Serviços Personalizados
Journal
Artigo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Acessos
Acessos
Links relacionados
-
 Similares em
SciELO
Similares em
SciELO
Compartilhar
Revista de Ciências Agrárias
versão impressa ISSN 0871-018X
Rev. de Ciências Agrárias vol.39 no.4 Lisboa dez. 2016
https://doi.org/10.19084/RCA16104
ARTIGO
Biological nitrogen fixation by Phaseolus vulgaris
Fixação biológica de azoto em Phaseolus vulgaris
Ricardo Soares, Jesus Tejo, Maria Manuela Veloso and Isabel Videira e Castro1
1 Instituto Nacional de Investigação Agrária e Veterinária, I.P. (INIAV, I.P), Quinta do Marquês, Av. República, Nova Oeiras, 2784-159 Oeiras, Portugal. *E-mail: isabel.castro@iniav.pt
ABSTRACT
Phaseolus vulgaris L. (common bean) is considered a promiscuous legume in its association with rhizobia since it can be nodulated by several species of the Rhizobiaceae family. In a field experiment in Oeiras (Portugal), strains were isolated from the root nodules of P. vulgaris 675. Each strain was inoculated in two P. vulgaris landraces: Pv 648 and Pv 675. Phylogeny of 16S rRNA, recA and nodC genes were inferred, using partial gene sequences. The results of plant inoculation assays have shown that only one of the isolated strains could successfully nodulate at least the P. vulgaris landrace (Pv 675), with an index of effectiveness of 31%. This strain was identified as belonging to Rhizobium sullae species by the 16S rRNA and recA genes but it also has a nodC gene from Rhizobium gallicum. The remaining strains were identified as belonging to R. gallicum, Agrobacterium tumefaciens and A. arsenijevicii.
Keywords: Biological nitrogen fixation, nodulation, Phaseolus vulgaris, phylogenetic characterization, rhizobia
RESUMO
Phaseolus vulgaris L., conhecido por feijão comum, é uma leguminosa promíscua que é nodulada por várias espécies de bactérias da família Rhizobiaceae. No decorrer de um estudo sobre bactérias Rhizobium que nodulam esta leguminosa, foram isoladas estirpes de bactérias dos nódulos radiculares de P. vulgaris (landrace 675), num ensaio de campo instalado em Oeiras, Portugal. Para a caracterização das estirpes recorreu-se à filogenia dos genes 16S rRNA, recA e nodC, usando sequências parciais destes genes. Os resultados dos ensaios efetuados com dois tipos de feijão comum, 675 e 648, revelaram que apenas uma estirpe foi capaz de nodular uma das landraces de P. vulgaris testadas, obtendo 31% de eficácia simbiótica com P. vulgaris 675. Esta estirpe foi identificada como Rhizobium sullae, possuindo no entanto o gene nodC relativo à espécie Rhizobium gallicum. As restantes estirpes isoladas foram identificadas como R. gallicum, Agrobacterium tumefaciens e A. arsenijevicii.
Palavras-chave: Fixação biológica de azoto, caraterização filogenética, nodulação, Phaseolus vulgaris, rizóbio
Introduction
Legumes (Fabaceae) are a family of dicotyledoneous plants. Most of these plants form a symbiosis with diazotrophic nitrogen fixing soil bacteria (collectively known as rhizobia) within a specialized structure, the root nodule (van Rhijn and Vanderleyden, 1995; Kamboj et al., 2008). Rhizobia can take up gaseous dinitrogen (N2) from the air and fix it into ammonia that can be subsequently assimilated into amino acids by the bacterium or the plant. In return, the plant provides the rhizobia with a carbon source in the form of dicarboxylic acids (Soussi et al., 1999). The multienzyme complex responsible for nitrogen fixation, nitrogenase, is irreversibly damaged when exposed to oxygen. The plant produces leghemoglobin, a protein related to human hemoglobin, for providing oxygen to the nodules. So, functional nodules generally have a pink color.
