Serviços Personalizados
Journal
Artigo
Indicadores
Links relacionados
Compartilhar
Ciência e Técnica Vitivinícola
versão impressa ISSN 0254-0223
Ciência Téc. Vitiv. vol.26 no.1 Dois Portos 2011
The Impact Of Nitrogen On Yeast Fermentation And Wine Quality
Ana Mendes-Ferreira, Catarina Barbosa, Patrícia Lage and Arlete Mendes-Faia
Institute for Biotechnology and Bioengineering, Centre of Genomics and Biotechnology, (IBB ⁄ CGB-UTAD), Universidade de Trás-os-Montes e Alto Douro, Vila Real, Portugal
ABSTRACT
Wine aroma is composed of primary, or varietal, aromas that arise directly from the grapes; secondary, or fermentation aromas, aromas produced by yeasts during the alcoholic fermentation; and the tertiary, or maturation bouquet that results from chemical reactions during wine ageing. Organic acids, higher alcohols, low-volatile organic sulphur compounds and esters, with their fruity notes, are significant sensorial components of wine and other fermented beverages and are the primary compounds that form a fermentation bouquet. At low levels, most of these compounds contribute to the perceived wine aroma, but at high levels, they can dominate the aroma, decreasing its complexity. The compounds are released during alcoholic fermentation in variable concentrations depending on the yeast strain, fermentation conditions, and nutrient concentration. The influence of nitrogen on alcoholic fermentation and aroma compound formation by yeast, and therefore its effect on wine quality, is discussed herein.
Keywords: yeast, nitrogen, alcoholic fermentation, aroma compounds, wine.
Impacto do Azoto na Actividade Fermentativa das Leveduras e na Qualidade do Vinho
SUMÁRIO
O aroma do vinho é composto pelo aroma primário ou varietal que surge directamente a partir das uvas, pelo aroma secundário ou bouquet de fermentação que inclui os compostos aromáticos produzidos pelas leveduras durante a fermentação alcoólica e pelo aroma terciário ou bouquet de maturação que resulta de reacções químicas que ocorrem durante o envelhecimento do vinho. Os ácidos orgânicos, álcoois superiores, compostos voláteis de enxofre e ésteres, com suas notas frutadas, são importantes componentes sensoriais do vinho e de outras bebidas fermentadas e constituem o principal grupo de compostos que formam o bouquet de fermentação. Em baixas concentrações, a maioria destes compostos contribui para o aumento da complexidade sensorial do vinho, enquanto que em elevadas concentrações o podem depreciar. São produzidos durante a fermentação alcoólica, em concentrações variáveis, dependendo da estirpe de levedura, das condições de fermentação e da concentração de nutrientes. O papel do azoto na fermentação alcoólica e na formação de compostos aromáticos pelas leveduras e, logo, na qualidade do vinho, é aqui apresentado.
Palavras-chave: leveduras, azoto, fermentação alcoólica, compostos aromáticos, vinho.
INTRODUCTION
There is a widely recognised consumer demand for more quality wines with distinct and diverse sensorial characteristics. In wine, some aroma compounds arise from the must with minor or no modifications, while others are products of yeast metabolism, in particular from the sugar and nitrogen compounds initially present in the grapes. Indeed, wine yeasts, particularly Saccharomyces cerevisiae, play a major part in this transformation process; they are responsible for the conversion of sugars to ethanol and carbon dioxide, which make a relatively small contribution to flavour, and hundreds of by-products, some of them not yet identified, that greatly contribute to the fermentation bouquet.
The recognition of the importance of nitrogen in grape-juice fermentation has largely materialised from empirical observations. Problems such as fermentation arrests and hydrogen sulphide (H2S) production are usually linked to a low nitrogen concentration in the grape-juice. Currently, strategies for the production of wines with specific characters and styles include combining the effects of different yeast strains and nutrient modulations, particularly nitrogen.
1.AN OVERVIEW OF SUGAR METABOLISM BY SACCHAROMYCES CEREVISIAE
Grape berry components are, in roughly decreasing order of concentration: water and other inorganic substances, carbohydrates, acids, phenols, nitrogen compounds, terpenoids, fats and lipoids and flavour compounds (Boulton et al., 1996). Glucose and fructose are the major sugars in berry and grape-must, with concentrations ranging from 150 to 300 g l-1. Sucrose and other sugars, such as xylose, rhamnose and arabinose, are present in very low concentrations, and it is widely accepted that S. cerevisiae strains do not use these sugars as carbon or energy sources during alcoholic fermentation (Bisson, 1993). In S. cerevisiae, glucose and fructose are the preferred carbon source and are transported into the cell by facilitated diffusion. Twenty different genes encoding glucose transport-related proteins have been identified (Özcan and Johnston, 1999); the members of this family include HXT1-HXT17, and GAL2 (which encodes a galactose transporter) as well as SNF3 and RGT2 (which encode putative sensors of high and low glucose concentrations) (Bisson et al., 1987; Kruckeberg, 1996). The ultimate target of the Snf3/Rgt2 pathway is Rgt1, a transcription factor that regulates expression of the HXT genes, which encode glucose transporters (Özcan and Johnston, 1999; Johnston and Kim, 2005; Kim and Johnston, 2006). There are two major glucose sensing and signalling pathways in yeast: the glucose induction mechanism, which is mediated by the cell surface sensors from the glucose transporter family, Rgt2 and Snf3, (Özcan and Johnston, 1999) and the glucose repression mechanism, which is mediated by the cytoplasm glucose sensor Snf1 (Gancedo, 1998; Hardie et al. 1998; Carlson, 1999; Hedbacker and Carlson, 2008). High intracellular glucose concentrations inhibit sensing through the glucose sensor Snf3 (Karhumaa et al., 2010).
Transcription of the HXT1 to HXT7 genes, which encode the metabolically relevant hexose transporters, is regulated in response to extracellular glucose. The low affinity transporters have been shown to be preferentially expressed at high glucose concentrations, whereas the high affinity ones predominate when this sugar is scarce (Özcan and Johnston, 1999; Perez et al., 2005). The remaining 10 Hxtp proteins (Hxt8p to Hxt17p) do not appear to play an important role in glucose uptake because the genes encoding these proteins are expressed only at very low levels (Diderich et al., 1999; Özcan and Johnston, 1999). Once inside the cells, glucose and fructose are converted into two moles of pyruvate by glycolytic pathway. Glycolysis occurs in two phases. In the first phase, glucose and fructose are phosphorylated by one of three enzymes: an HXK1- or HXK2-encoded hexokinases or GLK1-encoded glucokinase, which only phosphorylates glucose (Bisson, 1993). Differences in these two steps in the fermentation pathway, transport and phosphorylation, could explain the preferential S. cerevisiae utilisation of glucose over fructose. Besides, higher affinity of hexose transporters (HXT genes) for glucose (Bisson and Fraenkel, 1983), glucose can repress the expression of the specific fructose transporter Fsy1p (Gonçalves et al., 2000; Rodrigues de Sousa et al., 2004). Nevertheless, it has been recently reported that this discrepancy in sugar utilization during wine alcoholic fermentation is dependent on yeast strain and on environmental conditions (Berthels et al., 2004). Accordingly, ethanol had a stronger inhibitory effect on fructose than on glucose utilisation, and nitrogen supplementation stimulated fructose utilisation more than glucose utilisation.
After phosphorylation glucose-6-phosphate is converted by phosphoglucoisomerase to fructose-6-phosphate, and from this point, the metabolism of glucose and fructose are the same. Briefly, the cell hydrolyses two ATP molecules for sugar activation, and glucose is broken down to produce one mole of dihydroxyacetone phosphate and one of glyceraldehyde-3-phosphate. The enzyme triosephosphate isomerase converts dihydroxyacetone phosphate into glyceraldehyde-3-phosphate. In the second phase, glyceraldehyde-3-phosphate is oxidised and converted into two molecules of pyruvate in a series of five reactions. In this phase, four molecules of ATP together with two NADH molecules are generated, resulting in a net gain of ATP for use in biosynthetic pathways by the end of glycolysis (Walker, 1998). The branch between fermentative and oxidative metabolism occurs at the level of pyruvate. This metabolite can either be transported into the mitochondrion and oxidatively decarboxylated to acetyl-CoA by the pyruvate dehydrogenase complex, or decarboxylated to acetaldehyde by pyruvate decarboxylase in the cytosol (Pronk et al., 1996). Acetaldehyde can then be reduced to ethanol in a reaction catalysed by alcohol dehydrogenase (ADH) during which NADH is oxidised back to NAD+. Regeneration of NAD+ is necessary to maintain the redox balance and prevent the recess of glycolysis (Walker, 1998). S. cerevisiae cultivated on high sugar concentrations, such as those present in grape-juice, even under fully aerobic conditions, preferentially ferments sugars to ethanol and carbon dioxide, a phenomenon known as the Crabtree effect. In addition to the reduction of acetaldehyde to ethanol, other different strategies to regenerate NAD+ are used by yeast, leading to a diversity of end products from sugar catabolism; these products, such as glycerol, succinic acid, higher alcohols, diacetyl, acetoin, 2,3-butanediol, acetic and lactic acid, acetaldehyde, α-keto acids and several other metabolites, impact the complexity of the wine.
2.YEAST NITROGEN METABOLISM
Yeasts have the ability to use a wide range of nitrogen-containing compounds as sole nitrogen sources. S. cerevisiae can assimilate nitrogen from different sources, such as ammonium, urea, allantoin, or amino acids (reviewed by Cooper, 1982). In regards to the composition of grape-juice, the total nitrogen content is highly variable, ranging from 60 to 2400 mg l-1 and is mainly composed of amino acids, ammonium, peptides and proteins (Henschke and Jiranek, 1993). The major nitrogen constituents of the average grape-juice are proline, arginine, glutamate, glutamine, serine and threonine (Boulton et al., 1996), with proline and arginine accounting for 30-65% of the total amino acid content (Pretorius, 2000).
Similar to the response to glucose, nitrogen sensing and signalling mechanisms in yeast involve two distinct sensors. The cell surface amino acid sensor Ssy1p, which is related to amino acids permeases and acts analogously to the glucose sensors Rgt2p and Snf3p (Didion et al., 1998; Iraqui et al., 1999; Klasson et al., 1999) and the cytoplasmic nitrogen sensors Tor1p and Tor2p (Bertram et al., 2000, 2002). Some sensors, such as Mep2p (Lorenz and Heitman, 1998; van Nuland et al., 2006) and Gap1p (Donaton et al., 2003), are also effective transporters.
The concentration of ammonium in grape-juice ranges from 24 to 310 mg l-1 (average 123 mg l-1), and its ratio to the total nitrogen content is reasonably constant between vine varieties and regions (Ough, 1969). In S. cerevisiae, three permeases, Mep1p, Mep2p and Mep3p, are involved in the cellular transport of ammonium, which are regulated by nitrogen catabolite repression (Marini et al., 1997), and are expressed under low ammonium concentrations (ter Schure et al., 2000). Mep2p displays the highest affinity for NH4+ (Km 1-2 μM), followed by Mep1p (Km 5-10 μM) and finally by Mep3p, whose affinity is much lower (Km 1.4-2.1 mM) (Marini et al.,1997). Ammonium is assimilated mainly into glutamate and glutamine through the pathways of central nitrogen metabolism (ter Schure et al., 2000). Though this pathway, the yeast can produce glutamate by the reactive amination of α-ketoglutarate, catalysed by glutamate dehydrogenase, and/or incorporate ammonium into the amino group of glutamate, catalysed by glutamine synthetase, to produce glutamine (Magasanik, 2003).
The amino acids concentration in grape-musts is also highly variable, ranging from 300 to 1600 mg l-1 (Henschke and Jiranek, 1993) depending upon grape variety, time of harvest and fertilisation practices (Ough, 1969; Sponholz, 1991). In S. cerevisiae, amino acid transport across the plasma membrane occurs through a number of more-or-less specific amino acid permeases via a proton symport mechanism (Horak, 1997). In addition to specific amino acid permeases, S. cerevisiae has a general amino acid permease, encoded by GAP1, which is responsible for the uptake of all naturally occurring L-amino acids and related compounds, such as ornithine and citrulline, some D-amino acids, toxic amino acid analogues, and azetidine-2-carboxylate, as well as for the polyamines putrescine and spermidine (Jauniaux and Grenson, 1990; Andreasson et al., 2004; Uemura et al., 2005). It has been suggested that the Gap1 permease is important during wine fermentation, not only because of its role as transporter but also due to its amino acid sensor function (Chiva et al., 2009).
