Introduction
Cannabis is the most used regulated substance in the world, and its consumption continues to increase, counting 183 million consumers [1]. Cannabinoid hyperemesis syndrome (CHS) is characterized by paroxysmal and recurrent episodes of nausea, vomiting and abdominal pain in chronic cannabis users described for the first time in 2004 by Allen et al. [2]. A cohort study by Habboushe et al. [3] in patients admitted to the emergency department observed that 32.9% of self-reported frequent marijuana users met a CHS definition. Symptomatic relief with hot water baths is a specific sign of this condition (91-92.3% of cases) [2-4].
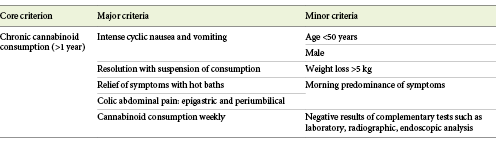
Several diagnostic criteria have been proposed (Allen et al., Sontineni et al., Simonetto et al., Pattathan et al., Sun et al., Cheung et al., Morris and Fisher) [2, 5-11]. Simonetto’s criteria (2012) are the most consensual, we have summarized them in Table 1. More recently, other criteria have been proposed, namely Roma IV criteria for cannabinoid hyperemesis (2016) and Venkatesan’s criteria (2019) [6].
Case Report
A 42-year-old previously healthy man was admitted several times to the emergency room within a 3-month period with complaints of generalized abdominal pain, epigastric region involuntary guarding, nausea and vomiting that improved with hot baths. During these admissions, he was examined by the internal medicine, gastroenterology and general surgery departments, undergoing several diagnostic exams (blood work, abdominal and thoracic X-ray, endoscopy and colonoscopy) that gave normal results. For symptomatic relief, several drugs were prescribed in ER: 40 mg of esomeprazole; 10 mg/2 mL EV metoclopramide; 8 mg/4ml EV of ondansetron and after discharge: 5 mg of chlordiazepoxide + 2.5 mg of clidinium bromide. Only partial and transient improvement was obtained, leading to frequent ER readmissions. To rule out a possible psychosomatic etiology, a psychiatric observation was requested. During the clinical interview, no other symptoms were reported and, at the examination, only mild anxious mood and naturalist (non-psychotic) beliefs were objectified. The patient reported a regular use of cannabinoid products (approximately 3 g, 12 units per day, smoked, prepared from marijuana leaves), for more than 5 years, with a recent exacerbation of consumption secondary to work-related stress. Due to the clinical history (onset of symptoms more than 6 months prior to observation, episodic vomiting, presentation after prolonged, excessive cannabis use and reliving of symptoms with hot baths), fair normality of the observation and the absence of major finds in the diagnostic exams performed, a working diagnosis of CHS was made. A brief psychoeducation, explaining the risks associated with consumption and its relationship with the clinical symptoms was performed, leading to the cessation of cannabinoid use and a complete resolution of the symptoms in the following days. No new episodes were reported during the following year.
Discussion
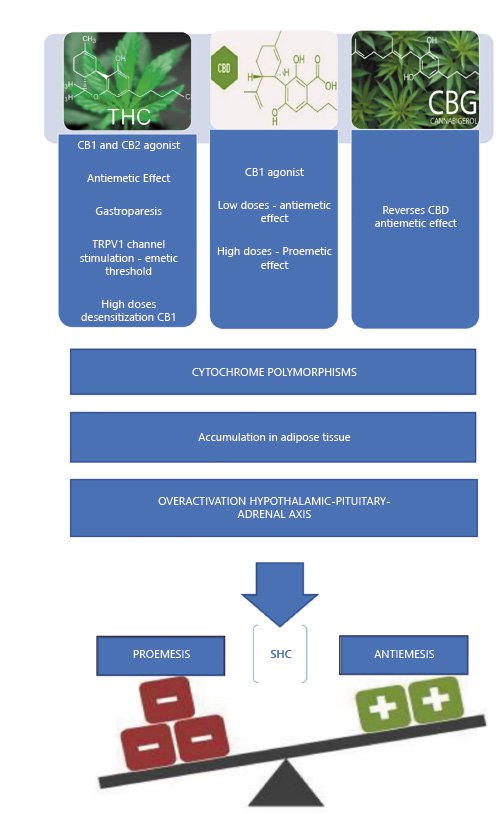
Cannabinoids bind to cannabinoid receptors CB1 and CB2. CB1 are located in the central nervous system (CNS) and at nerve endings along the gastrointestinal system, modulating gastric secretion, motility and inflammation. CB2 are located in the lymphoid tissues and are involved in the regulation of the immune system. Activation of CB1 by endogenous cannabinoids results in inhibition of the hypothalamic-pituitary-adrenal axis (HPA) and the sympathetic nervous system response to stress stimuli.