Legumes are important food and feed crops all over the world (Dita et al., 2006), partly due to their high nitrogen content, a result of the symbiosis with the rhizobia. The biological nitrogen fixation (BNF) plays an important role in the cropping system and some legumes could fix nitrogen better than others. For instance, faba bean (Vicia faba L.) was found to be very efficient while common bean (P. vulgaris) is in general rather poor (Hardarson and Danso, 1993; Carranca et al., 1999). An example is shown in Figura 1, indicating the quantities of total N fixed by grain legumes in Europe. It is seen that the highest N fixing species is faba bean and the least fixing is chickpea. However, the values are also a reflection of the areas occupied by each crop grown (Baddeley et al., 2013).
Legumes have also been widely used as green manure since the beginnings of agriculture. This practice adds nitrogen to the soil and improves soil quality by increasing the organic matter content but it has diminished due the availability of industrially produced fertilizers (Zahran, 1999).
There are 76 species of Phaseolus (Freytag and Debouck, 2002) that can be grouped in two clades (Delgado-Salinas et al., 2006). The most important crops of these species, used as food are: P. vulgaris (common bean), P. coccineus (scarlet runner bean), P. acutifolius (tepary bean), P. polyanthus (year bean) and P. lunatus (lima bean). The common bean (P. vulgaris) is the third most important grain legume growing worldwide in many parts of the tropics, subtropics and temperate regions, being only overcome by Glycine max L. (soybean) and Arachis hypogaea L. (peanut) (Singh, 1999). The domestication of common bean ocurred independently in South and Central Americas (Gepts and Bliss, 1986) and its dissemination into and across Europe was a result of introductions associated with several exchanges (Zeven, 1999; Angioi et al., 2010). The origin of Portuguese common bean germplasm although unclear is associated with the Era of the Discoveries, 16th century (Santalla et al., 1994; Rodiño et al., 2001). In Portugal, about 95% of the bean production is located in the North and Centre of the country, contributing up to 90% to the Portuguese national bean production (INE, 2014). A considerable proportion of agriculture in Portugal is still traditional and farmers cultivate their own landraces, common bean being usually intercropped with other species, mainly maize.
Rhizobia is currently composed of 16 genera that are distributed in two subclasses of Proteobacteria, most of them in the alfa-Proteobacteria, with 13 genera, six of them where rhizobia have been traditionally included, namely Rhizobium, Ensifer (formerly Sinorhizobium), Mesorhizobium, Bradyrhizobium, Azorhizobium, and Allorhizobium. Recently, bacteria isolated from legume nodules were phylogenetically placed out of the conventional groups of rhizobia, which include the following seven genera, Aminobacter, Devosia, Methylobacterium, Microvirga, Ochrobactrum, Phyllobacterium and Shinella. Also bacteria belonging to the subclasse of beta-Proteobacteria include three different genera namely Burkholderia, Cupriavidus and Herbaspirillum (Castro et al., 2016; Weir, 2016). Like other legumes, P. vulgaris forms nitrogen-fixing symbiosis with bacteria belonging to different genera and species, being studied worldwide, mainly in America where this legume has its distribution centers. Initially, based on the cross-inoculation-group concept, all bean-nodulating rhizobia were classified as Rhizobium leguminosarum bv. phaseoli (Jordan, 1984). However, common bean is very promiscuous in its association with rhizobia and several species of rhizobia have been described over the years to be endosymbionts of P. vulgaris besides R. leguminosarum, such as Rhizobium etli (Segovia et al., 1993), Rhizobium gallicum, Rhizobium giardinii (Amarger et al., 1997), Rhizobium tropici (Martínez-Romero et al., 1991), Rhizobium lusitanum (Valverde et al., 2006), Rhizobium multihospitium (Han et al., 2008), Rhizobium phaseoli (Ramírez-Bahena et al., 2008), Rhizobium vallis (Wang et al., 2011), Rhizobium leucaenae (Ribeiro et al., 2012), Rhizobium grahamii, Rhizobium mesoamericanum (López-López et al., 2012) and Rhizobium azibense (Mnasri et al., 2014).