Other amino acid permeases with low specificity are the low-affinity amino acid permease Agp1p, which accepts asparagine, glutamine and other amino acids, and the high-affinity glutamine permease Gnp1p, which accepts leucine, serine, threonine, cysteine, methionine, glutamine and asparagine (Regenberg et al. 1999). The genome of S. cerevisiae also encodes amino acid transporters with high specificity, i.e., MUP genes involved in methionine uptake (Isnard et al., 1996), Tat2p which can transport tryptophan and tyrosine and Put4p which is required for transport of proline. Can1p, Lyplp, and Alp1p are all specific for the cationic amino acids, lysine and arginine (Regenberg et al., 1999). After uptake into the cell, amino acids are assimilated and metabolised for biosynthesis and are either converted to other amino acids or degraded, releasing ammonium. Yeast is able to store amino acids in vacuoles for biosynthetic uses (Kitamoto et al.,1988). The biosynthesis of amino acids by cells grown in media with glucose and ammonium requires the utilisation of pyruvate, not only for energy production but also for α-keto acid biosynthesis, which can then be converted into the corresponding amino acid (Magasanik, 2003).
During the fermentation of grape-juice, nitrogen compounds are transported into the cells of S. cerevisiae in the first part of growth phase, although many factors, such as pH, ethanol concentration, temperature, carbon dioxide pressure, and the degree of aeration of medium, are known to affect the assimilation of these compounds (Henschke and Jiranek, 1993). The order of amino acid utilisation is influenced by the abundance of the various nitrogen sources, particularly ammonia, which is a preferred nitrogen source in this environment and readily utilised (Bisson, 1991). Yeast cells selectively uptake nitrogenous compounds from their environment through a mechanism called Nitrogen Catabolite Repression (NCR) (Cooper and Sumrada, 1983), which limits the yeast´s ability to use poor nitrogen sources in presence of good ones. The transcription of genes encoding proteins needed for the uptake and degradation of inefficient nitrogen sources (proline) is maintained at low levels when more readily usable or better nitrogen sources (ammonium, asparagine and glutamine) are available (Magasanik and Kaiser, 2002). Studies on NCR in S. cerevisiae during wine fermentation confirmed that yeast cells evolve from a nitrogen repressed-situation at the beginning of fermentation to a nitrogen de-repressed condition as nitrogen is consumed (Beltran et al., 2004). However, arginine and γ-aminobutyric acid are usually taken up during the latter stages of fermentation under enological conditions and are always detectable in the final wine (Boulton et al., 1996). Furthermore, proline, one of the major nitrogen sources found in grape-juice, requires molecular oxygen for its metabolism (Tomenchock and Brandiss, 1987) and is not taken up from grape juice under anaerobic fermentative conditions (Ingledew et al., 1987). Because of these factors, yeast assimilable nitrogen (YAN) is defined as the sum of total amino acids minus proline plus ammonia, and it is expressed as mg N l-1.
3.THE INFLUENCE OF NITROGEN ON ALCOHOLIC FERMENTATION AND FORMATION OF BY-PRODUCTS
Several factors have been recognised as a potential cause of problematic fermentations. Among these factors, the limited nitrogen content of some natural grape-juices has been associated with the occurrence of stuck and sluggish fermentations. In addition to these problems, nitrogen concentration also regulates the formation of by-products, such as H2S, fatty acids, higher alcohols, and esters, among others, which affect the chemical and sensorial proprieties of wine.
3.1. Stuck and sluggish fermentations
During winemaking, the external environment strongly influences the physiological behaviour of yeast cells. In the case of stuck or sluggish fermentation, the environmental changes during grape-juice fermentation may lead to an inability of the yeast cells to use all the existing sugars. In wineries, sluggish fermentation is defined as one in which the rate of sugar consumption is very low, and the end of fermentation is not accomplished in a reasonable amount of time. In these cases, even if dryness is accomplished, much attention must be paid to potential economics losses (Kunkee, 1991; Boulton et al., 1996). Incomplete or stuck fermentations are defined as those leaving a higher than desired residual sugar content in the wine at the end of the alcoholic fermentation (greater than 2 g l-1 for dry wines). Wines with high post-fermentation sugar content are very susceptible to microbial spoilage and cannot be bottled until it is known that they are microbiological stable. Furthermore, slow and incomplete fermentations are vulnerable to oxidation problems due to the lack of protective carbon dioxide release of during active fermentation (Bisson, 1999). Although a significant amount of research has been conducted in wine-producing countries to establish the causes of these problematic fermentations (reviewed in Bisson, 1999; Alexandre and Charpentier, 1998; Henschke, 1997), they are still common problems in modern winemaking.
Generally, grape-juices possess all the nutrients necessary to support yeast growth, of which the amounts of carbon and nitrogen the most important. While the carbon sources present in grape-juices greatly exceed the nutritional requirements of the yeast, the nitrogen compounds are highly variable in composition and concentration, as mentioned above. Initial low levels of assimilable nitrogen can limit growth and biomass accumulation, resulting in a slow fermentation rate (Salmon, 1989; Manginot et al., 1998; Bely et al., 1990; Mendes-Ferreira et al., 2004). Yeast growth, like the fermentation rate, is an exponential function of the initial nitrogen content of must (Agenbach, 1977), is highly sensitive to low nitrogen concentrations (less than 300 mg l-1) and is less responsive to higher concentrations (Jiranek et al., 1990, 1991).
Although, the fermentation rate may initially be related to yeast biomass, usually it becomes uncoupled from growth during the latter stages of fermentation. However, there is a direct relationship between the initial sugar content, fermentation rate and duration, and the amount of nitrogen utilised by yeast. The threshold concentration of nitrogen needed to achieve total sugar degradation is not only dependent on the initial sugar concentration and fermentation conditions but also most significantly on the yeast strain used (Jiranek et al., 1990, 1995a). This dependence highlights the choice of yeast strain as a useful tool for managing the fermentation of low nitrogen grape-musts.
A mean value of 140 mg l-1 (Agenbach, 1977) of assimilable nitrogen has been mentioned as being sufficient for complete fermentation of reasonably ripened grapes (25.8º Brix), and a value six times higher is needed to achieve the optimum level (Kunkee, 1991). More recently, using chemically defined grape-juice media, Mendes Ferreira et al. (2004) established that S. cerevisiae requires a minimum of 267 mg N l-1 for complete fermentation of 200 g l-1 of glucose in an industrially reasonable time and that lower nitrogen levels were sufficient to support growth but led to sluggish or stuck fermentations. Although nitrogen has been the most studied cause of sluggish and/or stuck fermentations, the basic mechanism responsible for sugar breakdown arrest is unknown. Upon depletion of nitrogen from the external media, a major decrease in sugar transport activity has been observed. The reduced fermentative activity has been explained by a strong decrease in the transport of sugar activity triggered by nitrogen starvation (Salmon, 1989; Salmon et al., 1993), although it has not been well established how nitrogen levels physiologically modulate carrier activity. Published results suggest that during alcoholic batch fermentation with high sugar concentration and nitrogen limitation, protein synthesis stops and the glucose transport begin to be irreversibly inactivated, when yeast cells sense intracellular nitrogen limitation, either due to a lack of nitrogen in the medium or by inactivation of ammonium transport systems (Lagunas et al., 1982; Busturia and Lagunas, 1986; Cardoso and Leão, 1992). This disabling of glucose transport ultimately leads to an alleviation of the Crabtree effect, with the activation of respiratory genes and decrease in fermentation rate (Mendes-Ferreira et al., 2007a). Accordingly, it has been suggested that the transcription activation of genes involved in the tricarboxylic acid cycle and respiration may be associated with a low sugar uptake capacity and/or redox imbalance (Jin et al., 2004). The obvious advantage of this association is that less sugar is needed by yeast cells to obtain the same amount of ATP (Thomsson et al., 2005) needed for cellular maintenance. However, in studies conducted under conditions mimicking the enological environment, it was observed that S. cerevisiae continues to ferment glucose for nearly a month in a nitrogen depleted media, indicating that yeast cells are able to sustain basal glucose up-take capacity during long periods of nitrogen deprivation (Mendes-Ferreira et al., 2007a, 2009). Both low- (HXT1 and HXT3) and high-affinity (HXT2, HXT6, and HXT7) glucose carriers were expressed at higher levels in the N-limiting than in the control fermentations; this expression profile indicates that the regulation of the glucose transporters is associated with translational or post-translational levels rather than occurring at the transcriptional level (Mendes-Ferreira et al., 2007a). Using a transposon mutagenesis approach, Gardner et al. (2005) have identified two genes, NGR1 and GID7, the disruption of which improved sugar catabolism throughout nitrogen-limited fermentations. The authors suggested that the absence of these genes could prevent deactivation of Hxt7p and, thus, enhance its ability to maintain catabolic activity under conditions of nitrogen starvation. Additionally, the results of high cell-density experiments showed that in spite of a lower sugar uptake, adding biomass obtained from sluggish cultures reduced the time to finish a problematic fermentation (Varela et al., 2004). However, even if a large population of active yeast cells can be introduced into the inoculated wines, resulting in a complete fermentation, this does not necessarily correlate to an improvement in the sensory characteristics of the wines (Carrau et al., 2010)
To overcome sluggish or premature fermentation arrest due to nitrogen limitation, winemakers supplement grape-juices prior to fermentation with diammonium phosphate. This strategy does not always succeed because extracellular nitrogen is not the only factor affecting intracellular pools of nitrogen in yeast. The efficiency of the nitrogen transport system may be reduced by the increasing ethanol formation (Cardoso and Leão, 1992), either by increasing membrane permeability to protons (Leão and van Uden, 1984) inhibiting the proton pumping membrane ATPases (van Uden, 1989) or by changes in the lipid composition of the yeast plasma membrane (Alexandre et al., 1994). In addition, attention must be given to the legislation pending on limiting nitrogen addition due to the possible formation of ethyl carbamate, a suspected carcinogen, and because excess nitrogen can cause microbial instability in wines.
The use of powerful techniques, such as DNA microarrays, has contributed to a better understanding of the molecular basis for the adaptation of wine yeast to the plethora of stress conditions that successively occur throughout alcoholic fermentation, leading to improvements in the winemaking process (Backhus et al., 2001; Rossignol, et al., 2003; Mendes-Ferreira et al., 2007a). Comparative gene expression profiling of a reference condition with N-sufficient concentration to a series of N-limitation/starvation concentration conditions in the yeast strain S. cerevisiae (Mendes-Ferreira et al., 2007a) led to the identification of a restricted group of genes that specifically respond to nitrogen availability (Mendes-Ferreira et al., 2007b). A designed DNA chip incorporating some of these signature N-responsive genes identified is being developed to assist the winemaker in assessing the nitrogen status of the fermenting grape-juice. Our ultimate goal is that such a chip could be used to predict premature fermentation arrest due to nitrogen shortage and to allow for the customisation of treatment strategies.
Meanwhile, an estimation of the initial nitrogen concentration in grape-juice continues to be of major interest for detecting the risk of problem fermentations. A great effort has been made to develop a rapid and accurate method of determining nitrogen content in grape-juice in order to limit the risk of fermentation becoming sluggish or stuck and the consequent depreciation of wine quality (Dukes and Butzke, 1998; Shively and Henick-Kling, 2001; Gump et al., 2002; Filipe-Ribeiro and Mendes-Faia, 2007). Furthermore, the deliberate use of strains with lower requirements for assimilable nitrogen might in itself be a useful strategy for reducing the incidence of problematic fermentations (Gardner et al., 2002, 2005) particularly for wineries where routine grape-juice nitrogen quantification is not readily achievable. In this way, better nitrogen utilisation ability will lead to a higher cell density with the same nitrogen contents, promoting a high and sustained fermentation rate (Bisson, 1999).
3.2. Production of hydrogen sulphide
Volatile sulphur compounds, such as H2S and various ethyl and methyl mercaptans, are generated by wine yeasts during alcoholic fermentation, and most are associated with unpleasant odours (Swiegers and Pretorius, 2007). H2S imparts a rotten eggs odour and has a profoundly negative effect on the sensory quality of wines when it exceeds its perception threshold (50 to 80 mg l-1). At lower levels, 20 to 30 mg -1, the compound has been considered to have a positive effect, providing a yeast-like smell (Rauhut, 1993).
H2S is a product of the sulphate reduction sequence (SRS) pathway (Fig. 1), which involves a set of sequential reactions that culminates with the incorporation of sulphur into a carbon skeleton (Thomas and Surdin-Kerjan, 1997).
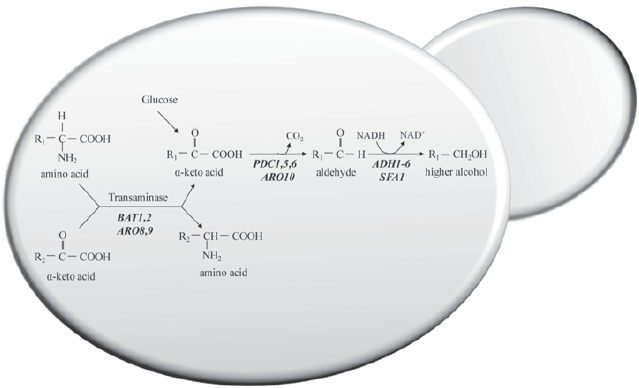
Fig. 1– Illustrative representation of the SRS pathway and the biosynthesis of sulphur amino acids in S. cerevisiae. The genes involved in each enzymatic step are in italics.
