initial erythematous plaque lesions on the hand and forearm worsened despite the administration of 1 g of intravenous ceftriaxone for 7 days and daily dressings with collagenase and chloramphenicol. The lesion extended to the elbow and arm, became more hyperpigmented with more bullae and excoriated/exulcerated areas with granulation tissue (Fig. 2). As lesions became more painful, oral gabapentin (300 mg of three times a day) was prescribed. During an incisional biopsy on the most flaccid bullous lesions, there was exudation of gelatinous and bloody material. His-tology revealed epidermal acanthosis and foci of exulcerations, and, in the dermis and hypodermis, there was exuberant edema, vascular ectasias, recent hemorrhage and mixed interstitial infiltrate, rich in lymphocytes, histiocytes and neutrophils, surrounding numerous oval yeast structures with a thick capsule, measuring between 4 and 10 microns (Fig. 3). Mayer's Mucicarmine staining (Fig. 4) showed the presence of a thick mucoid capsule, confirming the histopathological diagnosis of cutaneous Cryptococcosis.
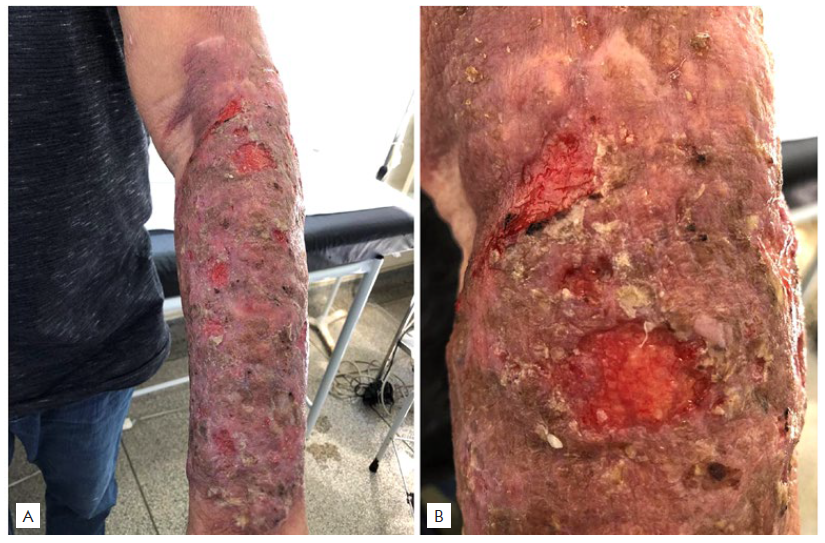
Figure 2 Late lesions after two weeks of the initial lesions. a) after biopsy, forearm with erythema and skin hyperpigmentation, with excoriated and crusted areas; b) lesions with a reddish, granular and crusted bottom.
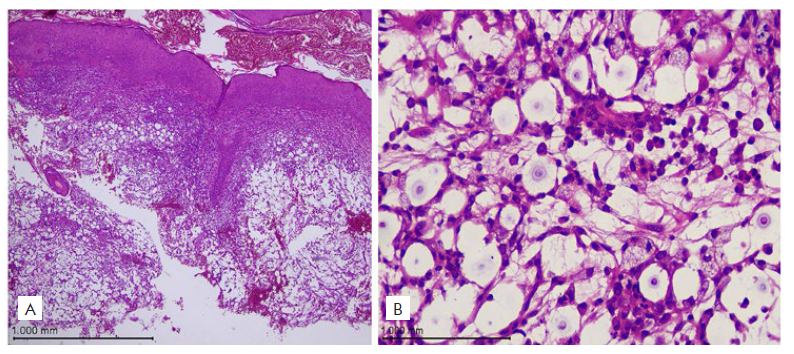
Figure 3 Histopathology, H&E stain. A) Epidermis with acanthosis and dermis with edema, vascular ectasias and foci of recent hemorrhage (40x); B) On the right, mixed inflammatory infiltrate and edema, observing oval structures with a double oval capsule (400x)
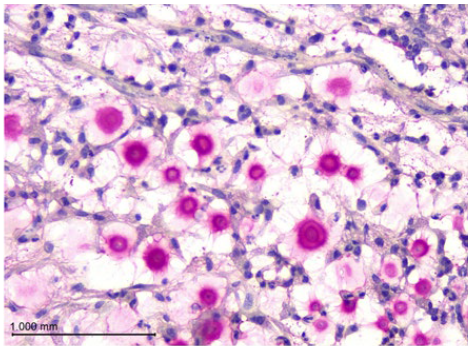
Figure 4 Mayer's Mucicarmine staining. Yeast structures stained in magenta, showing thick double capsule rich in mucin (400x).