Descriptions of bean rhizobia from the Iberian Peninsula refer to isolates that were originated from soils of Spain (Herrera-Cervera et al., 1999; Rodriguez-Navarro et al., 2000) and Portugal (Valverde et al., 2006). The majority of the isolates originating from Spain were characterized as R. etli, R. gallicum and R. giardinii, probably imported from the Americas along with the seeds (Silva et al., 2003; Zurdo-Piñeiro et al., 2004; Beyhaut et al., 2006). Isolates from Portugal were proposed to represent the new species R. lusitanum, having characteristics that are different from the rhizobial species from Spain (Valverde et al., 2006). To our knowledge, no evidence for the presence of R. lusitanum in soils of the Americas has been reported. However, in Portugal the information about rhizobia nodulating and fixing nitrogen in common bean is very scarce.
The objective of the present study was to investigate rhizobia endosymbionts which nodulate Portuguese landraces of P. vulgaris and to characterize their ability to promote plant growth, as well as their correspondent phylogeny using several genes sequences such as 16S rRNA, recA and nodC. The gene 16S rRNA is commonly used for taxonomic proposes. However, in many cases this conventional gene is not sufficiently discriminative (Fox et al., 1992; Clayton et al., 1995) and so, other genes, like recA and nodC were also included in this study to better determine taxonomic positions of the isolated strains. recA is an housekeeping gene with fast molecular evolution rates (Gaunt et al., 2001; Rivas et al., 2009; Lorite et al., 2012), while nodC is a symbiotic gene involved in the first steps of nodulation.
Materials and Methods
Isolation of bacteria from root nodules
A field experiment was sown with two Portuguese common bean landraces (Pv 648 and Pv 675) at Quinta do Marquês in Oeiras (Lisbon), Portugal, in the Spring-Summer of 2014. The landraces accessions were obtained during a germplasm collecting mission that took place in 1982 in the North of the country (Trás-os-Montes region). The seeds are maintained at the Research Unit of Biotechnology and Genetic Resources, INIAV – Oeiras.
Bacteria were isolated from root nodules of beans plants (landraces 648 and 675) in the end of growth season. A total of 12 nodules (8 from Pv 648 and 4 from Pv 675) were surface sterilized with 0.1% (w/v) HgCl2, washed extensively with sterilized water and crushed in a Petri dish. A droplet of the nodule suspension was streaked on yeast-mannitol agar (YMA) plate containing congo red (Vincent, 1970). Well isolated colonies (obtained only from three nodules), corresponding each to a pure isolate, Phv-675-1, Phv-675-2, Phv-675-3A and Phv-675-3B, were stored refrigerated at 4 ºC for further characterization.
Plant inoculation assay
Seeds of common bean Pv 648 and Pv 675 were surface-sterilized using 0.1% (w/v) of HgCl2 for 4 min and washed extensively with sterilized water. For each treatment, three pre-germinated seeds were transferred one to each flask containing inert sand with 50 ml liquid Jensen medium (Jensen, 1941) corresponding to three replica per treatment. Strains isolated in this study from root nodules were inoculated using 1 ml liquid Jensen (1/4 diluted) suspension with approximately 108 cells. Uninoculated (T0) and nitrogen (TN) controls were included by adding 1 ml liquid Jensen medium (¼ diluted) and 1 ml 1.75% (w/v) KNO3, respectively. A positive control was also included using the strain Phv-C, which belongs to the INIAV Collection of Rhizobia Bacteria, and that is known to nodulate efficiently P. vulgaris. This strain was inoculated in the same conditions used for the others strains. Plants were grown in a controlled environmental chamber with a photoperiod of 16 h light and 8 h dark cycle at 23 ? (day)/18 ? (night). After 8 weeks of growth, the presence of nodules was examined and shoots were dried in an oven at 80 ºC during 48 h. Data were analysed by one-way analysis of variance (ANOVA), followed by the Fisher LSD test at P = 0.05.
