Serviços Personalizados
Journal
Artigo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Acessos
Acessos
Links relacionados
-
 Similares em
SciELO
Similares em
SciELO
Compartilhar
Revista de Ciências Agrárias
versão impressa ISSN 0871-018X
Rev. de Ciências Agrárias vol.38 no.2 Lisboa jun. 2015
ARTIGO
Early events of grapevine resistance towards downy mildew by a systems biology approach
Compreensão dos mecanismos de resistência da videira ao míldio usando uma abordagem de biologia de sistemas
Andreia Figueiredo1, Mónica Sebastiana1, Joana Martins2, Filipa Monteiro1, Ana V. Coelho2 e Maria S. Pais1
1 Plant Molecular Biology and Biotechnology Group, Biosystems & Integrative Sciences Institute (BioISI), Science Faculty of Lisbon University, 1749-016 Lisboa, Portugal. E-mails: aafigueiredo@fc.ul.pt, author for correspondence; mgsebastiana@fc.ul.pt; fimonteiro@fc.ul.pt; msalomepais@gmail.com
2 Instituto de Tecnologia Química e Biológica António Xavier, Universidade Nova de Lisboa, Av. da Republica – EAN, 2780-157 Oeiras, Portugal. E-mails: jfmartins88@gmail.com; varela@itqb.unl.pt
ABSTRACT
Grapevine downy mildew, caused by the fungus Plasmopara viticola (Berk. & Curt.) Berl. & de Toni was introduced in European vineyards in the 1870s and quickly spread to all major grape-producing regions worldwide. The early events of grapevine resistance to P. viticola were accessed using transcriptomic, metabolomic and proteomics approaches in the cultivars, ‘Regent' and ‘Trincadeira', resistant and susceptible to downy mildew, respectively. At the transcript level the resistant genotype ‘Regent' presents signalling and defence-related transcripts up-regulated as soon as 6hpi. At the metabolome level, our results show that ‘Trincadeira' and ‘Regent' are intrinsically different as ‘Regent', the resistant cultivar, is able to activate the phenylpropanoid pathway and to accumulate linolenic acid, the precursor of jasmonate in the octadecanoid cascade. At the proteome level ‘Regent' presents upregulated proteins mainly belonging to the functional categories of photosynthesis, carbohydrate metabolism and defence, indicating that this cultivar makes a more efficient use of light, in CO2 assimilation and obtaining intermediate metabolites from photoassimilates for the biosynthesis of secondary metabolites and subsequent defence responses. Our data brings new insights on the molecular processes underlying resistance to Plasmopara viticola, which may eventually enable the design of novel strategies for pathogen control.
Keywords: grapevine, metabolomics, proteomics, resistance, transcritomics,
RESUMO
O míldio da videira, causado pelo fungo oomycete obrigatório Plasmopara viticola (Berk. & Curt.) Berl. & de Toni foi introduzido na Europa em 1870 e rapidamente se espalhou para todas as regiões produtoras de vinho no mundo. Neste estudo foram utilizadas abordagens de transcritómica, proteómica e metabolómica e duas cultivares de videira, ‘Regent' e ‘Trincadeira', resistente e suscetível ao fungo P. vitícola tendo em vista a caracterização dos mecanismos de defesa. Ao nível da transcritómica, o genótipo resistente ‘Regent' apresenta uma ativação de transcritos associados a processos de sinalização e defesa, 6 horas após inoculação. No que se refere à metabolómica, os resultados obtidos demonstram que os dois genótipos são inerentemente diferentes sendo que na cultivar ‘Regent' há uma ativação da via biossintética dos fenilpropanoides e uma acumulação de ácido linolénico, o precursor do ácido jasmónico logo após a inoculação com o P. viticola. Ao nível da proteómica verificou-se que no genótipo resistente há indução de proteínas ligadas à fotossíntese, metabolismo dos carbohidratos e defesa após a inoculação com o fungo, o que sugere que este genótipo é mais eficaz na assimilação de CO2, na produção de energia através da fotossíntese e na produção de compostos intermediários para subsequente ativação dos mecanismos de defesa. Os nossos resultados constituem um contributo para a compreensão dos processos moleculares subjacentes à ativação de mecanismos de defesa no genótipo resistente ‘Regent', o que se poderá refletir no estabelecimento de novas estratégias para o controlo desta doença.