After etiological confirmation, the patient initiated oral fluconazole (300 mg/day). A previous contact with pigeon feces was confirmed and the authors confirmed once more, that the patient was not under treatment with immunosuppressive drugs and had no evidence of immunosuppression. Blood cultures for Cryptococci were negative as well as serologies for HIV, hepatitis B and C and a complete blood count, coagulation tests, serum electrolytes, blood cultures, urinalysis, renal and hepatic function were normal. Nasopharyngeal swab for SARS-CoV-2 was negative.
Computed tomography of the chest without contrast showed traces of centrilobular and paraseptal emphysema in the upper fields, bronchial and bronchiolar inflammatory changes, related to chronic smoking. There was no mediastinal or hilar lymph node enlargement. Sparse calcified atheroma involved also the coronary (Fig. 5).
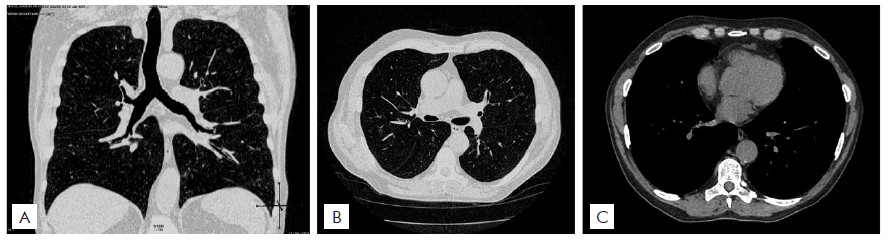
Figure 5 Computed tomography of the chest with traces of centrilobular and parasseptal emphysema in the upper fields, without other specific alterations.
After one month of fluconazole therapy (300 mg/day), blisters and ulcers resolved with crusts, residual erythema and atrophy, still with diffuse infiltration (Fig. 6). A new biopsy performed on the forearm revealed still the presence of polymorphonuclear leukocytes, without observation of microorganisms. Culture was also already negative for aerobic, anaerobic bacteria, fungi, mycobacteria and acid-alcohol resistant bacillus.
Fluconazole therapy was kept for 3 months with full recovery of the lesions. After 4 months of the end of the treatment, the patient is still doing well and is under monthly surveillance, with no evidence of disease.
DISCUSSION
Epidemiology and Etiologic Agent
Cryptococcosis is a systemic fungal infection, caused mainly by Cryptococcus neoformans (C. neoformans), an encapsulated opportunistic yeast that has worldwide distribution, found mainly in soil, decomposing wood, pigeon feces, fruit and vegetables. After the diagnosis was confirmed, the patient reported previous contact with pigeon feces, which contributes to the recognition of a possible source of infection.1,2
The yeast has a polysaccharide capsule which is an important virulence factor, allowing its visualization with Indian ink, methylene blue and Mayer's mucicarmine.
There are four serotypes: serotype A, represented by C. neoforans var. grubii, with a global distribution. Serotypes B and C, represented by C. neoformans var. gattii, are limited to tropical and subtropical areas, and serotype D represented by C. neoformans var. neoformans, is found mainly in Europe.1
The pathogens Cryptococcus neoformans and Cryptococcus gatti currently represent the main worldwide cause of fungal meningitis.1 C. neoformans mainly infects patients with low CD4+ T cell counts, while C. gatti also infects immunocompetent individuals.4,5
Cryptococcosis is rare in the population without HIV infection, or when there is no other state of immunocompromise, such organ transplantation, neoplasms or in chronic immunosuppressive treatment.6
Clinical features
Symptoms vary according to the functionality of the immune system. The pathogen, upon reaching the airways, can present as an asymptomatic respiratory infection to severe pneumonitis and acute respiratory failure. Chest radiographs can reveal unilateral, nodular or cavitary infiltrates.8,9From the lung infection it can spread to the central nervous system and other sites, such as skin, prostate or bones.3 Central nervous system involvement is more common in immunocompromised patients, and symptoms are nonspecific, including headache, memory loss, tremor, muscle weakness, disorientation and confusion.