The values of shoots dry weight (X) were also used to calculate the index of effectiveness (Es) according to the formula Es (%) = (Xs - XT0)/(XTN - XT0) x 100 (Ferreira and Marques, 1992), where Xs represents the mean dry weight of inoculated shoots; XTN the mean dry weight of plants with nitrogen control; XT0 the mean dry weight of uninoculated plants.
Phylogenetic analysis
Genomic DNA of bacterial isolates was extracted from logarithmic phase cultures pre-grown overnight in TY media (Beringer, 1974) using the Bio-Rad Aqua Pure Genomic DNA extraction kit. Primers 41f/1488r (Weisburg et al., 1991), recA63/recA555 (Gaunt et al., 2001) and nodCFu/nodCI (Laguerre et al., 2001) were used for the amplification of 16S rRNA, recA and nodC genes, respectively. Taq Master mix (Qiagen kit) and 20 - 40 ng of total genomic DNA of each strain were also used for amplification. For each strain, one amplified fragment was used for sequencing. RecA and nodC genes amplification and sequencing were only performed after the confirmation of genus Rhizobium by 16S rRNA gene sequencing. Chromas Lite software (version 2.1.1) was used for the visualization and edition of the sequences and the database BLASTn of NCBI was used to find homologous sequences. For phylogenetic analysis, Mega (version 6) software (Tamura et al., 2013) was used and the neighbor-joining (NJ) (Saitou and Nei, 1987) and bootstrap (Felsenstein, 1985) methods were selected. Phylogenetic distances were calculated using p-distance nucleotide substitution model and the pairwise deletion option. Confidence values were based on 1000 bootstrap replications.
Results
Plant inoculation assay
Plant inoculation assays were performed only with strains isolated from Pv 675, due to failure of obtaining bacteria isolates from Pv 648. This failure may be due to the fact that nodules of landrace 648 were white inside and seemed like large tumors. It was found that, among the strains isolated in this study, only strain Phv-675-1 was able to nodulate the original host, P. vulgaris 675 (Figure 2), and no strains were able to nodulate the landrace Pv 648. On other hand, the strain Phv-C, used as control, always formed nodules in both hosts (Table 1).

Data of the shoots dry weight of both landraces when inoculated separately with strains Phv-675-1, 675-2, and Phv-C (Figure 3) showed highest values in the assays with Pv 675, the original host. When the host plant was the landrace Pv 648, only the plants inoculated with strain Phv-C had higher values of dry weight than the uninoculated control (T0), although these differences were not statistically significant. Plants inoculated with Phv-C strain, in landrace Pv 675, also showed high dry weight values, statistically similar to the control TN, and without significant differences. On the other hand, values of dry weight of both P. vulgaris were very similar when inoculated with strains Phv-675-1 and Phv-675-2. Differently, the strain Phv-675-3A induced in landrace Pv 675 the lowest values, even much lower than the T0 control and having differences statistically significant.
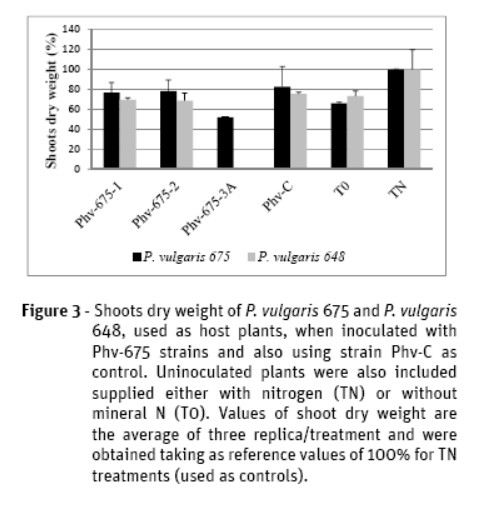
The index of effectiveness of rhizobia strains nodulating Pv 675 was calculated, having the strain Phv-675-1 an Es of 31% and the strain Phv-C an Es of 50%. However, this last strain had a low value of Es (about 10%) concerning Pv 648.