Palavras-chave: videira, metabolómica, proteómica, resistência, transcritómica
Introduction
Grapevine (Vitis vinifera L.) is currently the most important fruit plant cultivated worldwide due to its economic impact in the wine industry. This industry is highly strategic for the Portuguese economy since it accounts for more than 680 million euros per year of exports (Global Agricultural Information Network, 2013). Grapevine downy mildew is caused the oomycete fungus Plasmopara viticola (Berk. et Curt.) Berl. et de Toni, which has been introduced in Europe during the 19th century and is still considered one of the most destructive grapevine diseases affecting leaves, fruits, and shoots thereby causing significant yield losses and reduction in berry quality. Unfortunately, most Vitis vinifera cultivars, commonly used for wine production, are susceptible to downy mildew. Hence, typical viticulture requires extensive fungicide applications every growing season, with great impact in the economic costs and the environment (Gessler et al., 2011). Since nearly all V. vinifera cultivars are susceptible to P. viticola, resistance traits have been introduced from other Vitis species through breeding programs. American grapevines exhibit natural host resistance against P. viticola (Jurges et al., 2009) and have been used in breeding programs for resistance to grapevine pathogens resulting on the obtention of V. vinifera cultivars such as ‘Regent' and ‘Solaris' (Vitis International Variety Catalogue 2011). Grapevine resistance to downy mildew has a large genotypic component with different cultivars responding to infection with defense reactions presenting variable promptness and magnitude, timing, and intensity (Kortekamp and Zyprian 2003; Jurges et al., 2009). Advances towards the elucidation of this specific pathosystem have been made using approaches from transcriptional analysis (Figueiredo et al., 2008; Kortekamp et al., 2008; Polesani et al., 2008; Polesani et al., 2010; Wu et al., 2010; Malacarne et al., 2011) to quantitative trait loci (QTL) analysis of segregating populations (Dalbo et al., 2000; Fischer et al., 2004; Welter et al., 2007; Bellin et al., 2009; Peressotti et al., 2010; Blasi et al., 2011; Schwander et al., 2012; Venuti et al., 2013). Despite the knowledge gathered on grapevine-downy mildew interaction, recent reports documented the emergence of P. viticola resistance-breaking isolates in Europe (Peressotti et al., 2010). These findings support the need to continue identifying resistance candidate genes and to pursue breeding strategies for performing knowledge-based pyramiding of resistance in appropriate genetic backgrounds, thus contributing for the increase of resistance durability (Palloix et al., 2009).
In our previous studies (Figueiredo et al., 2008; Figueiredo et al., 2012; Ali et al., 2012; Monteiro et al., 2013; Figueiredo et al, unpublished data) we have compared two different grapevine genotypes (resistant and susceptible) prior and post-inoculation with downy mildew using field and greenhouse grown plants. We have used an ‘Omics approach towards elucidating the defence mechanisms of the resistant genotype, ‘Regent', towards P. viticola. In the next sections we highlight the outcomes of the different ‘Omics approaches, integrating knowledge gathered trough a systems biology approach. Future research directions will be defined.
Grapevine resistance to P. viticola: a systems biology approach
Two representative resistant and susceptible Vitis vinifera cultivars (‘Regent' and ‘Trincadeira') were compared using transcritome, metabolome and proteome analysis. ‘Regent', bred at the Institute for Grapevine Breeding Geilweilerhof in Germany, was chosen as a model since its resistance traits were achieved by multiple crosses introgressing resistance genes from American wild species (Eibach and Topfer, 2003). It combines high wine quality and resistance to downy and powdery mildew pathogens. The cultivar ‘Trincadeira' is a highly susceptible Portuguese cultivar used to produce much appreciated Portuguese wines with high economic interest (Figueiredo et al., 2008).
For the systems biology approach, we have studied both genotypes under field conditions in order to get the constitutive differences that may account for downy mildew resistance (non-inoculated). Afterwards we have used greenhouse controlled conditions (inoculated and non-inoculated) in order to access cultivar-specific kinetics of gene induction after downy mildew inoculation. We have used costume-made cDNA microarrays, proton NMR spectroscopy (1H NMR) and two dimension difference gel electrophoresis (2D-DIGE) for the transcriptome, metabolome and proteome analysis.