Sulphate up-take involves two high-affinity permeases. Once inside the cell, the transfer of the adenosyl-phosphoryl moiety of ATP to sulphate yields adenylyl sulphate (APS), which is in turn phosphorylated to yield phosphoadenylyl sulphate (PAPS). PAPS is reduced to sulphite and then to sulphide. In the presence of nitrogen, sulphide is incorporated in nitrogenous precursors to form cysteine and methionine. When insufficient nitrogen is available, sulphide diffuses out of the cell.
Representação da via de assimilação e redução do sulfato (SRS) e da biossíntese de aminoácidos sulfurados em S. cerevisiae. Os genes envolvidos em cada etapa enzimática estão apresentados em itálico.
A assimilação do sulfato envolve duas permeases de alta afinidade. Uma vez dentro da célula, a transferência do grupo adenosil-fosforil do ATP para o sulfato origina adenilil-sulfato (APS), que por sua vez é fosforilado originando fosfoadenilil-sulfato (PAPS). O PAPS é reduzido a sulfito e depois a sulfureto. Na presença de azoto, o sulfureto é incorporado nos precursores azotados para formar cisteína e metionina. Quando o azoto disponível é insuficiente, o sulfureto difunde-se para fora da célula.
The levels of H2S produced by yeasts depends on the levels of elemental sulphur in grapes (Mendes-Ferreira et al., 2002; Schutz and Kunkee, 1977; Thomas et al., 1993), naturally present as sulphate at an average concentration of 200 mg l-1 (Rauhut, 1993), and on the amount of sulphite (Acree et al., 1971; Stratford and Rose, 1985) commonly added (usually 50-200 mg l-1) in grape-must prior to fermentation due to its antioxidant and antimicrobial proprieties. The amount of organic compounds containing sulphur (Acree et al., 1972), vitamin deficiencies (Wang et al., 2003; Bohlscheid and Edwards, 2004; Bohlscheid et al., 2007) and fermentation conditions (Schutz and Kunkee, 1977) also influence H2S production under winemaking conditions. The amount of sulphide released is highly dependent on the genetic background of the yeast (Henschke and Jiranek, 1993; Jiranek et al., 1995b; Spiropoulos et al., 2000; Mendes-Ferreira et al., 2002, 2009, 2010), probably due to differences in the activity levels of the enzymes involved in incorporating sulphide into amino acids (Spiropoulos et al., 2000) or the differences in the yeasts ability to retain a greater amount of H2S within the cells (Rupela and Tauro, 1985). The complexity of the production and release makes it difficult to devise strategies to prevent hydrogen sulphide formation during winemaking.
Racking and aeration and the addition of trace quantities of copper sulphate are two effective means of eliminating the most volatile sulphides (Boulton et al., 1996), but the remainder left behind by these treatments represents a troublesome source of off-odours in wine.
The association between a shortage of assimilable nitrogen and H2S production during wine fermentations was first established by Vos and Gray (1979). Later, it was suggested that H2S production is dependent on yeast requirements for amino acids and is a response to nitrogen deficiency when cells are grown in the presence of sulphite (Henschke and Jiranek, 1991). In the presence of sufficient nitrogen, sulphide is incorporated in the nitrogenous precursors O-acetyl serine and O-acetyl homoserine, which form cysteine and methionine, respectively. The two sulphur-containing amino acids, cysteine and methionine, are usually found in very low concentrations in grape-juice, 0-2 mg l-1 and 0-119 mg l-1, respectively (Sponholz et al., 1991). Therefore, biosynthesis of these amino acids by yeast during growth is necessary (Henschke and Jiranek, 1993). Under nitrogen sufficient conditions, an adequate supply of precursors is available to prevent the net accumulation of sulphide, but when insufficient or unsuitable nitrogen sources are available, the pool of precursors becomes depleted and the SRS pathway is activated in an attempt to restore sulphur amino acids biosynthesis. This process leaves no available precursors to combine with the sulphide, and, thus, it diffuses out as the sensorially offensive compound H2S (Henschke and Jiranek, 1991; Jiranek et al., 1995b). Recently, an inverse relationship between the H2S produced by yeasts and the initial nitrogen concentration was found in concentrations ranging from 267 to 402 mg N l-1 (Mendes-Ferreira et al., 2009).
At lower levels, e.g., 66 mg N l-1, very little sulphide is liberated, in agreement with previous transcriptomic data in which several MET genes, essential for the SRS pathway, were specifically down-regulated under nitrogen-deficient conditions (Mendes-Ferreira et al., 2007a ,b). In contrast, a clear relationship between the yeast strain and the liberation of H2S in response to nitrogen availability (267 or 402 mg N l-1) with gene expression levels could not be found, making it difficult to propose a mechanism for any cross-talk between gene expression and H2S liberation (Mendes-Ferreira et al., 2010). However, using a different approach, Linderholm et al. (2008) identified four genes involved in the SRS pathway (MET17, CYS4, HOM2 and HOM6) that impact H2S formation.
In contrast to what has been extensively reported, recent data provided evidence that DAP supplementation not only does not reduce H2S formation but also, in some cases, even exacerbates the problem; the timing of such addition is critical to ensure that fermentation proceeds without an excessive release of H2S (Mendes-Ferreira et al., 2010a). This observation reinforces the importance of an accurate estimate of the initial YAN concentration and leads to questions about the wisdom of prophylactic DAP supplementation prior to fermentation.
In sum, further strategies should be used to study wine yeast strains under conditions that exacerbate the strain differences in H2S production, in order to clarify the regulatory effects of this particular group of genes on H2S release under winemaking conditions.
3.3. Production of fatty acids
Fatty acids constitute one of the classes of compounds produced by yeast during fermentation that impart unpleasant aromas, and they have been associated with fatty, rancid and cheese-like odours (Peinado et al., 2004; Ugliano and Henschke, 2009; Bisson and Karpel, 2010). Short- and medium-chain fatty acids (MCFA) are mostly consumed during alcoholic fermentation (Henschke and Jiranek, 199; Ancín et al., 1998).
The pathway leading to the biosynthesis of fatty acids involves the formation of Acetyl-CoA (Nikänen, 1986), which is produced by oxidative decarboxylation of pyruvic acid. This series of reactions leading to the synthesis takes place in a multienzyme complex known as fatty acid synthase (Walker, 1998). The production of fatty acids by yeasts is related to the metabolism of carbohydrates through glucose, which is the main precursor of acetyl-CoA. MCFA are produced by yeast as intermediates in the biosynthesis of long-chain fatty acids, rather than as a result of acid catabolism (Lambrechts and Pretorius, 2000). The unsaturated fatty acids, palmitoleic and oleic, play important roles in yeast physiology, such as membrane integrity, and increase the resistance of yeast to elevated concentrations of ethanol (Walker, 1998). At certain levels, MCFA such as decanoic and octanoic acids may become inhibitory to S. cerevisiae growth (Sá-Correia et al., 1989; Bisson, 1999). This inhibitory effect is more prevalent in white wine fermentation, probably because these types of wines are fermented at low temperatures and without aeration (Zamora, 2009). In addition to the yeast strain, the medium composition (Alexandre and Charpentier, 1998) and fermentation temperature (Beltran et al., 2006) have been reported to influence the formation of fatty acids. Oxygen is another important factor for yeast metabolism because it is required for the biosynthesis of essential unsaturated fatty acids and sterols (Walker, 1998).
The production of MCFA, hexanoic, octanoic and decanoic, and their respective ethyl estersethyl hexanoate, ethyl octanoate, and ethyl decanoate, is stimulated by high assimilable nitrogen levels (Mendes-Ferreira et al., 2009). Also, the timing of nitrogen addition affects MCFA released by yeasts (Barbosa et al., 2009); a later addition of DAP, during stationary growth-phase, leads to a significant decrease in the production of these compounds.
Acetic acid is the principle component of volatile acidity in fermented beverages (Boulton et al., 1996; Ribéreau-Gayon et al., 2000). At high concentrations, acetic acid gives a negative sensory attribute (vinegar-character) to wine, with an acceptable concentration from 0.2 to 0.7 g l-1. It is produced not only by spoilage bacteria (lactic acid bacteria and acetic bacteria) but is also formed by yeast during fermentation in the range of 0.1 to 0.3 g l-1. The biochemical pathway for the formation of acetic acid by wine yeasts has not yet been clearly identified (Boulton et al., 1996, Ribéreau-Gayon et al., 2000). The possible enzyme reactions in yeast that could lead to acetic acid formation are as follows: 1) reversible formation from acetyl Co-A and acetyl adenylate through acetyl Co-A synthetase; 2) cleavage of citrate by citrate lyase; 3) production from pyruvate by pyruvate dehydrogenase; 4) reversible formation from acetyl-phosphate by acetyl kinase and 5) oxidation of acetaldehyde by aldehyde dehydrogenase (Boulton et al., 1996).
The amount of acetic acid produced by yeasts during alcoholic fermentation was found to depend on the yeast strain (Shimazu and Watanabe, 1981; Millan and Ortega, 1988; Erasmus et al., 2004; Torrens et al., 2008), fermentation temperature (Monk and Cowley, 1984; Beltran et al., 2008) and the chemical composition of grape-juice, such as the sugar level and vitamins concentration (Nordström, 1964; Radler, 1993). In wine, acetic acid production seems to be inversely correlated with the initial nitrogen levels (Agenbach, 1977; Tromp, 1984; Bely et al., 2003). The lowest acetic acid concentrations occur around 200–250 mg/L of yeast assimilable nitrogen with increases of up to twofold at nitrogen concentrations outside this range (Bely et al. 2003; Vilanova et al. 2007). These levels are apparently unaffected by the initial sugar concentration and osmotic stress (Ugliano and Henschke, 2009). However, in slow fermentation under nitrogen limitation/starvation conditions with 67 mg N l-1, yeasts produce less acetic acid (Mendes-Ferreira et al., 2009). Nitrogen additions prior to fermentation were more effective than later additions at preventing acetic acid accumulation in wine (Ugliano and Henschke, 2009). Conflicting results have been obtained in synthetic grape-juice media in which Barbosa et al., (2009) found that nitrogen additions later, during stationary phase (72 h), decreased acetic acid concentration; these conflicting results caution that further work should be carried out in natural grape-juices.
3.4. Production of alcohols
Higher alcohols, also known as fusel oils, are aliphatic and aromatic alcohols containing more than two carbon atoms. They are quantitatively the largest group of the flavour compounds found in alcoholic beverages (Nikänen and Nikänen, 1977; Nikänen, 1986). These compounds can have either a positive or negative impact on the aroma and flavour of wine. Total higher-alcohols content in wine ranges from 100 to 500 mg l-1, and isoamyl alcohol is the major component (Boulton et al., 1996). In concentrations exceeding 400 mg l−1 higher- alcohols can impart a strong, pungent smell and taste, whereas optimal levels below 300 mg l−1 confer fruity characters (Lambrechts and Pretorius, 2000; Swiegers and Pretorius, 2005).
The amount of higher alcohols formed during fermentation depends on the yeast strain (Carrau et al., 2008; Jimenéz-Martí et al., 2007; Mendes-Ferreira et al., 2009; Miller et al., 2007; Molina et al., 2007; Nikänen and Nikänen, 1997; Romano et al., 2003; Thurston et al., 1982; Rankine, 1967, Singh and Kunkee, 1977), fermentation temperature (Molina et al., 2007; Beltran et al., 2006; Webb and Ingraham, 1963; Rankine, 1967), pH of the grape-juice (Rankine, 1967), amount of insoluble solids (Klingshirn et al., 1987) and aeration conditions (Webb and Ingraham, 1963; Guymon et al., 1961) as well as on the initial nitrogen content of the media (Ough and Bell, 1980; Rapp and Versini, 1991; Vilanova et al., 2007; Hernandez-Orte et al., 2006; Mendes-Ferreira et al., 2009).
Aliphatic alcohols include 1-propanol, 2-methyl 1-propanol, 2-methyl 1-butanol (active amyl alcohol) and 3-methyl 1-butanol (isoamyl alcohol). Isoamyl alcohol, active amyl alcohol and isobutyl alcohol are produced by wine yeasts from the branched-chain amino acids, leucine, isoleucine and valine, respectively. Basically, two different metabolic pathways (Fig. 2) are hypothesised to be involved in the production of the higher alcohols: 1) anabolic pathways involved in de novo synthesis of branched-chain amino acids through their biosynthetic pathway from glucose, and 2) the catabolism of valine, leucine, isoleucine, and 2-phenylalanine, also known as the Ehrlich pathway.