Phylogenetic analysis
The PCR reaction of each sample produced expected bands of approximately 1500, 500 and 900 bp when amplifying the 16S rRNA, recA and nodC genes, respectively. As shown in Figures 4 and 5, and Table 2, strain Phv-675-1 was clustered with R. sullae WSM1592, having 99.7% of sequence identity with 16S rRNA and 99.3% with recA genes. Strain Phv-675-2 was clustered with R. gallicum bv. gallicum R620, in both 16S rRNA and recA genes, with 100% and 99.8% of identity, respectively. When analyzing the symbiotic nodC gene (Figure 6) strains Phv-675-1 and Phv-675-2 were clustered together with 2 strains of R. gallicum bv. gallicum, R602 and RHM47 (isolated from P. vulgaris), and with one strain of R. gallicum, SPT1-23a (isolated from Ammopiptanthus sp.), showing at least 99.7% of sequence identity. However, Phv-675-1 and Phv-675-2 had the highest similarity (99.8 and 99.9%, respectively) with R. gallicum bv. gallicum R602 (see Table 2).
Strain Phv-C, used as control in plant inoculation assays, was previously identified as R. azibense, through the amplification and subsequent sequencing of the 16S rRNA gene (data not published), and was included herein in the phylogenetic tree of the 16S rRNA gene along with the remaining strains (Figure 4).
The 16S rRNA phylogenetic analysis of strains Phv-675-3A and Phv-675-3B (Figure 4) showed that these strains were clustered with 99.6% of identity with a different species of bacteria, A. tumefaciens Ach5 and A. arsenijevicii AL5.1, respectively.
Figure 5Discussion
The phylogenetic analysis of strains isolated in this study revealed the existence of several species among them, which is not surprising because P. vulgaris is considered very promiscuous in its association with rhizobia. One of the strains, Phv-675-1, was identified as R. sullae, through 16S rRNA and recA genes. R. sullae has never been isolated before from P. vulgaris, although R. sullae is described as being the endossymbiont of sulla (Hedysarum coronarium L.) (Squartini et al., 2002), but few studies report information about its host range, ecology and phylogeny. In fact, R. sullae is considered to be highly specific to Hedysarum coronarium L. and no cross-nodulations with other host legume species were reported (Casella et al., 1984; Glatzle et al., 1986). However, when analyzing the sequence of the nodC gene, this strain was identified has belonging to R. gallicum, suggesting that the transference of symbiotic plasmid could be involved between these two Rhizobium species. It is known that the essential information for symbiotic and saprophytic life cycles of Rhizobium genus is encoded in plasmids (García-de los Santos et al., 1996). The symbiotic genes, nod (for nodulation), nif and fix (for nitrogen fixation), are located in a megaplasmid, and for this reason lateral transference of symbiotic genes intra and inter-species usually occur and are part of Rhizobium evolution (Nuti et al., 1979; Hombrecher et al., 1981). On the other hand, in the field of the study area there was no previous record of common bean cultivation, having sulla grown spontaneously over the years. Together with the fact that P. vulgaris is a very promiscuous host, this could explain the appearance of this unusual and not described endosymbiont of P. vulgaris. The results obtained in this study, from plant inoculation assays, although preliminary, reveal that strain Phv-675-1 nodulated only one of the P. vulgaris landraces tested and was poorly effective to fix nitrogen (Es=31%). The strain used in this work as a positive control, Phv-C, nodulated both landraces. Interestingly, this strain was identified as R. azibense, which represents a novel Rhizobium species recently characterized (Mnasri et al., 2014) of nitrogen fixing bacteria isolated from root-nodules of P. vulgaris. Although, Phv-C was more effective with landrace 675 (Es=50%), it is not considered highly effective following the criteria adopted by Ferreira and Marques (1992) (where values of Es should be higher than 75% to be considered highly effective strains in nitrogen fixation). However, for the landrace 648 this strain was considered as ineffective. These results showed the importance of host plant in the symbiosis, i.e., in the nodulation and nitrogen fixation.