Transcript and metabolic profiling of ‘Regent' and ‘Trincadeira' leaves under field conditions
A combined metabolomics and transcriptomics approach was used to identify transcripts and metabolites which could constitutively discriminate ‘Regent' and ‘Trincadeira' (Figueiredo et al., 2008). Under field conditions the resistant genotype ‘Regent' presents an induction of several transcripts known to be associated with defence against stress, such as subtilisin-like protease, S-adenosyl methionine synthase (AdoMet synthase), phenylalanine ammonia lyase (PAL), WD-repeat protein like, and J2P protein. ‘Regent' also presents a higher accumulation of metabolites such as inositol, alanine, glutamine, glutamate and caffeic acid (Figueiredo et al., 2008). All together, these data suggest that secondary metabolite production is constitutively higher in ‘Regent' than in ‘Trincadeira' which is corroborated by the higher accumulation of caffeic acid in the resistant cultivar (Figueiredo et al., 2008). Moreover, the constitutive up-regulation of a subtilisin-like protein and accumulation of inositol in the resistant cultivar may be related to a higher signal perception and transduction capacity, enabling a rapid response to pathogen attack (Hamzehzarghani et al., 2005; Ramirez et al., 2013).
Transcript, metabolic and proteomic profiling of ‘Regent' and ‘Trincadeira' leaves under greenhouse conditions
Prior to pathogen challenge, differences in basal gene expression levels between the two grapevine cultivars were determined by comparing uninfected samples of both genotypes under greenhouse conditions. The differential expression of 22 genes was found between both genotypes with the major differences being associated to photosynthesis (27.2%) and general metabolism (18.2%) (Figueiredo et al., 2012). Also, ‘Regent' constitutively accumulated more phenolic compounds (caffeoyl derivative: trans-caftaric acid, feruloyl derivative: trans-fertaric acid, and quercetin-3-O-glucoside) than ‘Trincadeira' (Table 1) (Ali et al., 2012). At a proteome level, proteins related to photosynthesis, carbohydrate and metabolism discriminated both genotypes (Figueiredo et al., submitted). The results obtained by these approaches suggest that ‘Regent' may be more efficient than ‘Trincadeira' in cellular mechanisms like CO2 assimilation, transformation of light into chemical energy, and production of intermediate metabolites from photoassimilates, needed for the biosynthetic pathways. There has long been evidence that a constitutive expression of resistance leads to a reduction in plant growth and fitness as a consequence of “metabolic competition” directed towards the synthesis of defense elements (Walters and Heil, 2007). The increased intensity of spots corresponding to components of the primary metabolism and biosynthetic machinery in ‘Regent' could pay for the fitness cost of the constitutive resistance, hence resulting in more stress-resistant plants.
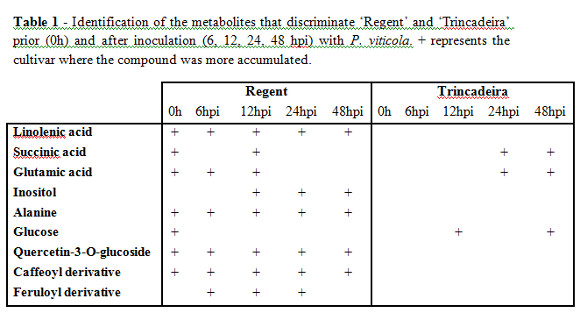
After pathogen challenge a broad reprogramming of the transcriptome occurs (Fig.1A) with major changes associated to the stress response and biotic stimulus and with the regulation of metabolic and biological processes. Several transcripts appear to be preferentially activated in ‘Regent' when compared to ‘Trincadeira', representing a number of functional categories whose expression appears to be coordinated, such as signalling and defense (Figueiredo et al., 2012). Signalling and defense appear to be more activated in ‘Regent' as soon as 6 hours post inoculation (hpi), with transcripts encoding signal transduction components like subtilisin-like proteins, IQ calmodulin-binding region- Apoptosis regulator Bcl-2 protein, cyclophilin and CBL-interacting serine/threonine protein kinase induced specifically in ‘Regent' (Figueiredo et al., 2012; Monteiro et al., 2013). Apart from signalling events, several defense-related transcripts are preferentially induced in ‘Regent' at 6 and 12hpi as is the case of a protein with homology to leucine-rich repeat (LRR) receptor like proteins, PR10 proteins and a chitinase-like protein. Of the metabolism-related transcripts we have shown an activation of a lipid binding protein and of a chalcone synthase suggesting activation of both lipid and phenylpropanoid metabolisms (Figueiredo et al., 2012). The metabolome analysis by 1HNMR revealed considerable differences between both genotypes with a higher accumulation of secondary metabolites in ‘Regent' upon pathogen challenge (Table 1). The accumulation of metabolites such as trans-caftaric acid (caffeoyl derivative), trans-fertaric acid (feruloyl derivative), and quercetin-3-O-glucoside, in the resistant genotype suggests their possible involvement in successful defense against downy mildew. Also the first 12hpi seems to be very critical with ‘Regent' showing an increased synthesis of phenolics in this time period. Moreover it was shown that linolenic acid accumulates in ‘Regent' after inoculation with P. viticola (Ali et al., 2012). Since linolenic acid is the precursor of jasmonate in the octadecanoid cascade, these results open new insights into jasmonate signaling in grapevine resistance against a biotrophic fungus as P. viticola.