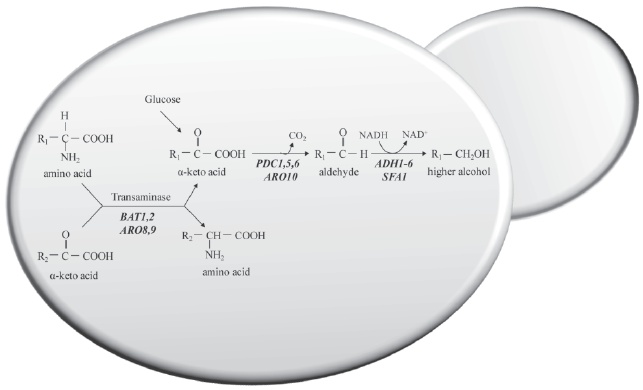
Figure 2 – Formation of higher alcohols from sugar and by the Ehrlich pathway.
The α-keto acids, derived from sugars via pyruvate or from transamination/deamination of amino acids, are decarboxylated to the correspondent aldehyde. This aldehyde is then reduced to the correspondent alcohol by alcohol dehydrogenase. Genes of S. cerevisiae involved in each enzymatic step are in italics.
Formação de álcoois superiores a partir dos açúcares e pelo mecanismo de Ehrlich.
Os α-cetoácidos, derivados dos açúcares via piruvato ou formados por transaminação/desaminação dos aminoácidos, são descarboxilados ao aldeído correspondente. O aldeído é depois reduzido ao respectivo álcool pela álcool desidrogenase. Os genes de S. cerevisiae envolvidos em cada etapa enzimática estão apresentados em itálico.
The pathway of formation of higher alcohols via the Ehrlich mechanism is identical to that involved in the biosynthesis of the analogous amino acids valine, leucine and isoleucine, up to the last intermediate, i.e., the appropriate α-keto acid (Boulton et al., 1996). The α-keto acids derived from the sugars via pyruvate (Nikänen, 1986) or from transamination/deamination of amino acids are decarboxylated to the corresponding aldehyde and then reduced by the alcohol dehydrogenase to the corresponding alcohol (Webb and Ingraham, 1963; Kunkee et al., 1966, 1972; Singh and Kunkee, 1977; Hazelwood et al., 2008). The aldehydes formed can then be either reduced to form fusel alcohol or oxidised to form a fusel acid. The ratio of fusel acids to fusel alcohols is largely dictated by the redox status of the yeast cell and whether reduced or oxidised cofactors are in short supply (Bisson and Karpel, 2010).
Under low nitrogen concentration, the majority of α-keto acids are mainly synthesised from sugars due to a lack of alpha amino acids necessary for transamination purposes (Nikänen and Nikänen, 1977; Nikänen, 1986; Oshita et al., 1995). At low levels of assimilable nitrogen, the biosynthetic pathway predominates, whereas at high levels the Ehrlich pathway becomes prominent as a result of feedback and/or repression of key enzymes in the biosynthetic pathway (Yoshimoto et al., 2002). Changes in isoamyl alcohol production, influenced by the nitrogen source, appear to be due to transcriptional co-regulation of LEU and BAT genes and production of isoamyl acetate is dependent on isoamyl alcohol production and ATF transcription (Yoshimoto et al., 2002). Overexpression of a branched-chain amino acid transferase gene, BAT1, enhanced the production of isoamyl acetate (Lilly et al., 2006), which appeared to be linked to an increased production of isoamyl alcohol, the precursor of isoamyl acetate.
It is commonly accepted that the most important pathway leading to 2-phenylethanol, which is described with pleasant terms such as old rose, sweetish, and perfumed alcohol, is the Ehrlich pathway. L-phenylalanine is transaminated to phenylpyruvate, decarboxylated to phenylacetaldehyde and finally reduced to the alcohol. However, Etschmann et al. (2002) described an alternative route, where 2-phenylethanol is obtained by de novo synthesis via the Shikimate pathway, in which erythrose-4-phosphate from glycolysis and phosphoenolpyruvate from the pentose-phosphate pathway are condensed with chorismate and prephenate to produce phenylpyruvate. This alcohol is produced in high quantities by wine yeast in low nitrogen conditions, (Mendes-Ferreira et al., 2009), and, as expected, when DAP is added, its concentration is diminished (Barbosa et al., 2009). However, to enhance 2-phenylethanol formation it is recommended to supplement the musts during the yeast stationary growth phase (Barbosa et al., 2009, Mendes-Ferreira et al., 2010a).
3.5. Production of esters
Ethyl esters of fatty acids (ethyl hexanoate, ethyl octanoate, ethyl decanoate) and acetates of higher alcohols (phenylethyl acetate, isoamyl acetate) are qualitatively one of the most important flavours in wine, because they contribute to a desirable fruity taste (Bartowsky and Pretorius, 2009; Fujii et al., 1994, 1997; Mason and Dufour, 2000; Nikänen, 1986; Saerens et al., 2010; Ugliano and Henschke, 2009; Fugelsang and Edwards, 2007; Bisson and Karpel, 2010). Exceptions are ethyl lactate and diethyl succinate, which do not have any sensory impact at normal concentrations (Zamora, 2009), and ethyl acetate gives an unpleasant solvent or nail-polish character.
A fruity character is usually associated with ethyl esters, while the tropical fruit notes are linked to the acetates of higher alcohols (Clemente-Jimenez et al., 2005). The most significant esters that contribute to the pleasant fruity aroma of wines are as follows: isoamyl acetate (banana-like aroma); ethyl caprylate and ethyl caproate (apple-like aroma); 2-phenylethyl acetate (fruity flower flavour); ethyl butanoate (floral or fruity); ethyl caproate or ethyl octanoate and ethyl caprylate, also called ethyl hexanoate (sour apple); and phenylethyl acetate (flowery, roses, or honey) (Lambrechts and Pretorius, 2000; Verstrepen et al., 2003a). Their contribution to wine aroma and bouquet largely depends on their relative concentrations: at low levels, esters contribute to perceived wine aroma complexity, while at high levels, they may decrease aroma complexity.
Esters found in wine are originated directly in the grapes (Rapp and Versini, 1991) but the majority result from the activity of yeasts during the alcoholic fermentation (Younis and Stewart, 1998; Lambrechts and Pretorius, 2000; Saerens et al., 2008). Esters are synthesised by yeasts via lipid and acetyl-CoA metabolism during fatty acid biosynthesis or are produced by chemical esterification of alcohols with acids during wine-ageing (Lambrechts and Pretorius, 2000). The majority of acetyl-CoA is formed by the oxidative decarboxylation of pyruvate, while most of the other acyl-CoAs come from the acylation of free CoA catalysed by acyl-CoA synthase in fatty acid metabolism (Mason and Dufour, 2000). Factors that stimulate fatty acid biosynthesis also stimulate fatty acid ester formation (Bardi et al., 1998; Lilly et al., 2006; Saerens et al., 2008, 2010), and mutations that block fatty acid biosynthesis also block ester formation, suggesting that esters derive from fatty acid biosynthesis and not from its degradation (Thurston et al., 1982).
In short, esters are formed via an intracellular process catalysed by acyltransferase or ester synthase (Mason and Dufour, 2000). Ethyl esters derive from the reaction of acyl-CoA compounds with ethanol catalysed by acyl-CoA :ethanol O-acyltransferases, whereas acetate esters formation results from the reactions of acetyl-CoA with higher alcohols, catalysed by alcohol acetyltransferases (Boulton et al., 1996; Bisson and Karpel, 2010).
In ethyl ester formation, the condensation of acyl-CoA with ethanol is catalysed by acyltransferases, encoded by the EEB1 and EHT1 genes and YMR210W (Saerens et al., 2006, 2008, 2010). The formation of acetate esters results from the condensation of acetyl-CoA with higher alcohols by acetyltransferases, encoded by the ATF1 and ATF2 genes (Fujii et al., 1994, 1997; Fujiwara et al., 1998, 1999; Lilly et al., 2000, 2006; Verstrepen et al. 2003b; Yoshimoto et al., 1998, 1999; Nagasawa et al., 1998). In fact, over-expression of the ATF1 gene significantly increased the production of fruity esters by yeasts (Lilly et al., 2000, 2006; Verstrepen et al., 2004). Mutational analysis of these genes suggests that other unknown transferases may also be involved in ester synthesis (Bisson and Karpel, 2010; Ugliano and Henschke, 2009). However, deletion of the IAH1 gene, which encodes isoamyl acetate hydrolysing esterase, leads to increased acetate esters production (Fukuda et al., 1998). This esterase is required to balance alcohol acetyltransferase in order to maintain optimal amounts of isoamyl acetate and is particularly important in saké brewing (Fukuda et al., 2000). In sum, the balance between ester-synthesising enzymes (alcohol acetyltransferases) and ester-degrading enzyme (esterases) is important for the net rate of ester accumulation and overall flavour profile of the wine (Fukuda et al., 1998; Verstrepen et al., 2003b; Lilly et al., 2006; Ugliano and Henschke, 2009).
Esters production is susceptible to quantitative variations; however, acetates and ethyl esters do not always vary in the same way, reflecting their different metabolic pathways (Ugliano and Henschke, 2009). The amount of esters formed by yeast during fermentation primarily depends on the yeast strain (Carrau et al., 2008; Jimenez-Marti et al., 2007; Mendes-Ferreira et al., 2009; Miller et al., 2007; Molina et al., 2007; Romano et al., 2003; Saerens et al., 2008, Soles et al., 1982; Thurston et al. 1982), fermentation temperature (Beltrán et al., 2006; Killian and Ough, 1979; Molina et al., 2007), oxygen and nitrogen availability (Vos and Gray, 1979; Bell et al., 1979; Ough and Lee, 1981), lipid and higher alcohol contents (Nikänen and Nikänen, 1977), and inoculum size (Carrau et al., 2010). A lower fermentation temperature tends to increase the production of esters (Beltrán et al., 2006, Molina et al., 2007), not due to retention of volatile esters or inhibition of esterase activity (Bisson and Karpel, 2010), but due to changes in the expression profiles of several genes involved in esters synthesis under low temperature conditions (Beltrán et al., 2006).
The concentrations of oxygen, nitrogen, lipids and higher alcohols affect the types of esters formed during fermentation, via regulation of the Ehrlich, fatty acid and ester synthesis pathways (Swiegers and Pretorius, 2005). Usually, more esters are produced by high nitrogen demanding yeasts (Torrea et al., 2003). Addition of ammonium to grape-musts, a common practice in the wine industry, increases the concentration of acetate esters and ethyl butyrate but does not increase the higher molecular weight fatty acid ethyl esters, ethyl hexanoate, ethyl octanoate and ethyl decanoate (Béltran et al., 2005; Barbosa et al., 2009; Miller et al., 2007).
3.6. Production of volatile thiols
Organic sulphur compounds have a considerable influence on the sensorial quality of wines due to their associated unpleasant odours and low detection thresholds. However, some contribute positively to wine aroma. For example, the tropical characters found in Sauvignon Blanc wines come primarily from volatile thiols, with contributions from fermentation-derived esters (Dubourdieu et al., 2006; Swiegers et al., 2009). These volatile thiols do not exist in grape-juice and are released during alcoholic fermentation by yeast utilisation of the S-cysteine conjugates present in the grapes (Dubourdieu et al., 2006, Swiegers et al., 2009). Most of wine yeast strains produce a relatively similar fermentation bouquet, but some strains possess a strong ability to hydrolyse the cysteine conjugates responsible for the Sauvignon Blanc character (Dubourdieu et al., 2000; Murat et al., 2001). A moderate nitrogen supply to the vines increases the amount of aromatic precursors in the grapes (Peyrot des Gachons et al., 2005). The amount of volatile thiols released from chain-cysteinylated precursors is repressed by the Ure2p regulator via the general NCR mechanism (Thibon et al., 2008), which regulates the uptake of nitrogen sources. Additionally, the Ure2p protein repressed the transcription of IRC7, which encodes putative cystathionine beta-lyase, the main enzyme involved in thiol bioconversion (Thibon et al., 2008). An inverse correlation between nitrogen content and production of volatile thiols by yeast has been observed in natural and synthetic grape juice (Subileau et al., 2008). In this study, the involvement of Gap1p in the up-take of the cysteinylated precursor was only demonstrated in synthetic grape juice, and thus, further work should be carried out to identify the key step(s) on which NCR eventually acts.