Strain Phv-675-2 was identified as R. gallicum, which is one of the previously described rhizobial species found to nodulate and fix nitrogen with P. vulgaris (Herrera-Cervera et al., 1999) and first identified from common bean nodules in France (Amarger et al., 1997). However, this strain was not able to nodulate any landraces of P. vulgaris tested here (Table 1). Probably other factors, besides the inexistence of nod genes, had influenced in the plant nodulation assay, like the loss of symbiotic plasmid.
Strain Phv-675-3A did not nodulate the landraces 675 of P. vulgaris and had strong homologies with tumor inducing strains like A. tumefaciens and A. arsenijevicii in the 16S rRNA gene. Moreover, in the plant assays, the inoculation with strain Phv-675-3A had an unfavorable effect when compared with T0 control plants. For these reasons, the strains Phv-675-3A and Phv-675-3B may have been isolated from a tumor rather than a root nodule, but several authors (Mhamdi et al., 2005; Mrabet et al., 2006) had shown that Agrobacterium strains could colonize mature nodules and this hypothesis must also be considered.
Conclusion
The diversity of rhizobia nodulating P. vulgaris has been widely studied, but, because of the promiscuous nature of this plant concerning its association with rhizobia, novel endosymbionts should be expected as more ecological niches are examined. Interestingly, in this study a rhizobia strain with high percentage of similarity with R. sullae was identified. This specie had never been described before as endosymbiont of common bean.
The present work analyzed few strains nodulating landraces of common bean used by local farmers from the region of Bragança, in the north-east of Portugal. However, this study points to the need to study the diversity of rhizobia nodulating common bean in soils where these traditional varieties have been used by farmers for centuries, in our country. Such research will allow better understanding of the symbiosis between P. vulgaris and rhizobia, and thus will contribute to reduce the need for nitrogen fertilization. It will be possible to select strains for common bean with high nitrogen fixing capacity to be used as inoculants. This procedure can contribute to the sustainability of traditional cropping systems.
Acknowledgments
Jesus Trejo was granted by Programa Leonardo da Vinci, Fundecyt, Spain. The authors are grateful to Nádia Castanheira for the support in the statistical analysis.
References
Amarger, N.; Macheret, V. and Laguerre, G. (1997) - Rhizobium gallicum sp. nov. and Rhizobium giardinii sp. nov., from Phaseolus vulgaris nodules. International Journal of Systematic Bacteriology, vol. 47, n. 4, p. 996–1006. [ Links ]
Angioi, S.A.; Rau, D.; Attene, G.; Nanni, L.; Belluci, E.; Logozzo, G.; Negri, V.; Spagnoletti Zeuli, P. L. and Papa R. (2010) - Beans in Europe: origin and structure of the European landraces of Phaseolus vulgaris L. Theoretical and Applied Genetics, vol. 121, p. 828-843. http://dx.doi.org/10.1007/s00122-010-1353-2 [ Links ]