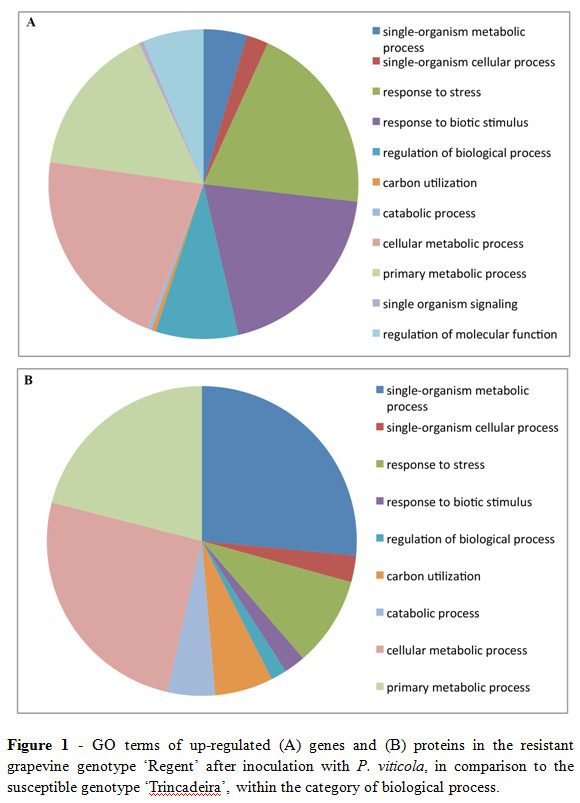
At a proteome level, the major differences between resistant and susceptible cultivars account for proteins involved in response to stress and biotic stimulus and also to the regulation of primary and cellular metabolic processes (Fig.1B).
Oxidative stress and defense-related proteins were found to be down-regulated in ‘Regent' during the early interaction (up to 24hpi). However, at 24hpi defense related proteins appear up-regulated in the resistant genotype. Also, a putative plastid lipid associated protein increases its expression at 12hpi, corroborating both transcriptome and metabolome data and suggesting that there is an activation of lipid metabolism in ‘Regent'. This activation may be related to jasmonic acid signalling.
Conclusions
This systems biology approach allowed us to characterize the genotype-associated differences prior and after P. viticola challenge. We have shown that both genotypes are inherently different and that constitutive metabolic differences may account for a faster response to a pathogen stimulus in the resistant cultivar. We also demonstrated that the susceptible cultivar ‘Trincadeira' appears to mount a resistance reaction, which is neither fast nor robust enough to prevent the pathogen from spreading. With these approaches new research strategies are already under development, such as the characterization of subtilisin-like proteins as targets for introgression in breeding programs and the confirmation of jasmonic acid as a key player on grapevine signalling against the biotrophic fungus P. viticola.
Acknowledgments
This work was funded by Portuguese National Funds through Fundação para a Ciência e a Tecnologia (FCT) in the frame of the project PTDC/AGR-GPL/119753/2010 and grant SFRH/BPD/ 63641/2009.