3.7. Production of monoterpenes
Monoterpenes are the most important odour compounds. Many, such as linalool, nerol, geraniol, and citronellol, have pleasant floral odours and are the key aroma compounds of Muscat grapes (Rapp and Versini, 1991). Terpenes, metabolites derived from mevalonic acid, are characterised by multiples of branched five-carbon unit that resemble isoprene. The monoterpenes are 10-carbon compounds, many of which are volatile and odorous (Boulton et al., 1996). In grapes, many of these compounds are stored in a water-soluble, glycosidically bound form. Formation of terpenes during fermentation was first detected by Wurz et al. (1988) and Zea et al. (1995). The role of yeasts on terpene formation in wines has been considered irrelevant or has even been ignored because the increase of terpenic compounds found in the must during fermentation was attributed solely to β-glucosidase activity, which is more active in non-Saccharomyces yeasts (Rosi et al., 1994, Mendes-Ferreira et al., 2001). β-glucosidase cleaves the sugar moieties from the glycosides and can have a specific impact on the sensory profile of wine as it releases the more volatile aroma compounds.
Recently, a few studies, using a synthetic grape-juice medium lacking terpene precursors showed that yeasts can produce monoterpenes during fermentation (Carrau et al., 2005; Mendes-Ferreira et al., 2009). Earlier, Javelot et al. (1991) verified that a mutation in a gene involved in sterol biosynthesis gene ERG20, which codes for farnesyl pyrophosphate synthetase which (the enzyme responsible for catalysing the formation of C15 farnesyl pyrophosphate units for isoprenoid and sterol biosynthesis) increases the ability of the yeast to produce terpenes, giving a strong Muscat note to the wines fermented with that strain. Also, increasing yeast geraniol synthase activity increases the production of geraniol, in contrast to what occurs in defective farnesyl diphosphate synthase mutants that excrete geraniol and linalool in similar amounts (Oswald et al., 2007). Assimilable nitrogen and oxygen are presumed to be regulators of mevalonic and sterol formation; therefore, intermediates such as geranyl pyrophosphate could be a monoterpene precursor (Vaudano et al., 2004).
In contrast, Carrau et al., (2005) proposed that biosynthesis of the C10 terpenes derives from the leucine-mevalonic acid pathway and, thus, is independent of sterol metabolism. Strikingly, the amount of assimilable nitrogen and oxygen content in the growth media were noted to affect monoterpene formation by yeast (Carrau et al., 2005; Bell and Henschke, 2005). Also, Mendes-Ferreira et al., (2009) observed that the amounts of linalool produced were yeast strain dependent, but the authors were not able to establish a link between nitrogen levels and terpene production by wine yeasts.
4. FINAL REMARKS
The effect of initial nitrogen concentration on the growth and fermentation ability of wine yeast has been extensively studied in the last few decades. This information has provided valuable data for the control and prevention of slow and premature fermentation arrest during winemaking. Recently, a few studies have addressed the impact of nitrogen concentration, nitrogen source and the timing of nitrogen addition on aroma formation by yeast. On the basis of these studies, alternative strategies using conventional techniques could be devised to optimise the release of aroma compounds by yeasts during alcoholic fermentation. Despite this progress, further investigation on the mechanisms underlying aroma compounds formation by yeasts is essential.
REFERENCES
Acree T.E., Sonoff E.P., Splittstoesser D.F., 1971. Determination of hydrogensulfide in fermentation broths containing SO2. Appl. Microbiol., 22, 110-12. [ Links ]
Acree T. E., Sonoff E.P., Splittstoesser D. F., 1972. Effect of yeast strain and type of sulphur compound on hydrogen sulphide production. Am. J. Enol. Vitic., 23, 6-9. [ Links ]
Agenbach W.A., 1977. A study of must nitrogen content in relation to incomplete fermentations, yeast production and fermentation activity. In: Proceedings of the South African Society of Enology and Viticulture. 66–87. Cape Town. Stellenbosch. [ Links ]
Alexandre H., Rousseaux I., Charpentier C., 1994. Ethanol adaptation mechanisms in Saccharomyces cerevisiae. Biotechnol. Appl. Biochem., 20, 173–183. [ Links ]
Alexandre H., Charpentier C., 1998. Biochemical aspects of stuck and sluggish fermentation in grape-must. J. Industrial Microbiol. Biotechnol. 20, 20-27. [ Links ]
Ancín C., Ayestarán B. , García A., Garrido J., 1998. Evolution of fatty acid contents in Garnacha and Viura musts during fermentation and the aging of wine. Z. Lebensm. Unters. Forsch. 226, 143-147. [ Links ]
Andreasson C., Neve E. P., Ljungdahl P. O., 2004. Four permeases import proline and the toxic proline analogue azetidine-2-carboxylate into yeast. Yeast, 21, 193-199. [ Links ]
Backhus L. E., DeRisi J., Brown P. O., Bisson L. F., 2001. Functional genomic analysis of a commercial wine strain of Saccharomyces cerevisiae under differing nitrogen conditions. FEMS Yeast Res., 1,111-125. [ Links ]
Barbosa C., Falco V., Mendes-Faia A., Mendes-Ferreira A., 2009. Nitrogen addition influences formation of aroma compounds, volatile acidity and ethanol in nitrogen deficient media fermented by Saccharomyces cerevisiae wine strains. J. Biosci. Bioeng., 108, 99–104. [ Links ]
Bardi L., Crivelli C., Marzona M., 1998. Esterase activity and release of ethyl esters of medium-chain fatty acids by Saccharomyces cerevisiae during anaerobic growth. Can. J. Microbiol., 44, 1171–76. [ Links ]
Bartowsky E.J., Pretorius I.S., 2009. Microbial formation and modification of flavor and off-flavor compounds in wine. In: Biology of microorganisms on grapes, in must and in wine. 209-232. Konig H., Unden, G. and Frohlich J. (ed.), Springer-Verlag, Heidelberg. [ Links ]
Bell A.A., Ough C.S., Kliewer W.M., 1979. Effects on must and wine composition, rate of fermentation, and wine quality of nitrogen fertilization of Vitis vinifera var. Thompson Seedless grapevines. Am. J. Enol. Vitic., 30, 124-129. [ Links ]
Bell S.J., Henschke P. A., 2005. Implications of nitrogen nutrition for grapes, fermentation and wine. Aust. J. Grape Wine Res., 11, 242-295. [ Links ]
Beltran G., Novo M., Rozes N., Mas A., Guillamon J.M., 2004. Nitrogen catabolite repression in Saccharomyces cerevisiae during wine fermentations. FEMS Yeast Res., 4, 625−632. [ Links ]
Beltran G., Esteve-Zarzoso B., Rozès N., Mas A., Guillamón J.M., 2005. Influence of the timing of nitrogen additions during synthetic grape-must fermentations on fermentation kinetics and nitrogen consumption. J. Agric. Food Chem., 53, 996–1002. [ Links ]
Beltran G., Novo M., Leberre V., Sokol S., Labourdette D., Guillamon J. M., Mas A., Francois J., Rozes N., 2006. Integration of transcriptomic and metabolic analyses for understanding the global responses of low-temperature winemaking fermentations. FEMS Yeast Res., 6, 1167-1183. [ Links ]
Beltran G., Novo M., Guillamón J. M., Mas A., Rozès N., 2008. Effect of fermentation temperature and culture media on the yeast lipid composition and wine volatile compounds. Int. J. Food Microbiol. , 121, 169–177. [ Links ]
Bely M., Sablayrolles J.M., Barre P., 1990. Automatic detection of assimilable nitrogen deficiencies during alcoholic fermentation in enological conditions. J. Ferment. Bioeng., 70, 246-252. [ Links ]
Bely M., Rinaldi A., Dubourdieu D., 2003. Influence of assimilable nitrogen on volatile acidity production by Saccharomyces cerevisiae during high sugar fermentation. J. Biosci. Bioeng., 96, 507-512. [ Links ]
Berthels N.J., Cordero Otero R.R., Bauer F.F., Thevelein J.M., Pretorius I.S., 2004. . Discrepancy in glucose and fructose utilisation during fermentation by Saccharomyces cerevisiae wine yeast strains. FEMS Yeast Res., 4, 683-689 [ Links ]
Bertram P.G., Choi J.H., Carvalho J., Ai W., Zeng W., Chan T.F., Zheng X.F., 2000. Tripartite regulation of Gln3p by TOR, Ure2p, and phosphatases. J. Biol. Chem. 275, 35727-35733. [ Links ]
Bertram P.G., Choi J.H., Carvalho J., Chan T.F., Ai W., Zheng X.F., 2002. Convergence of TOR-nitrogen and Snf1-glucose signalling pathways onto Gln3. Mol. Cell Biol., 22, 1246-1252. [ Links ]
Bisson L.F., Fraenkel D.G., 1983. Involvement of kinases in glucose and fructose uptake by Saccharomyces cerevisiae. Proc. Natl. Acad. Sci., 80, 1730-1734. [ Links ]
Bisson L.F., Neigeborn L., Carlson M., Fraenkel D.G., 1987. The SNF3 gene is required for high-affinity glucose transport in Saccharomyces cerevisiae. J. Bacteriol., 169, 1656–1662 [ Links ]
Bisson, L.F., 1991. Influence of nitrogen on yeast and fermentation of grapes. In: Proceedings of the International Symposium on nitrogen in grapes and wine. 78-89. American Society for Enology and Viticulture: Davis, CA. Seattle, USA. [ Links ]
Bisson L.F., 1993. Yeasts-Metabolism of sugars. In: Wine Microbiology and Biotechnology. 55-75. G.H. Fleet (ed.), Harwood Academic Publishers, Chur, Switzerland. [ Links ]
Bisson L.F., 1999. Stuck and sluggish fermentations. Am. J. Enol. Vitic., 50,107-119. [ Links ]
Bisson L.F., Karpel J.E., 2010. Genetics of Yeast Impacting Wine Quality. Annu. Rev. Food Sci. Technol., 1, 139–62.
Bohlscheid J.C., Edwards C.G., 2004. Interactive effects of nitrogen and biotin on yeast growth, fermentation rate, and volatile production. Am. J. Enol. Vitic., 55, 310A. [ Links ]
Bohlscheid J.C., Fellman J.K., Wang X.D., Ansen D., Edwards C.G., 2007. The influence of nitrogen and biotin interactions on the performance of Saccharomyces in alcoholic fermentations. J. Appl. Microbiol., 102, 390-400. [ Links ]
Boulton B., Singleton V.L., Bisson L.F., Kunkee R.E., 1996. Yeast and biochemistry of ethanol fermentation. In: Principles and Practices of Winemaking. 139-172. Chapman and Hall (eds.), New York. [ Links ]
Busturia A., Lagunas R., 1986. Catabolite inactivation of the glucose transport system in Saccharomyces cerevisiae. J. Gen. Microbiol., 132, 379-85.
Cardoso H., Leão C., 1992. Sequential inactivation of ammonium and glucose transport in Saccharomyces cerevisiae during fermentation. FEMS Microbiol. Lett., 73, 155-159. [ Links ]
Carlson M., 1999. Glucose repression in yeast. Curr Opin Microbiol., 2, 202-207. [ Links ]
Carrau F.M., Medina K., Boido E., Farina L., Gaggero C., Dellacassa E., Versini G., Henschke P.A., 2005. De novo synthesis of monoterpenes by Saccharomyces cerevisiae wine yeasts. FEMS Microbiol. Lett., 243, 107-115. [ Links ]
Carrau F.M., Medina K., Farina L., Boido E., Gaggero C., Henschke P.A., Dellacassa E., 2008. Production of fermentation aroma compounds by Saccharomyces cerevisiae wine yeasts: effects of yeast assimilable nitrogen on two model strains. FEMS Yeast Res., 8, 1196-1207. [ Links ]
Carrau F., Medina K., Fariña L., Boido E., Dellacassa E., 2010. Effect of Saccharomyces cerevisiae inoculum size on wine fermentation aroma compounds and its relation with assimilable nitrogen content. Int. J. Food Microbiol., 143, 81-85. [ Links ]
Cherest H., Davidian J.C., Thomas D., Benes V., Ansorge W., Surdin-Kerjan Y., 1997. Molecular characterization of two high affinity sulfate transporters in Saccharomyces cerevisiae. Genetics, 145, 627-35.