The occurrence of CHS is somewhat paradoxical since the main psychoactive component of marijuana THC (tetrahydrocannabinol-9) is an effective antiemetic. The antiemetic effect results both from the activation of the CB1 receptors (central) and consequent non-competitive inhibition of 5HT3 serotonergic receptors (emetogenic), and the release of serotonin via pre-synaptic CB1 receptors. The CB1 activation in the medulla oblongata inhibits gastric motor function and proemetic dopamine activity in the CNS.
Several theories have been proposed to explain this paradoxical relationship, namely [12-17]: (1) accumulation of cannabinoids (THC) or their metabolites in the CNS and fat tissue; (2) extreme CB1 stimulation may desensitize the receptor; (3) role of other cannabinoids; (4) cytochrome P450 genetic polymorphisms, altering the metabolism of cannabinoids; and (5) deregulation of the normal functioning of the hypothalamic-pituitary-adrenal axis and the sympathetic nervous system in response to stress and disruption of the endocanabinoid system.
In summary, CHS probably results from the combination of factors described above that promote emesis (Fig. 1). CHS is usually divided into three phases, pre-emetic or prodromal (lasting for months or years - early nauseas, abdominal discomfort), hyperemetic (intense and persisting nausea, episodic vomiting, diffusing abdominal pain), and the recovery phase. CHS frequently implies recurrent evaluations in the emergency department as a result of difficulty in controlling vomiting, which leads to several complementary diagnostic tests. Nevertheless, most of the performed complementary diagnostic tests have unspecific results (laboratory tests may show hydroelectrolytic alterations and leukocytosis in the emetic phase), making differential diagnosis difficult. Patients are often subjected to various cycles of antiemetics with poor response and potential adverse effects. The main complications of CHS include renal failure, hydroelectrolytic disorders, esophageal injuries and pneumomediastinum [13].
Regarding the differential diagnosis, CHS shares several similarities with the cyclic vomiting syndrome and it can be hard to differentiate them. Both syndromes are characterized by cyclic stereotypic episodes of vomiting but, according to the Rome IV criteria, in CHS a chronic and excessive cannabis use is present and the relief of vomiting episodes is achieved by sustained cessation of its consumption[14].
The treatment in the acute phase (emetics) involves supportive measures (hydration, antiemetics, etc.), which are generally not very effective.
The evidence of pharmacological treatment is limited, with benzodiazepines being the drugs most reported to be effective in the acute treatment of CHS, followed by haloperidol and other antipsychotics. Topical capsaicin (in the form of a skin patch, transdermal patch or ointment), which works by promoting analgesia via inactivation and desensitization of sensory neurons, has showed promising results [15-17].
A systematic review by Richards et al. [16], with 63 eligible studies and a sample of 205 individuals, demonstrated that quality evidence for the treatment of CHS is limited. Studies with level of evidence V (n = 44), benzodiazepines, metoclopramide, haloperidol, ondansetron, morphine and capsaicin were identified as effective treatments in the acute phase and tricyclic antidepressants in the maintenance phase. Nevertheless, the only known effective treatment of basic pathology (CHS) is a complete suspension of cannabinoid consumption. Otherwise, the probability of recurrence is high [18, 19].
Research on this topic is at an early stage, lacking more robust scientific evidence for better diagnostic and therapeutic definition.
Conclusions
This is an illustrative case of a poorly reported clinical entity, despite having a probable significant and increasing prevalence. With this case report, we want to raise awareness in order that clinicians identify and properly manage these cases, avoiding multiple emergency department admissions and redundant complementary diagnostic exams.
There is an increasing prevalence of CHS which is correlated to the worldwide increasing use of cannabis. However, it is still a rare clinical entity that most clinicians are not aware of, resulting in underdiagnosing and late diagnosis. Cannabis consumption should be ruled out, especially in young patients with unexplained vomiting, to avoid overlooking CHS.