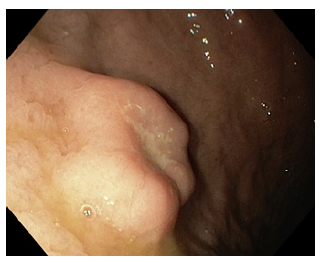
A 59-year-old man with unremarkable medical history underwent upper endoscopy evaluation due to dyspeptic symptoms. A 20 mm ulcerated subepithelial lesion was found in the greater curvature of the gastric body (Fig. 1). Biopsies showed only chronic active gastritis, without specific changes. After therapy with proton pump inhibitors, a subsequent endoscopy showed complete resolution of the superficial ulceration and an endoscopic ultrasound (Fig. 2) showed a well-defined heterogeneous, hypoechoic mass originating in the second layer of the gastric wall, with small anechoic areas. Ultrasound-guided fine needle aspiration (22 G FNA needle) and bite-on-bite biopsies were again unremarkable. The patient opted for addition follow-up and further attempts at tissue acquisition as he did not intend surgery with an uncertain diagnosis. The lesion was followed up by endoscopic ultrasound (at 3 and 6 months) and endoscopy (a year after the last endoscopic ultrasound), remaining stable for 3 years, with unremarkable histology (two instances of fine needle aspiration and one instance of bite-on-bite endoscopic biopsies). Due to an absence of diagnosis, the patient finally opted for surgical removal (opting against endoscopic removal). The surgical specimen showed overlying eroded mucosa, subepithelial glandular proliferation, cystically dilated, without cytologic atypia (Fig. 3). Overall, these findings were suggestive of gastritis cystica profunda. Epstein-Barr virus serologies were obtained and were not consistent with current infection.
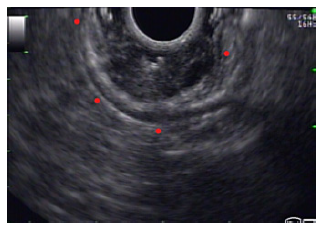
Fig. 2 Endoscopic ultrasound demonstrated a well-defined heterogeneous lesion originating in the second layer of the gastric wall. Small anechoic cysts can be seen inside the lesion.
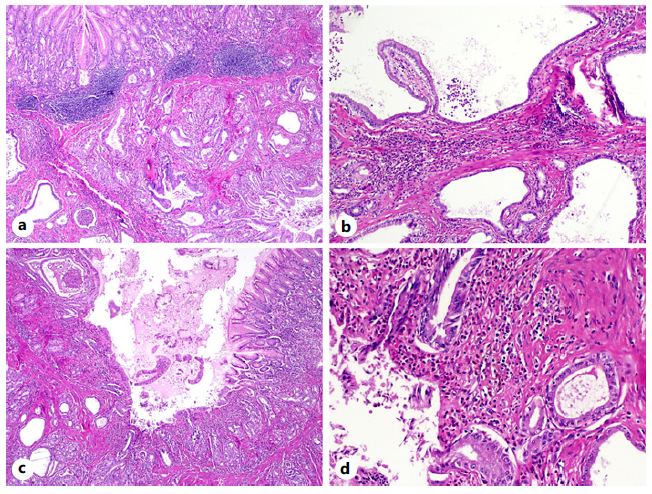
Fig. 3 On hematoxylin and eosin staining (H&E), a glandular proliferation was found within the submucosa of the gastric body (H&E, ×20) (a). Some of these glands were cystically dilated, lined by foveolar epithelium and without significant cytologic atypia (H&E, ×40) (b). The overlying gastric mucosa displayed foci of erosion and ulceration (H&E, ×20) (c), in association with a mixed inflammatory infiltrate and hemosiderinladen macrophages (H&E, ×100) (d). These features are suggestive of gastritis cystica profunda.
Gastritis cystica profunda is a rare form of subepithelial gastric lesion, typically found in post-gastrectomy patients, with around 65 cases described in the literature [1, 2]. In postsurgical patients, it is thought to arise from subepithelial migration of epithelial cells after surgical mucosal disruption. In patients without surgery, other factors such as EBV infection, ischemia, and chronic inflammation have been proposed but the underlying mechanism is unknown [3]. Animal studies show that GCP has malignant potential but the data on humans are still scarce [4]. The most frequent endoscopic ultrasound features are anechoic cysts in the submucosal layer and GCP should be suspected in this setting [2]. Although the sparse evidence available demonstrates a subpar diagnostic yield for endoscopic ultrasound fine needle aspiration, tissue sampling is still useful to exclude differential diagnosis such as gastrointestinal stromal tumors [2].
It could be argued that by utilizing larger caliber or FNB-type needles we could increase the diagnostic yield but there is no published evidence to support these claims. The diagnosis is nevertheless difficult as there are no defined diagnosis criteria; clinical, radiological and imaging results have to be integrated; nevertheless most patients are only diagnosed after surgery. In our case, this was further complicated as the patient had no history of gastric surgery, the only proven risk factor. Additionally, we found no evidence of further gastric disease, systemic disorders, or medication known to impact gastric mucosa.