Serviços Personalizados
Journal
Artigo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Acessos
Acessos
Links relacionados
-
 Similares em
SciELO
Similares em
SciELO
Compartilhar
Revista de Ciências Agrárias
versão impressa ISSN 0871-018X
Rev. de Ciências Agrárias vol.41 no.spe Lisboa dez. 2018
https://doi.org/10.19084/RCA.17062
ARTIGO
Potato cyst nematodes’ routine analyses: morphology and/or PCR?
Análises de rotina para detecção de nemátodes de quisto da batateira: morfologia e/ou PCR?
Maria L. Inácio*, Maria J. Camacho, Leidy Rusinque, Filomena Nóbrega and Eugénia Andrade
Instituto Nacional de Investigação Agrária e Veterinária (INIAV), Unidade de Sistemas Agrários e Florestais e Sanidade Vegetal, Oeiras
(*E-mail: lurdes.inacio@iniav.pt)
ABSTRACT
Potato cyst nematodes (PCN), Globodera rostochiensis and G. pallida, are a serious threat to potato crops worldwide. Due to the risk these two species pose, a national survey was implemented starting in 2010 aiming to know the presence and distribution of these quarantine nematodes in Portugal. This fact has led to an increasing demand to accurately identify the PCN species present in the fields. Usually, the identification is based on morphology followed by molecular analysis. Considering there is at least one laboratory in the EU certified for morphological identification of PCN, the aim of this study was to look into the possibility of using only morphological diagnostic characters for routine purposes. Forty cysts of Globodera were used, for morphological and molecular analyses. Morphological analysis allowed the identification of 23 cysts as G. rostochiensis and 17 cysts as G. pallida and the molecular results revealed that 9 cysts were G. rostochiensis and 31 cysts were G. pallida. Thus, morphological and molecular analyses matched 65%, concluding that in our routine analyses only an initial morphological approach followed by molecular methods enables a reliable identification of PCN species. So, it is recommended to combine both methods. PCR can be ruled out only if the purpose is to detect Globodera nematodes, which is the case of the seed potato fields where the presence of round-cyst nematodes will interdict the seed production.
Keywords: cysts, Globodera, national survey, quarantine, molecular studies
RESUMO
Os nemátodes de quisto da batateira (NQB), Globodera rostochiensis e G. pallida, são uma ameaça para a cultura da batata a nível mundial. Devido ao risco que estas duas espécies representam, foi implementado um programa de prospeção nacional a partir de 2010 com o objectivo de detetar a presença e conhecer a distribuição destes nemátodes de quarentena em Portugal. Este facto intensificou a necessidade de determinar com precisão as espécies de NQB presentes nos campos de batata. Geralmente, esta identificação é baseada na avaliação das características morfológicas, confirmada de seguida pela análise molecular. Considerando que existe pelo menos um laboratório na União Europeia certificado para a identificação morfológica de NQB, o objetivo deste estudo foi investigar a possibilidade de identificar estas espécies apenas através das características morfológicas, em análises de rotina. Para as análises morfológicas e moleculares foram utilizados 40 quistos de Globodera sp.. A análise morfológica permitiu identificar 23 quistos como G. rostochiensis e 17 quistos como G. pallida, enquanto que os resultados moleculares para os mesmos quistos permitiram a identificação de 9 quistos como G. rostochiensis e 31 quistos como G. pallida. As análises morfológicas e moleculares apenas coincidiram em 65% dos casos. Concluiu-se que nas nossas análises de rotina apenas a utilização das duas abordagens permite a identificação precisa de espécies de NQB, sendo por isso recomendado combinar sempre os dois métodos. A análise molecular pode ser excluída somente se o objetivo visar a deteção de nemátodes do género Globodera, como é o caso dos campos de batata de semente, onde a presença de qualquer espécie destes nemátodes interditará a produção de semente.
Palavras-chave: Globodera, prospeção nacional, quarentena, quistos, estudos moleculares
INTRODUCTION
Potato cyst nematodes (PCN), Globodera pallida and G. rostochiensis, are a serious threat to potato crops worldwide, and both species have a quarantine status (EPPO, 2017a). In Portugal, G. rostochiensis, currently present in all potato producing regions of the country, was first identified in 1956 (Macara, 1963) and G. pallida later in 1988 (Santos and Fernandes, 1988), both in Trás-os-Montes region.
Before the national survey started, infestations detected in Portugal mainland were almost entirely due to G. rostochiensis (Santos & Fernandes, 1988; Santos et al., 1995; Martins et al., 1996, Conceição et al., 2003, Cunha et al., 2004, 2006; Cunha et al., 2012). The few pure G. pallida populations found in Portugal may suggest that it was introduced after G. rostochiensis or that there were only a few introductions; due to their low natural mobility, those populations could be restricted to the areas where they were introduced (Santos et al., 1995). In 2012, Cunha et al. reported that all Portuguese isolates of G. rostochiensis were ro1/ro4, and G. pallida isolates could have come from two different introductions (one for the isolate from Aveiro and another for the isolate from Vila Real). According to the authors, this lack of differentiation may be caused by PCN introductions that could have been made in different regions from the same origin or to the same region from different origins (Cunha & Conceição, 2012). Recently, the analysis of soils sampled in Portuguese potato fields revealed a spread of G. pallida, which contradicts the expectations because G. rostochiensis used to be the most widespread species in Portugal. The reasons for that reverse situation are still not fully understood (Camacho et al., 2017a,b).