References
Ali, K.; Maltese, F.; Figueiredo, A.; Rex, M., Fortes, A.M.; Zyprian, E.; Pais, M.S.; Verpoorte, R. and Choi, Y.H. (2012) - Alterations in grapevine leaf metabolism upon inoculation with Plasmopara viticola in different time-points. Plant Science, vol. 191, p. 100-107. [ Links ]
Bellin, D.; Peressotti, E.; Merdinoglu, D.; Wiedemann-Merdinoglu, S.; Adam-Blondon, A.; Cipriani, G.; Morgante, M.; Testolin, R. and Di Gaspero, G. (2009) - Resistance to Plasmopara viticola in grapevine 'Bianca' is controlled by a major dominant gene causing localised necrosis at the infection site. Theoretical and Applied Genetics, vol. 120, n. 1, p. 163-176. [ Links ]
Blasi, P.; Blanc, S.; Wiedemann-Merdinoglu, S.; Prado, E.; Ruhl, E.; Mestre, P. and Merdinoglu, D. (2011) - Construction of a reference linkage map of Vitis amurensis and genetic mapping of Rpv8, a locus conferring resistance to grapevine downy mildew. Theoretical and Applied Genetics, vol. 123, n. 1, p. 43-53. [ Links ]
Dalbo, M.; Ye, G.; Weeden, N.; Steinkellner, H.; Sefc, K. and Reisch, B. (2000) - A gene controlling sex in grapevines placed on a molecular marker-based genetic map. Genome, vol. 43, n. 2, p. 333-340. [ Links ]
Eibach R. and Toepfer R. (2003) - Success in resistance breeding ‘Regent' and its steps into the market. Acta Horticulturae, vol. 603, p. 687–691. [ Links ]
Figueiredo, A.; Fortes, A.M.; Ferreira, S.; Sebastiana, M., Choi, Y.H., Sousa, L.; Acioli-Santos, B., Pessoa, F.; Verpoorte, R. and Pais, M.S. (2008) - Transcriptional and metabolic profiling of grape (Vitis vinifera L.) leaves unravel possible innate resistance against pathogenic fungi. Journal of Experimental Botany, vol. 59, n. 12, p. 3371-3381. [ Links ]
Figueiredo, A.; Monteiro, F.; Fortes, A., Bonow-Rex, M.; Zyprian, E.; Sousa, L. and Pais, M. (2012) - Cultivar-specific kinetics of gene induction during downy mildew early infection in grapevine. Functional & Integrative Genomics, vol. 12, n. 2, p. 379-386. [ Links ]
Fischer, B.; Salakhutdinov, I.; Akkurt, M.; Eibach, R.; Edwards, K.; Topfer, R. and Zyprian, E. (2004) - Quantitative trait locus analysis of fungal disease resistance factors on a molecular map of grapevine. Theoretical and Applied Genetics, vol. 108, n. 3, p. 501-515. [ Links ]
Gessler, C.; Pertot, I. and Perazzolli, M. (2011) - Plasmopara viticola: a review of knowledge on downy mildew of grapevine and effective disease management. Phytopathologia Mediterranea, vol. 50, n. 1, p. 3-44. [ Links ]
Hamzehzarghani, H.; Kushalappa, A.; Dion, Y.; Rioux, S.; Comeau, A.; Yaylayan, V.; Marshall, W. and Mather, D. (2005) - Metabolic profiling and factor analysis to discriminate quantitative resistance in wheat cultivars against fusarium head blight. Physiological and Molecular Plant Pathology, vol. 66, n. 4, p. 119-133. [ Links ]
Jurges, G.; Kassemeyer, H.; Durrenberger, M.; Duggelin, M. and Nick, P. (2009) - The mode of interaction between Vitis and Plasmopara viticola Berk. & Curt. Ex de Bary depends on the host species. Plant Biology, vol. 11, n. 6, p. 886-898. [ Links ]
Kortekamp, A.; Welter, L., Vogt, S.; Knoll, A.; Schwander, F., Topfer, R. and Zyprian, E. (2008) - Identification, isolation and characterization of a CC-NBS-LRR candidate disease resistance gene family in grapevine. Molecular Breeding, vol. 22, n. 3, p. 421-432. [ Links ]
Kortekamp, A. and Zyprian, E. (2003) - Characterization of Plasmopara-resistance in grapevine using in vitro plants. Journal of Plant Physiology, vol. 160, n. 11, p. 1393-1400. [ Links ]