Chiva R., Baiges I., Mas A., Guillamon J.M., 2009. The role of GAP1 gene in the nitrogen metabolism of Saccharomyces cerevisiae during wine fermentation. J. Appl. Microbiol., 107, 235-244. [ Links ]
Clemente-Jimenez J.M., Mingorance-Cazorla L., Martínez-Rodríguez S., Las Heras-Vázquez F.J., Rodríguez-Vico F., 2005. Influence of sequential yeast mixtures on wine fermentation. Int. J. Food Microbiol., 98, 301– 308. [ Links ]
Cooper T.G., 1982. Nitrogen metabolism in Saccharomyces cerevisiae. In: The Molecular Biology of the Yeast Saccharomyces: Metabolism and Gene Expression. 39-99. Strathern J.N., Jones E.W. and Broach J.R. (eds.), Cold Spring Harbor Press, New York. [ Links ]
Cooper T.G., Sumrada R.A., 1983. What is the function of nitrogen catabolite repression in Saccharomyces cerevisiae? J. Bacteriol., 155, 623-627. [ Links ]
Dickinson J.R., Salgado L.E., Hewlins M.J., 2003. The catabolism of amino acids to long chain and complex alcohols in Saccharomyces cerevisiae. J. Biol. Chem., 278, 8028-34. [ Links ]
Diderich J.A., Schepper M., van Hoek P., Luttik M.A., van Dijken J.P., Pronk J.T., Klaassen P., Boelens H.F., de Mattos M.J., van Dam K., Kruckeberg A.L., 1999. Glucose uptake kinetics and transcription of HXT genes in chemostat cultures of Saccharomyces cerevisiae. J. Biol. Chem., 274, 15350-15359. [ Links ]
Didion T., Regenberg B., Jorgensen M.U., Kielland-Brandt M.C., Andersen H.A., 1998. The permease homologue Ssy1p controls the expression of amino acid and peptide transporter genes in Saccharomyces cerevisiae. Mol. Microbiol., 7, 643-650. [ Links ]
Donaton M.C., Holsbeeks I., Lagatie O., Van Zeebroeck G., Crauwels M., Winderickx J., Thevelein J.M., 2003. The Gap1 general amino acid permease acts as an amino acid sensor for activation of protein kinase A targets in the yeast Saccharomyces cerevisiae. Mol. Microbiol., 50, 911–929. [ Links ]
Dubourdieu D.T., Tominaga I., Masneuf C., 2000. The role of yeasts in grape flavor development during fermentation: the example of Sauvignon blanc. 50th Annual Meeting, Seattle American Society of Enology and Viticulture. 196–203. 19–23 June 2000, Seattle, WA. [ Links ]
Dubourdieu D., Tominaga T., Masneuf I., Peyrot des Gachons C., Murat M.L., 2006. The Role of Yeasts in Grape Flavor Development during Fermentation: The Example of Sauvignon blanc. Am J. Enol. Vitic., 57, 81-88. [ Links ]
Dukes B.C., Butzke C.E., 1998. Rapid determination of primary amino acids in grape-juice using an o-phthaldialdehyde/N-acetyl-L-cysteine spectrophotometric assay. Am. J. Enol. Vitic., 49, 125-134. [ Links ]
Erasmus D.J., Cliff M., van Vuuren H.J.J., 2004. Impact of yeast strain on the production of acetic acid, glycerol, and the sensory attributes of Icewine. Am. J. Enol. Vitic., 55, 371–378. [ Links ]
Etschman M.M.W., Bluemke W., Sell D., Schrader J., 2002. Biotechnological production of 2-phenylethanol. Appl. Microbiol. Biotechnol., 59, 1-8. [ Links ]
Filipe-Ribeiro L., Mendes-Faia A., 2007. Validation and comparison of analytical methods used to evaluate the nitrogen status of grape juice. Food Chem., 100, 1272-1277. [ Links ]
Fugelsang K.C., Edwards C.G., 2007. Yeasts. In: Wine Microbiology. Pratical applications and procedures . 3-28. Springer Science Business Media (ed.), LLC, New York, USA. [ Links ]
Fujii T., Nagasawa N., Iwamatsu A., Bogaki T., Tamai W., Hamachi M., 1994. Molecular cloning, sequence analysis, and expression of the yeast alcohol acetyltransferase gene. Appl. Environ. Microbiol., 60, 2786–92. [ Links ]
Fujii T., Kobayashi O., Yoshimoto H., Furukawa S., Tamai Y., 1997. Effect of aeration and unsaturated fatty acids on expression of the Saccharomyces cerevisiae alcohol acetyltransferase gene. Appl. Environ. Microbiol., 63, 910–915. [ Links ]
Fujiwara D., Yoshimoto H., Sone H., Harashima S., Tamai Y., 1998. Transcriptional co-regulation of Saccharomyces cerevisiae alcohol acetyltransferase gene ATF1 and D-9 fatty acid desaturase gene OLE1 by unsaturated fatty acids. Yeast, 14, 711–721. [ Links ]
Fujiwara D., Kobayashi O., Yoshimoto H., Harashima S., Tamai Y., 1999. Molecular mechanism of the multiple regulation of the Saccharomyces cerevisiae ATF1 gene encoding alcohol acetyltransferase. Yeast, 15, 1183–97. [ Links ]
Fukuda K., Yamamoto N., Kiyokawa Y., Yanagiuchi T., Wakai Y., Kitamoto K., Inoue Y., Kimura A., 1998. Balance of activities of alcohol acetyltransferase and esterase in Saccharomyces cerevisiae is important for production of isoamyl acetate. Appl. Environ. Microbiol., 64, 4076–78. [ Links ]
Fukuda K., Kiyokawa Y., Yanagiuchi T., Wakai Y., Kitamoto K., Inoue Y., Kimura A., 2000. Purification and characterization of isoamyl acetate-hydrolyzing esterase encoded by the IAH1 gene of Saccharomyces cerevisiae from a recombinant Escherichia coli. Appl. Microbiol. Biotechnol., 53, 596-600. [ Links ]
Gancedo J.M., 1998. Yeast carbon catabolite repression. Microbiol. Mol. Biol. Rev., 62, 334-361. [ Links ]
Gardner J.M., Poole K., Jiranek V., 2002. Practical significance of relative assimilable nitrogen requirements of yeast: a preliminary study of fermentation performance and liberation of H2S. Aust. J. Grape Wine Res., 8,175-179. [ Links ]
Gardner J.M., McBryde C., Vystavelova A., Barros Lopes M., Jiranek V., 2005. Identification of genes affecting glucose catabolism in nitrogen-limited fermentation. FEMS Yeast Res., 5, 791–800. [ Links ]
Gonçalves P., Rodrigues de Sousa H., Spencer-Martins I., 2000. FSY1, a novel gene encoding a specific fructose/ H(+) symporter in the type strain of Saccharomyces carlsbergensis. J. Bacteriol., 182, 5628-5630. [ Links ]
Gump B.H., Zoecklein B.W., Fugelsang K.C., Whiton R.S., 2002. Comparison of analytical methods for prediction of prefermentation nutritional status of grape juice. Am. J. Enol. Vitic., 53, 325-329. [ Links ]
Guymon J.F., Ingraham J.L., Crowell E.A., 1961. The influence of aeration on the formation of higher alcohols by yeast. Am. J. Enol. Vitic., 12, 60-66. [ Links ]
Hardie D.G., Carling D., Carlson M., 1998. The AMP-activated/SNF1 protein kinase subfamily: metabolic sensors of the eukaryotic cell? Annu. Rev. Biochem., 67, 821-55. [ Links ]
Hazelwood L.A., Daran J.M., van Maris A.J.A., Pronk J.T., Dickinson J.R., 2008. The Ehrlich Pathway for Fusel Alcohol Production: a Century of Research on Saccharomyces cerevisiae Metabolism. Appl. Environ. Microbiol., 74, 2259–2266. [ Links ]
Hedbacker K., Carlson M., 2008. SNF1/AMPK pathways in yeast. Front. Biosci., 13, 2408–2420. [ Links ]
Henschke P.A., Jiranek. V., 1991. Hydrogen sulfide formation during fermentation: effect of nitrogen composition in model grape-must. In: Proceedings of the international symposium on nitrogen in grapes and wine. 172-184. Rantz J.M. (ed.). Seattle, Washington, USA. [ Links ]
Henschke P.A., Jiranek. V., 1993. Yeasts-Metabolism of nitrogen compounds. In: Wine Microbiology and Biotechnology. 77-164. Fleet G.H. (ed.). Harwood Academic Publishers, Chur, Switzerland. [ Links ]
Henschke P.A., 1997. Wine yeast. In: Yeast sugar metabolism: biochemistry, genetics, biotechnology, and applications. 527–560. Zimmermann F.K., Entian K.D. (eds.), Technomic, Lancaster, Pennsylvania-USA. [ Links ]
Hernandéz-Orte P., Bely M., Cacho J., Ferreira V., 2006. Impact of ammonium additions on volatile acidity, ethanol, and aromatic compound production by different Saccharomyces cerevisiae strains during fermentation in controlled synthetic media. Aust. J. Grape Wine Res., 12, 150–160. [ Links ]
Horak J., 1997. Yeast nutrient transporters. Biochim. Biophys. Acta, 1331, 41-79. [ Links ]
Ingledew W.M., Magnus C.A., Sosulski F.W., 1987. Influence of oxygen on proline utilization during the wine fermentation. Am. J. Enol. Vitic., 38, 246-248. [ Links ]
Iraqui I., Vissers S., Bernard F., de Craene J.O., Boles E., Urrestarazu A., Andre B., 1999. Amino acid signaling in Saccharomyces cerevisiae: a permease-like sensor of external amino acids and F-Box protein Grr1p are required for transcriptional induction of the AGP1 gene, which encodes a broad-specificity amino acid permease. Mol. Cell Biol., 19, 989-1001. [ Links ]
Isnard A.D., Thomas D., Surdin-Kerjan Y, 1996. The study of methionine uptake in Saccharomyces cerevisiae reveals a new family of amino acid permeases. J. Mol. Biol., 262, 473-484 [ Links ]
Jauniaux J.C., Grenson M., 1990. GAP1, the general amino acid permease gene of Saccharomyces cerevisiae. Nucleotide sequence, protein similarity with the other bakers yeast amino acid permeases, and nitrogen catabolite repression. Eur. J. Biochem., 190, 39-44. [ Links ]
Javelot C., Girard P., Colonna-Ceccaldi B., Vladescu B., 1991. Introduction of terpene-producing ability in a wine strain of Saccharomyces cerevisiae. J. Biotechnol., 21, 239-251. [ Links ]
Jiménez-Martí E., Aranda A., Mendes-Ferreira A., Mendes-Faia A., del Olmo M., 2007. The nature of the nitrogen source added to nitrogen depleted vinifications conducted by a Saccharomyces cerevisiae strain in synthetic must affects gene expression and the levels of several volatile compounds. Antonie van Leeuwenhoek, 92, 61-75. [ Links ]
Jin Y.S., Laplaza J.M., Jeffries T.M., 2004. Saccharomyces cerevisiae engineered for xylose metabolism exhibits a respiratory response. Appl. Environ. Microbiol., 70, 6816–6825. [ Links ]
Jiranek V., Langridge P., Henschke P.A., 1990. Nitrogen requirement of yeast during wine fermentation. Proceedings of the seventh Australian wine industry technical conference. Williams, P.J.; Davidson, D.M.; Lee, T.H. (Eds) .; 166-171, Adelaide, SA. Adelaide, SA: Australian Industrial Publishers. [ Links ]
Jiranek V., Langridge P., Henschke P.A., 1991. Yeast nitrogen demand: selection criterion for wine yeasts for fermenting low nitrogen musts. Proceedings of the international symposium on nitrogen in grapes and wine. Rantz, J.M.(Ed). 266-269.Seattle, Washington, USA. Davis, CA: American Society for Enology and Viticulture. [ Links ]
Jiranek V., Langridge P., Henschke P.A., 1995a. Amino acid and ammonium utilization by Saccharomyces cerevisiae wine yeasts from a chemically defined medium. Am. J. Enol. Vitic., 46, 75-83. [ Links ]
Jiranek V., Langridge P., Henschke P.A., 1995b. Regulation of hydrogen sulphide liberation in wine producing S. cerevisiae strains by assimilable nitrogen. Appl. Environ. Microbiol., 61, 461-67. [ Links ]
Johnston M.J., Kim H., 2005. Glucose as a hormone: receptor-mediated glucose sensing in the yeast Saccharomyces cerevisiae. Biochem. Soc. Trans., 33, 247–252. [ Links ]
Karhumaa K., Wu B., Kielland-Brandt M.C., 2010. Conditions with high intracellular glucose inhibit sensing through glucose sensor Snf3 in Saccharomyces cerevisiae. J. Cellular Biochem., 110, 920–925. [ Links ]
Killian E., Ough C.S., 1979. Fermentation Esters Formation and Retention as Affected by Fermentation Temperature. Am. J. Enol. Vitic., 30, 301–305. [ Links ]
Kim J.H., Johnston M., 2006. Two glucose-sensing pathways converge on Rgt1 to regulate expression of glucose transporter genes in Saccharomyces cerevisiae. J. Biol. Chem., 281, 26144–26149. [ Links ]
Kitamoto K., Yoshizawa K., Ohsumi Y., Anraku Y., 1988. Dynamic aspects of vacuolar and cytosolic amino acid pools of Saccharomyces cerevisiae. J. Bacteriol. 170, 2683-2686. [ Links ]
Klasson H., Fink G.R., Ljungdahl P.O., 1999. Ssy1p and Ptr3p are plasma membrane components of a yeast system that senses extracellular amino acids. Mol. Cell Biol., 19, 5405-5416. [ Links ]
Klingshirn L.M., Liu J.R., Gallander J.F., 1987. Higher Alcohol Formation in Wines as Related to the Particle Size Profiles of Juice Insoluble Solids. Am. J. Enol. Vitic., 38, 207–210. [ Links ]
Kruckeberg A.L., 1996. The hexose transporter family of Saccharomyces cerevisiae. Arch. Microbiol., 166, 283-92. [ Links ]
Kunkee R.E., Guymon J.F., Crowell E.A., 1966. The formation of n-propyl alcohol by cell-free extracts of Saccharomyces cerevisiae. J. Inst. Brew., 72, 530-536. [ Links ]
Kunkee R.E., Guymon J.F., Crowell E.A., 1972. Studies on the control of higher alcohol formation by yeasts through metabolic inhibition. In: 1st Specialized International Symposium on yeasts. Kockova-Kratochivilova A. and Minarik E. (eds.), Publishing House of the Academy Sciences, Bratislava, Slovakia. [ Links ]
Kunkee R.E., 1991. Relationship between nitrogen content in must and sluggish fermentation. In: Proceedings of the international symposium on nitrogen in grapes and wine. 148-155. Rantz J.M., (ed.), American Society for Enology and Viticulture. Seattle, Washington, USA. [ Links ]