Baddeley, J.A.; Jones, S.; Topp, C.F.E.; Watson, C.A; Helming, J. and Stoddard, F.L. (2014) - Biological nitrogen fixation (BNF) by legume crops in Europe. Legume Futures Report 1.5; Legume-Supported Cropping Systems for Europe [cit.2016-07-25]. http://www.legumefutures.de/images/Legume_Futures_Report_1.5.pdf [ Links ]
Beringer, J.E. (1974) - R factor transfer in Rhizobium leguminosarum. Journal of General Microbiology, vol. 84, n. 1, p. 188–198. http://dx.doi.org/10.1099/00221287-84-1-188 [ Links ]
Beyhaut, E.; Tlusty, B.; van Berkum, P. and Graham, P.H. (2006) - Rhizobium giardinii is the microsymbiont of Illinois bundleflower (Desmanthus illinoensis (Michx.) Macmillan) in midwestern prairies. Canadian Journal of Microbiology, vol. 52, n. 9, p. 903–907. http://dx.doi.org/10.1139/w06-051 [ Links ]
Carranca, C.; de Varennes, A. and Rolston, D. (1999) - Biological nitrogen fixation by fababean, pea and chickpea, under field conditions, estimated by the isotope dilution technique. European Journal of Agronomy, vol. 10, n. 1, p. 49–56. http://dx.doi.org/10.1016/S1161-0301(98)00049-5 [ Links ]
Casella, S.; Gault R.R.; Reynolds, K.C.; Dyson, J.R. and Brockwell., J. (1984) - Nodulation studies on legumes exotic to Australia: Hedysarum coronarium. FEMS Microbiology Letters, vol. 22, n. 1, p. 37–45. http://dx.doi.org/10.1111/j.1574-6968.1984.tb00350.x [ Links ]
Castro, I.V.; Fareleira, P. and Ferreira, E. (2016) - Nitrogen Fixing Symbioses in a Sustainable Agriculture. In: Hakeem, K.R.; Akhtar, M.S. & Abdullah, S.N.A. (Eds) - Plant, Soil and Microbes. Volume 1: Implications in Crop Science, p. 55-91. [ Links ]
Clayton, R.A.; Sutton, G.; Hinkle, P.S. Jr; Bult, C. and Fields, C. (1995) - Intraspecific variation in small-subunit rRNA sequences in GenBank: why single sequences may not adequately represent prokaryotic taxa. International Journal of Systematic Bacteriology, vol. 45, n. 3, p. 595–599. [ Links ]
Delgado-Salinas, A; Bibler, R. and Lanvin M. (2006) - Phylogeny of the genus Phaseolus (Leguminosae): a recent diversification in an ancient landscape. Systematic Botany, vol. 31, n. 4, p. 779-791. http://dx.doi.org/10.1600/036364406779695960 [ Links ]
Dita, M.A.; Rispail, N.; Prats, E.; Rubiales, D. and Singh, K.B. (2006) - Biotechnology approaches to overcome biotic and abiotic stress constraints in legumes. Euphytica, vol. 147, n. 1-2, p. 1–24. http://dx.doi.org/10.1007/s10681-006-6156-9 [ Links ]
Felsenstein, J. (1985) - Confidence limits on phylogenies: an approach using the bootstrap. Evolution, vol. 39, n. 4, p. 783–791. [ Links ]
Ferreira, E.M. and Marques, J.F. (1992) - Selection of Portuguese Rhizobium leguminosarum bv. trifolii strains for production of legume inoculants. Plant and Soil, vol. 147, n. 1, p. 151–158. http://dx.doi.org/10.1007/BF00009381 [ Links ]
Fox, G.E.; Wisotzkey, J.D. and Jurtshuk, P. (1992) - How close is close: 16S rRNA sequence identity may not be sufficient to guarantee species identity. International Journal of Systematic Bacteriology, vol. 42, n. 1, p. 166–170. http://dx.doi.org/10.1099/00207713-42-1-166 [ Links ]