There are, in the literature, cases of familial cryptococcosis, related to inherited immunodeficiency diseases.10,11The disease is more frequent among men, which may suggest protective effects of estrogen.7
Cutaneous manifestations
Cutaneous cryptococcosis occurs mainly due to hematogenous dissemination. It affects about 20% of patients with disseminated cryptococcosis. C. neoformans serotype D, due to its dermatotropism, is associated with an increased risk of developing the cutaneous form.12
There is a wide spectrum of dermatological manifestations, including acneiform lesions, ulcerations, nodules, domeshaped papules with central umbilication, and it may also present as bacterial cellulitis, as in the present case, as sarcoidosis-like or panniculitis.13,14
AIDS patients commonly present with molluscum contagiosum-like lesions, which may suggest one of the first clinical signs of infection spread.7 Skin involvement in cryptococcosis also frequently occurs in liver transplant recipients using tacrolimus.1
The patient described did not have any evidence of immunosu-ppression, however he presented an extensive form of cutaneous involvement, without clinically or radiologically systemic signs, which demonstrates the importance of considering cryptococcosis as a differential diagnosis of skin lesions regardless of the immunological status of the patient.
Diagnosis
The diagnosis can be made by performing a skin biopsy with histopathological examination, microscopic evaluation and tissue culture. C. neoformans can be grown in various microbiological media with temperatures between 3ºC and 30ºC, with a maximum tolerance of 40º C.15 Mucoid colonies and budding cells similar to encapsulated round yeasts with 5 µm to 10 µm in diameter can be found.16 After staining with Hematoxylin and Eosin, a clear space can be observed around the cells, due to the capsule, which is stained with Alcian Blue or Mayer's Mucicarmine, the latter used in this patient’s skin sample.
Infection can also be confirmed by biochemical tests such as the cryptococcal polysaccharide antigen and the fungus can be detected in the cerebrospinal fluid (CSF) and blood through the latex agglutination test or ELISA.17
Disseminated cryptococcosis is confirmed by a positive culture from at least two different sites or a positive blood culture.18,19Blood cultures of our patient were negative and the culture of the lesion after the treatment was also negative.
Treatment
Treatment depends on the patient's immune status and the location of the infection. It is important to rule out involvement of the central nervous system in disseminated cryptococcosis as, because of the risk of meningoencephalitis, the drugs used must penetrate the blood-brain barrier. 20
In the guidelines for the management of disseminated cryptococcosis from the Infectious Diseases Society of America (IDSA), 2010, induction therapy is performed for 2 weeks, using liposomal amphotericin B at a dose of 3-4 mg/kg/day intravenously or amphotericin B lipid complex 5 mg/kg/day in combination with flucytosine 100 mg/day followed by consolidation therapy using fluconazole at a dose of 400-800 mg daily for 8 weeks, which should be reduced to 200-400 mg/day and continued for 6 months to 1 year.20
Recent protocols describe the management for immunocompetent patients, but do not include a non-disseminated exclusive cutaneous form of the disease.21
Unfortunately, mortality in patients with cryptococcosis remains high, as patients are normally already in a deficient immune status, and drug resistance may occur due to changes in the target enzyme encoded by the ERG11 gene, lanosterol 14α-demethylase, and the growth of biofilm. Research regarding new antifungal agents, immunomodulators, and other drugs that can be used as adjunct to the existing therapy is currently being carried out.22,23
A clinical improvement was noticed in the reported patient with the use of a intermediate dose of fluconazole, between 200-400 mg/day for 3 months, without the administration of amphotericin B because the patient had the non-disseminated form of the disease. A pos-sible hypothesis for a good prognosis of this case may be attributed to a localized cutaneous form of the disease and the healthy immune status of the patient, which helped to control the infection.
CONCLUSION
Cryptococcosis is a fungal infection with a wide spectrum of clinical manifestations that can evolve to systemic involvement and death. Skin lesions are nonspecific and varied, occurring in approximately 20% of patients, but are usually indications of the disseminated form of the disease. Despite being an opportunistic infection, affec-ting more commonly immunocompromised patients, it is extremely important to consider this condition in the differential diagnosis of the most varied dermatological manifestations regardless of the patient's immunological status, thus avoiding a late diagnosis and progression to a poor prognosis.