Lagunas R., Dominguez C., Busturia A., Saez S.J., 1982. Mechanisms of appearance of the Pasteur effect in Saccharomyces cerevisiae: Inactivation of sugar transport systems. J. Bacteriol., 152, 19-25. [ Links ]
Lambrechts M.G., Pretorius I.S., 2000. Yeast and its importance to wine aroma. A review. South African J. Enol. Vitic., 21, 97–129. [ Links ]
Leão C., van Uden N., 1984. Effects of ethanol and other alkanols on passive proton influx in the yeast Saccharomyces cerevisiae. Biochim. Biophys. Acta, 7741, 43-48. [ Links ]
Lilly M., Lambrechts M.G., Pretorius I.S., 2000. Effect of Increased Yeast Alcohol Acetyltransferase Activity on Flavor Profiles of Wine and Distillates. Appl. Environ. Microbiol., 66, 744-753. [ Links ]
Lilly M., Bauer F.F., Styger G., Lambrechts M.G., Pretorius I.S., 2006. The effect of increased branched-chain amino acid transaminase activity in yeast on the production of higher alcohols and on the flavour profiles of wine and distillates. FEMS Yeast Res., 6, 726–743. [ Links ]
Linderholm A.L., Findleton C.L., Kumar G., Hong Y., Bisson L.F., 2008. Identification of genes affecting hydrogen sulphide formation in Saccharomyces cerevisiae. Appl. Environ. Microbiol., 74, 1418-27. [ Links ]
Lorenz M.C., Heitman J., 1998. The MEP2 ammonium permease regulates pseudohyphal differentiation in Saccharomyces cerevisiae. EMBO J., 17, 1236-1247. [ Links ]
Magasanik B., Kaiser C.A., 2002. Nitrogen regulation in Saccharomyces cerevisiae. Gene, 290, 1-18. [ Links ]
Magasanik B., 2003. Ammonia assimilation by Saccharomyces cerevisiae. Eukaryot Cell, 2, 827-9. [ Links ]
Manginot C., Roustan J.L., Sablayrolles J.M., 1998. Nitrogen demand of different yeast strains during alcoholic fermentation. Importance of the stationary phase. Enzyme Microb. Tech., 23, 511-517. [ Links ]
Marini A.M., Soussi-Boudekou S., Vissers S., Andre. B., 1997. A family of ammonium transporters in Saccharomyces. Mol. Cell Biol., 17, 4282-4293. [ Links ]
Mason A.B., Dufour J.P., 2000. Alcohol acetyltransferases and the significance of ester synthesis in yeast. Yeast, 16, 1287-1298. [ Links ]
Mendes-Ferreira A., Clímaco M.C., Mendes Faia A., 2001. The role of non-Saccharomyces species in releasing glycosidic bound fraction of grape aroma components - a preliminary study. J. Appl. Microbiol., 91, 67-71. [ Links ]
Mendes-Ferreira A., Mendes-Faia A., Leão C., 2002. Survey of hydrogen sulphide production by wine yeasts. J. Food Prot., 65, 1033-1037. [ Links ]
Mendes-Ferreira A., Mendes-Faia A., Leão C., 2004. Growth and fermentation patterns of Saccharomyces cerevisiae under different ammonium concentrations and its implications in winemaking industry. J. Appl. Microbiol., 97, 540-545. [ Links ]
Mendes-Ferreira A., del Olmo M., Garcia-Martinez J., Jiménez-Martí E., Mendes-Faia A., Perez-Ortin J., Leão C., 2007a. Transcriptional response of Saccharomyces cerevisiae to different nitrogen concentrations during alcoholic fermentation. Appl. Environ. Microbiol., 73, 3049-60. [ Links ]
Mendes-Ferreira A., del Olmo M., Garcia-Martinez J., Jiménez-Martí E., Leão C., Mendes-Faia A., Perez-Ortin J., 2007b. Saccharomyces cerevisiae signature genes for predicting nitrogen deficiency during alcoholic fermentation. Appl. Environ Microbiol., 73, 5363-5369. [ Links ]
Mendes-Ferreira A., Barbosa C., Falco V., Leão C., Mendes-Faia A., 2009. Hydrogen sulphide and other aroma compounds production by wine strains of Saccharomyces cerevisiae in synthetic media with different nitrogen concentrations. J. Ind. Microb. Biotechn., 36, 571-583. [ Links ]
Mendes-Ferreira, A., Barbosa, C., Inês, A., Mendes-Faia, A. 2010a. The timing of diammonium phosphate supplementation of wine must affects subsequent H2S release during fermentation. J. Appl. Microbiol. 108, 540-549. [ Links ]
Mendes-Ferreira A., Barbosa C., Jiménez-Martí E., del Olmo M., Mendes-Faia A., 2010b. The wine yeast strain-dependent expression of genes implicated in sulfide production in response to nitrogen availability. J. Microbiol. Biotechn., 20, 1314-1321. [ Links ]
Millan M.C., Ortega J.M., 1988. Production of ethanol, acetaldehyde and acetic acid in wine by various yeast races: role of alcohol and aldehyde dehydrogenases. Am. J. Enol. Vitic., 39, 107–112. [ Links ]
Miller A.C., Wolff S.R., Bisson L.F., Ebeler S.E., 2007. Yeast Strain and Nitrogen Supplementation: Dynamics of Volatile Ester Production in Chardonnay Juice Fermentations. Am. J. Enol. Vitic., 58, 470-483. [ Links ]
Molina A.M., Swiegers J.H., Varela C., Pretorius I.S., Agosin E., 2007. Influence of wine fermentation temperature on the synthesis of yeast-derived volatile aroma compounds. Appl. Microbiol. Biotechnol., 77, 675-687. [ Links ]
Monk P.R., Cowley R.J., 1984. Effect of nicotinic acid and sugar concentration of grape-juice and temperature on accumulation of acetic acid during yeast yeast fermentation. J. Ferment. Technol., 62, 515–521. [ Links ]
Murat M.L., Masneuf I., Darriet P., Lavigne V., Tominaga T., Dubourdieu D.. 2001. Effect of the Saccharomyces cerevisiae yeast strains on the liberation of volatile thiols in Sauvignon blanc wines. Am. J. Enol. Vitic., 52, 136–139. [ Links ]
Nagasawa N., Bogaki T., Iwamatsu A., Hamachi M., Kumagai C., 1998. Cloning and nucleotide sequence of the alcohol acetyltransferase II gene (ATF2) from Saccharomyces cerevisiae Kyokai No. 7. Biosci. Biotechnol. Biochem., 62, 1852–1857. [ Links ]
Nikänen L., Nikänen I., 1977. Production of esters by different yeast strains in sugar fermentations. J. Inst. Brew., 83, 30-31. [ Links ]
Nikänen L., 1986. Formation and occurrence of flavor compounds in wine and distilled alcoholic beverages. Am. J. Enol. Vitic., 37, 84-96. [ Links ]
Nordström K., 1964. Formation of ethyl volatile acidity in fermentation with brewer's yeast. V. Effects of some vitamins and mineral nutrients. J. Inst. Brew., 70, 209-221. [ Links ]
Oshita K., Kubota M., Uchida M., Ono M., 1995. Clarification of the relationship between fusel alcohol formation and amino acid assimilation by brewing yeast using 13C-labeled amino acid. In: Proceedings of the European Brewing Convention. 387–394. Bruxelles. [ Links ]
Oswald M., Fischer M., Dirninger N., Karst F., 2007. Monoterpenoid biosynthesis in Saccharomyces cerevisiae. FEMS Yeast Res., 7, 413-21. [ Links ]
Ough, C.S., 1969. Ammonia content of California grapes. Am. J. Enol. Vitic., 20, 213-220. [ Links ]
Ough, C.S., Bell A.A., 1980. Effects of nitrogen fertilization of grapevines on amino acid metabolism and higher alcohol formation during grape-juice fermentation. Am. J. Enol. Vitic., 31, 122-123. [ Links ]
Ough C.S., Lee T.H., 1981. Effect of vineyard nitrogen fertilization level on the formation of some fermentation esters. Am. J. Enol. Vitic., 32, 125-127. [ Links ]
Özcan S., Johnston M., 1999. Function and regulation of yeast hexose transporters. Microbiol Mol. Biol. Rev., 63, 554-69. [ Links ]
Peinado R.A., Moreno J., Bueno J.E., Moreno J.A., Mauricio J.C., 2004. Comparative study of aromatic compounds in two young white wines subjected to pre-fermentative cryomaceration. Food Chem., 84, 585-590. [ Links ]
Perez M., Luyten K., Michel R., Riou C., Blondin B., 2005. Analysis of Saccharomyces cerevisiae hexose carrier expression during wine fermentation: Both low- and high-affinity Hxt transporters are expressed. FEMS Yeast Res., 5, 351–361. [ Links ]
Peyrot des Gachons C., Van Leeuwen C., Tominaga T., Soyer J.P., Gaudillère J.P., Dubourdieu D., 2005. Influence of water and nitrogen deficit on fruit ripening and aroma potential of Vitis vinifera L. cv. Sauvignon blanc in field conditions. J. Sci. Food Agric., 85, 73–85. [ Links ]
Pretorius I.S., 2000. Tailoring wine yeast for the new millennium: novel approaches to the ancient art of winemaking. Yeast, 16, 675-729. [ Links ]
Pronk J.T., de Steensma H.Y., Van Dijken J.P., 1996. Pyruvate metabolism in Saccharomyces cerevisiae. Yeast, 12, 1607-33. [ Links ]
Radler F., 1993. Yeasts-metabolism of organic acids. In: Wine microbiology and biotechnology. 165–223. Fleet G.H. (ed.), Harwood Academic Publishers, Chur, Switzerland. [ Links ]
Rankine B.C., 1967. Formation of higher alcohols by wine yeasts, and relationship to taste threshold. J. Sci. Fd. Agr., 17, 584-589. [ Links ]
Rapp A., Versini G., 1991. Influence of nitrogen compounds in grapes on aroma compounds of wine. In: Proceedings of the International Symposium on Nitrogen in Grapes and Wines. 156–164. Rantz J.M. (ed.), CA: American Society for Enology and Viticulture. Seattle, Washington, USA. [ Links ]
Rauhut D., 1993. Yeasts - production of sulfur compounds. In: Wine Microbiology and Biotechnology. 183-223. Fleet G.H. (ed.), Harwood Academic Publishers, Chur, Switzerland. [ Links ]
Regenberg B., During-Olsen L., Kielland-Brandt M.C., Holmberg S., 1999. Substrate specificity and gene expression of the amino-acid permeases in Saccharomyces cerevisiae. Curr Genet., 36, 317-328. [ Links ]
Ribéreau-Gayon P., Dubourdieu D., Donèche B., Lonvaud A., 2000. Biochemistry of alcoholic fermentation and metabolic pathways of wine yeasts. In: Handbook of Enology, Volume 1. The microbiology of wine and vinifications. 53-69. John Wiley & Sons Lda. Chichester, UK. [ Links ]
Rodrigues de Sousa H., Spencer-Martins I., Gonçalves P., 2004. Differential regulation by glucose and fructose of a gene encoding a specific fructose/H+ symporter in Saccharomyces sensu stricto yeasts.Yeast, 21, 519-530. [ Links ]
Romano P., Fiore C., Paraggio M., Caruso M., Capece A., 2003. Function of yeast species and strains in wine flavour. Intern. J. Food Microbiol., 86, 169-180. [ Links ]
Rosi Y., Vinella M., Domizio P., 1994. Characterization of P-glucosidase activity in yeasts of oenological origin. J. Appl. Bact., 77, 519-527. [ Links ]
Rossignol T., Dulau L., Julien A., Blondin B., 2003. Genome-wide monitoring of wine yeast gene expression during alcoholic fermentation. Yeast, 16, 1369-1385. [ Links ]
Rupela O.P, Tauro P., 1985. Genetic control of hydrogen sulfide retention in Saccharomyces cerevisiae. Biotechnol. Lett., 7, 279-282. [ Links ]
Sá-Correia I., Salgueiro S.P., Viegas C.A., Novais J.M., 1989. Leakage induced by ethanol and octanoic and decanoic acids in Saccharomyces cerevisiae. Yeast (special issue), 5, S123–S127. [ Links ]
Saerens S.M., Verstrepen K.J., Van Laere S.D.M., Voet A.R.D., Van Dijck P., Delvaux F.R., Thevelein J.M., 2006. The Saccharomyces cerevisiae EHT1 and EEB1 genes encode novel enzymes with medium-chain fatty acid ethyl ester synthesis and hydrolysis capacity. J Biol Chem 281, 4446-56. [ Links ]
Saerens S.M.G., Delvaux F., Verstrepen K.J., Van Dijck P., Thevelein J.M., Delvaux F.R., 2008. Parameters Affecting Ethyl Ester Production by Saccharomyces cerevisiae during Fermentation. Appl. Environ. Microbiol., 74, 454-461. [ Links ]
Saerens S.M., Delvaux F.R., Verstrepen K.J., Thevelein J.M., 2010. Production and biological function of volatile esters in Saccharomyces cerevisiae. Microb. Biotechnol., 3, 165-177. [ Links ]
Salmon J.M., 1989. Effect of sugar inactivation on sluggish and stuck oenological fermentations. Appl. Env. Microbiol,. 55, 953–958. [ Links ]
Salmon J.M., Vincent O., Mauricio J.C., Bely M., Barre P., 1993. Sugar Transport Inhibition and Apparent Loss of Activity in Saccharomyces cerevisiae as a Major Limiting Factor of Enological Fermentations. Am. J. Enol. Vitic., 44, 56-64. [ Links ]
Schutz M., Kunkee R.E., 1977. Formation of Hydrogen Sulfide from Elemental Sulfur During Fermentation by Wine Yeast. Am. J. Enol. Vitic., 28, 137-144. [ Links ]
Shimazu Y., Watanabe M., 1981. Effects of yeast strains and environmental conditions on formation of organic acids in must during fermentation. J. Ferment. Technol., 59, 27-32. [ Links ]
Shively C.E., Henick-Kling T., 2001. Comparison of two procedures for assay of free amino nitrogen. Am. J. Enol. Vitic., 52, 400-401. [ Links ]
Singh R., Kunkee R.E., 1977. Multiplicity and control of alcohol dehydrogenase isoenzymes in various strains of wine yeasts. Arch. Microbiol., 114, 255-259. [ Links ]
Soles R.M., Ough C.S., Kunkee R.E., 1982. Ester Concentration Differences in Wine Fermented by Various Species and Strains of Yeasts. Am. J. Enol. Vitic., 33, 94 - 98. [ Links ]
Spiropoulos A., Tanaka J., Flerianos I., Bisson L.F., 2000. Characterization of hydrogen sulfide in commercial and natural wine isolates of Saccharomyces. Am. J. Enol. Vitic., 51, 233-248. [ Links ]
Sponholz W.R., 1991. Nitrogen compounds in grapes, must and wine. In: Proceedings of the international symposium on nitrogen in grapes and wine. 67-77. J.M. Rantz J.M. (ed.), CA: American Society for Enology and Viticulture. Seattle, Washington, USA.