Singh, S.P. (1999) - Integrated genetic improvement. In: Singh, S.P. (Ed.). Common bean improvement in the twenty-first century. (Developments in plant breeding, Vol. 7). Kluwer Academic Publishers. Dordrecht, p. 133-165. [ Links ]
Soussi, M.; Lluch C. and Ocana, A. (1999) - Comparative study of nitrogen fixation and carbon metabolism in two chick-pea (Cicer arietinum L.) cultivars under salt stress. Journal of Experimental Botany, vol. 50, n. 340, p. 1701–1708. http://dx.doi.org/10.1093/jxb/50.340.1701 [ Links ]
Squartini, A.; Struffi, P.; Döring, H.; Selenska-Pobell, S.; Tola, E.; Giacomini, A.; Vendramin, E.; Velázquez, E.; Mateos, P.F.; Martínez-Molina, E.; Dazzo, F.B.; Casella, S. and Nuti, M.P. (2002) - Rhizobium sullae sp. nov. (formerly 'Rhizobium hedysari'), the root-nodule microsymbiont of Hedysarum coronarium L. International Journal of Systematic and Evolutionary Microbiology, vol. 52, n. 4, p. 1267-1276. http://dx.doi.org/10.1099/ijs.0.01821-0 [ Links ]
Tamura, K.; Stecher, G.; Peterson, D.; Filipski, A. and Kumar, S. (2013) - MEGA6: molecular evolutionary genetics analysis version 6.0. Molecular Biology and Evolution, vol. 30, p. 2725–2729. http://dx.doi.org/10.1093/molbev/mst197 [ Links ]
Valverde, A.; Igual, J.M.; Peix, A.; Cervantes, E. and Velázquez, E. (2006) - Rhizobium lusitanum sp. nov. a bacterium that nodulates Phaseolus vulgaris. International Journal of Systematic and Evolutionary Microbiology, vol. 56, n. 11, p. 2631–2637. http://dx.doi.org/10.1099/ijs.0.64402-0 [ Links ]
Vincent, J.M. (1970) - A Manual for the Practical Study of Root-nodule Bacteria, IBP Handbook no. 15. Blackwell Scientific Publications. Oxford. [ Links ]
Wang, F.; Wang, E.T.; Wu, L.J.; Sui, X.H.; Li, Y. Jr and Chen, W.X. (2011) - Rhizobium vallis sp. nov., isolated from nodules of three leguminous species. International Journal of Systematic and Evolutionary Microbiology, vol. 6, n. 11, p. 2582–2588. http://dx.doi.org/10.1099/ijs.0.026484-0 [ Links ]
Weir, B.S. (2016) - The current taxonomy of rhizobia. NZ Rhizobia website. [ Links ]
Weisburg, W.G.; Barns, S.M.; Pelletier, D.A. and Lane, D.J. (1991). 16S ribosomal DNA amplification for phylogenetic study. Journal of Bacteriology, vol. 173, n. 2, p. 697–703. http://dx.doi.org/10.1128/jb.173.2.697-703.1991 [ Links ]
Zahran, H.H. (1999) - Rhizobium-legume symbiosis and nitrogen fixation under severe conditions and in an arid climate. Microbiology and Molecular Biology Reviews, vol. 63, n. 4, p. 968–989. [ Links ]
Zeven, A.C. (1999) -The traditional inexplicable replacement of seed and seed ware of landraces and cultivars: A review. Euphytica, vol. 110, n. 3, p. 181-191. http://dx.doi.org/10.1023/A:1003701529155 [ Links ]
Zurdo-Piñeiro, J.L.; Velázquez, E.; Lorite, M.J.; Brelles-Mariño, G.; Schröder, E.C.; Bedmar, E.J.; Mateos, P.F. and Martínez-Molina, E. (2004) - Identification of fast-growing rhizobia nodulating tropical legumes from Puerto Rico as Rhizobium gallicum and Rhizobium tropici. Systematic and Applied Microbiology, vol. 27, n. 4, p. 469–477. http://dx.doi.org/10.1078/0723202041438437 [ Links ]
Received/recebido: 2016.08.08
Received in revised form/recebido em versão revista: 2016.11.04
Accepted/aceite: 2016.11.24