Malacarne, G.; Vrhovsek, U.; Zulini, L.; Cestaro, A.; Stefanini, M.; Mattivi, F.; Delledonne, M.; Velasco, R. and Moser, C. (2011) - Resistance to Plasmopara viticola in a grapevine segregating population is associated with stilbenoid accumulation and with specific host transcriptional responses. BMC Plant Biology, vol. 11, p. 114. [ Links ]
Monteiro, F.; Sebastiana, M.; Pais, M.S. and Figueiredo, A. (2013) - Reference Gene Selection and Validation for the Early Responses to Downy Mildew Infection in Susceptible and Resistant Vitis vinifera Cultivars. Plos One, vol. 8, n. 9, p. e72998. [ Links ]
Palloix, A.; Ayme, V. and Moury, B. (2009) - Durability of plant major resistance genes to pathogens depends on the genetic background, experimental evidence and consequences for breeding strategies. New Phytologist, vol. 183, n. 1, p. 190-199. [ Links ]
Peressotti, E.; Wiedemann-Merdinoglu, S.; Delmotte, F.; Bellin, D.; Di Gaspero, G.; Testolin, R.; Merdinoglu, D. and Mestre, P. (2010) - Breakdown of resistance to grapevine downy mildew upon limited deployment of a resistant variety. BMC Plant Biology, vol. 10, p. 147. [ Links ]
Polesani, M.; Bortesi, L.; Ferrarini, A.; Zamboni, A.; Fasoli, M.; Zadra, C.; Lovato, A.; Pezzotti, M.; Delledonne, M. and Polverari, A. (2010) - General and species-specific transcriptional responses to downy mildew infection in a susceptible (Vitis vinifera) and a resistant (V. riparia) grapevine species. BMC Genomics, vol. 11, p. 117. [ Links ]
Polesani, M.; Desario, F.; Ferrarini, A.; Zamboni, A.; Pezzotti, M.; Kortekamp, A. and Polverari, A. (2008) - CDNA-AFLP analysis of plant and pathogen genes expressed in grapevine infected with Plasmopara viticola. BMC Genomics, vol. 9, p. 142. [ Links ]
Ramirez, V.; Lopez, A., Mauch-Mani, B.; Gil, M. and Vera, P. (2013) - An Extracellular Subtilase Switch for Immune Priming in Arabidopsis. Plos Pathogens, vol. 9, n. 6, p. e1003445. [ Links ]
Schwander, F.; Eibach, R.; Fechter, I.; Hausmann, L.; Zyprian, E. and Topfer, R. (2012) - Rpv10: a new locus from the Asian Vitis gene pool for pyramiding downy mildew resistance loci in grapevine. Theoretical and Applied Genetics, vol. 124, n. 1, p. 163-176. [ Links ]
Venuti, S.; Copetti, D.; Foria, S.; Falginella, L.; Hoffmann, S.; Bellin, D.; Cindric, P., Kozma, P.; Scalabrin, S.; Morgante, M.; Testolin, R. and Di Gaspero, G. (2013) - Historical Introgression of the Downy Mildew Resistance Gene Rpv12 from the Asian Species Vitis amurensis into Grapevine Varieties. Plos One, vol. 8, n. 4, p. e61228. [ Links ]
Walters, D. and Heil, M. (2007) - Costs and trade-offs associated with induced resistance. Physiological and Molecular Plant Pathology, vol. 71, n. 1-3, p. 3-17. [ Links ]
Welter, L.; Gokturk-Baydar, N.; Akkurt, M.; Maul, E.; Eibach, R.; Topfer, R. and Zyprian, E. (2007) - Genetic mapping and localization of quantitative trait loci affecting fungal disease resistance and leaf morphology in grapevine (Vitis vinifera L). Molecular Breeding, vol. 20, n. 4, p. 359-374. [ Links ]
Wu, J.; Zhang, Y.; Zhang, H.; Huang, H.; Folta, K. and Lu, J. (2010) - Whole genome wide expression profiles of Vitis amurensis grape responding to downy mildew by using Solexa sequencing technology. BMC Plant Biology, vol. 10, p. 234. [ Links ]
Received/Recebido: 2015.02.27
Accepted/Aceite: 2015.04.30