Stratford M., Rose A.H., 1985. Hydrogen sulphide production from sulphite by Saccharomyces cerevisiae. J. Gen. Microbiol., 131, 1417-1424. [ Links ]
Subileau M., Schneider R., Salmon J.M., Degryse E., 2008. Nitrogen catabolite repression modulates the production of aromatic thiols characteristic of Sauvignon Blanc at the level of precursor transport. FEMS Yeast Res., 8, 771-780. [ Links ]
Swiegers J.H., Pretorius I.S., 2005. Yeast modulation of wine flavor. Adv. Appl. Microbiol., 57, 131-175. [ Links ]
Swiegers J.H., I.S. Pretorius. 2007. Modulation of volatile sulfur compounds by wine yeast. Appl. Microbiol. Biotechnol., 74, 954-960.
Swiegers J.H., Kievit R.L., Siebert T., Lattey K.A., Bramley B.R., Francis I.L., King E.S., Pretorius I.S., 2009. The influence of yeast on the aroma of Sauvignon Blanc wine. Food Microbiol., 26, 204–211. [ Links ]
ter Schure E.G., van Riel N.A., Verrips C.T., 2000. The role of ammonia metabolism in nitrogen catabolite repression in Saccharomyces cerevisiae. FEMS Microbiol. Rev., 24, 67-83. [ Links ]
Thibon C., Marullo P., Claisse O., Cullin C., Dubourdieu D., Tominaga T., 2008. Nitrogen catabolic repression controls the release of volatile thiols by Saccharomyces cerevisiae during wine fermentation. FEMS Yeast Research., 8, 1076-1086. [ Links ]
Thomas C.S., Boulton R.B., Silacci M.W., Gubler W.D., 1993. The effect of elemental sulfur, yeast strain and fermentation medium on hydrogen sulfide production during fermentation. Am. J. Enol. Vitic., 44, 211-216. [ Links ]
Thomas D., Surdin-Kerjan Y., 1997. Metabolism of sulfur amino acids in Saccharomyces cerevisiae. Microbiol. Mol. Biol. Rev., 61, 503-532. [ Links ]
Thomsson E., Gustafsson L., Larsson C., 2005. Starvation response of Saccharomyces cerevisiae grown in anaerobic nitrogen- or carbon-limited chemostat cultures. Appl. Environ. Microbiol., 71, 3007–3013. [ Links ]
Thurston P.A., Quain D.E., Tibb R.S., 1982. Lipid metabolism and the regulation of volatile ester synthesis in Saccharomyces cerevisiae. J. Inst. Brew., 88, 90–94. [ Links ]
Tomenchok D.M., Brandriss C.M., 1987. Gene-enzyme relationships in the proline biosynthetic pathway of Saccharomyces cerevisiae. J. Bacteriol., 169, 5364-5372. [ Links ]
Torrea D., Fraile P., Garde T., Ancín C., 2003. Production of volatile compounds in the fermentation of chardonnay musts inoculated with two strains of Saccharomyces cerevisiae with different nitrogen demands. Food Control, 14, 565–571. [ Links ]
Torrens J., Urpí P., Riu-Aumatell M., Vichi S., López-Tamames E., Buxaderas S., 2008. Different commercial yeast strains affecting the volatile and sensory profile of cava base wine. Int. J. Food Microbiol., 124, 48-57. [ Links ]
Tromp A., 1984. The effect of yeast strain, grape solids, nitrogen and temperature on fermentation rate and wine quality. S. Afr. J. Enol. Vitic., 5, 1-6. [ Links ]
Uemura T., Kashiwagi K., Igarashi K., 2005. Uptake of putrescine and spermidine by Gap1p on the plasma membrane in Saccharomyces cerevisiae. Biochem. Biophys. Res. Commun., 328, 1028-1033. [ Links ]
Ugliano M., Henschke P.A., 2009. Yeasts and wine flavour. In: Wine Chemistry and Biochemistry. 313-392. Moreno-Arribas M.V. & Polo M.C. (eds.), Springer Science Business, New York. [ Links ]
van Nuland A., Vandormael P., Donaton M., Alenquer M., Lourenço A., Quintino E., Versele M., Thevelein J.M., 2006. Ammonium permease-based sensing mechanism for rapid ammonium activation of the protein kinase A pathway in yeast. Mol. Microbiol., 59, 1485–1505. [ Links ]
van Uden N., 1989. Effects of alcohols on membrane transport in yeasts. In: Alcohol Toxicity in Yeasts and Bacteria. 135-146. van Uden N. (ed.), CRC Press, Boca Raton, Florida, USA. [ Links ]
Varela C., Pizarro F., Agosin E., 2004. Biomass Content Governs Fermentation Rate in Nitrogen-Deficient Wine Musts. Appl. Environ. Microbiol., 70, 3392-3400. [ Links ]
Vaudano E., Moruno E.G., Di Stefano R., 2004. Modulation of Geraniol Metabolism During Alcohol Fermentation. J. Inst. Brew., 110, 213–219. [ Links ]
Verstrepen K.J., Derdelinckx G., Dufour J-P., Winderickx J., Thevelein J.M., Pretorius I.S., Delvaux F. R.. 2003a. Flavour-active esters: adding fruitiness to beer. J. Biosci. Bioeng., 96, 110-118. [ Links ]
Verstrepen K.J., Van Laere S.D.M, Vanderhaegen B.M.P., Derdelinckx G., Dufour J-P, Pretorius I.S., Winderickx J., Thevelein J.M., Delvaux F.R., 2003b. Expression levels of the yeast alcohol acetyltransferase genes ATF1, Lg-ATF1, and ATF2 control the formation of a broad range of volatile esters. Appl. Environ. Microbiol., 69, 5228-37. [ Links ]
Verstrepen K.J., Van Laere S.D.M., Vercammen J., Dufour J-P., Pretorius I.S., Winderickx J., Thevelein J.M., Delvaux F.R., 2004. The Saccharomyces cerevisiae alcohol acetyl transferase Atf1p is localized in lipid particles. Yeast, 21, 367-77. [ Links ]
Vilanova M., Ugliano M., Varela C., Siebert T., Pretorius I.S., Henschke P.A., 2007. Assimilable nitrogen utilisation and production of volatile and non-volatile compounds in chemically defined medium by Saccharomyces cerevisiae wine yeasts. Appl. Microbiol. Biotechnol., 77, 145–157. [ Links ]
Vos P.J.A., Gray R.S., 1979. The origin and control of H2S during fermentation of grape-must. Am. J. Enol. Vitic., 30, 187-197. [ Links ]
Walker G.M., 1998. Yeast Physiology and Biotechnology. 350 p. John Wiley & Sons, Inc., New York. [ Links ]
Wang X.D., Bohlscheid J.C., Edwards C.G., 2003. Fermentative activity and production of volatile compounds by Saccharomyces grown in synthetic grape-juice media deficient in assimilable nitrogen and/or pantothenic acid. J. Appl. Microbiol., 94, 1-11. [ Links ]
Webb A.D., Ingraham J.L., 1963. Fusel oil. Adv. Appl. Microbiol., 5, 317–353. [ Links ]
Wurz R.E.M., Kepner R.E., Webb A.D., 1988. The Biosynthesis of Certain Gamma-Lactones from Glutamic Acid by Film Yeast Activity on the Surface of Flor Sherry. Am. J. Enol. Vitic., 39, 234-238. [ Links ]
Yoshimoto H., Momma T., Fujiwara D., Sone H., Kaneko Y., Tamai T., 1998. Characterization of the ATF1 and Lg-ATF1 genes encoding alcohol acetyltransferases in the bottom fermenting yeast Saccharomyces pastorianus. J. Ferment. Bioeng., 86, 15–20. [ Links ]
Yoshimoto H., Fujiwara D., Momma T., Tanaka K., Sone H., Nagasawa N., Tamai T., 1999. Isolation and characterization of the ATF2 gene encoding alcohol acetyl transferase II in the bottom fermenting yeast Saccharomyces pastorianus. Yeast, 15, 409–417. [ Links ]
Yoshimoto H., Fukushige T., Yonezawa T., Sone H., 2002. Genetical and physiological analysis of branched-chain alcohols and isoamyl acetate production in Saccharomyces cerevisiae. Appl. Microbiol. Biotechnol., 59, 501–8. [ Links ]
Younis O.S., Stewart G.G., 1998. Sugar uptake and subsequent ester and higher alcohol production by Saccharomyces cerevisiae. J. Inst. Brew., 104, 255–64. [ Links ]
Zamora F., 2009. Biochemistry of alcoholic fermentation. In: Wine Chemistry and Biochemistry. 3-26. Moreno-Arribas M.V. & Polo M.C. (eds.), Springer Science Business, New York. [ Links ]
Zea L., Moreno J., Ortega J.M., Medina M., 1995. Content of Free Terpenic Compounds in Cells and Musts Vinification with three Saccharomyces cerevisiae. J. Agric. Food Chem., 43, 1110-1114. [ Links ]
ACKNOWLEDGMENTS
The research carried out in our laboratory and presented in this paper was financially supported by FEDER, by Fundação para a Ciência e Tecnologia, (FCT) and by PTDC projects (contracts PTDC/AGR-ALI/71460/2006, PTDC/AGR-ALI/68284/2006 and PTDC/AGR-ALI/111224/2009). C.B. is a recipient of a Ph.D. grant from FCT (SFRH/BD/61881/2009).