As PCN pose a risk to potato crops in Portugal, the implementation of a national survey took place in 2010 intending to know the presence and distribution of those nematodes in potato cropping areas. Along with this came an increasing demand to accurately identify the PCN species present in Portuguese potato fields.
The distinction between the two species is essential in order to know their distribution in the national territory, re-evaluate the control measures so far implemented and to adopt more effective practices. Protocols to address quarantine and regulation requirements, speed, sensitivity, specificity and broad applicability features of the PCN identification are required.
The extraction of cysts from soil using elutriation principles remains essential especially when working with larger volumes of soil. Moreover, the morphological study of PCN is crucial as a previous step of the PCR tools preventing cross-reactivity with closely related species, namely Heterodera spp.
As PCN are very similar, robust methodologies are essential for their identification. Currently, the identification is based on the cysts and juveniles morphology, frequently followed by molecular analysis. However, there is no guarantee that the cysts used for the molecular studies are of the same species of the cysts used for morphological analysis when mixed populations are present. According to Camacho et al. (2017a), the species identification should be based on morphology/morphometry characterization followed by molecular analysis of the same cyst. However, there are accredited laboratories for morphological identification of both PCN species, such as the National Phytosanitary Laboratory - Nematology in Romania. Considering the importance of an accurate identification of PCN species, the aim of this research was to study the possibility of using only morphological diagnostic features for routine purposes.
MATERIAL & METHODS
Forty cysts were collected from Portuguese infested soils from the main producing potato regions, using Fenwick’s can method (Fenwick, 1940) and its modified version described by Oostenbrinck (1950). Globodera cysts from both species from Wageningen, The Netherlands, were used for morphological comparison and as controls for molecular analyses.
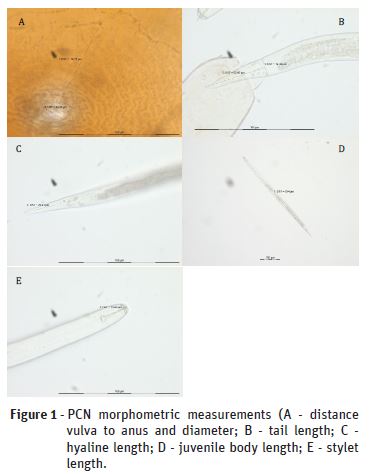
These 40 cysts, as well as second-stage juveniles (J2) recovered from each cyst, were used for morphological and molecular analyses. Morphological study of PCN was based on the observation of cyst colour and a combination of J2 and cyst features: body length, stylet length, tail and hyaline region, fenestra diameter, distance fenestra to anus, Granek’s ratio (distance from anus to the nearest edge of vulval basin divided by vulval basin diameter), and the number of cuticular ridges between vulva and anus, using an Olympus BX-41 bright field light microscope; pictures were taken with an Olympus DP10 digital camera. The observed characters of cysts and J2 were compared with those from the reference materials and descriptions in the literature (Marks & Brodie, 1998; EPPO, 2017b). Measurements were made using a ProgResSpeedXT – Jenoptik image software and dimensions are expressed in μm.
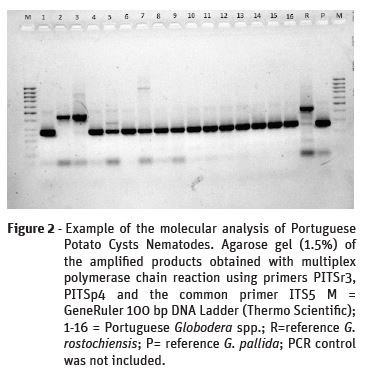
Half of each cyst was used in the morphological characterisation and the other half was used for DNA extraction, to assure one cyst went through both analyses. The internal transcribed spacer region (ITS) of the ribosomal DNA repeat unit was amplified by multiplex PCR. Multiplex PCR reactions were performed in a 25 μl final volume using the Promega GoTaq Flexi DNA Polymerase Kit (Promega, Madison, USA), containing 1 μl template DNA, 5 μl GoTaq Flexi PCR buffer, 1.5 mM MgCl2, 0.20 mM each dNTPs, 1.25 U GoTaq Flexi DNA Polymerase (Promega, Madison) and 0.4 μM of each primer in a Biometra TGradient thermocycler (Biometra, Gottingen, Germany). The set of primers for G. rostochiensis were: ITS5 (5’-GGA AGT AAA AGT CGT AAC AAG G-3’) and PITSr3 (5’-AGC GCA GAC ATG CCG CAA-3’) and for G. pallida were: ITS5 (5’-GGA AGT AAA AGT CGT AAC AAG G-3’) and PITSp4 (5´-ACA ACA GCA ATC GTC GAG-3’) (Bulman & Marshall, 1997). The amplification profile for ITS-rRNA consisted of an initial denaturation step of 94 °C for 2 min followed by 35 cycles of 94 °C for 30 s, 55 °C for 30 s and 72 °C for 30s and a final extension of 72 °C for 7 min (EPPO, 2017b). The amplified products were loaded onto a 1.5% agarose gel containing 0.5 μg.ml-1 ethidium bromide and 0.5×Tris-borate-EDTA (TBE) buffer and electrophoresed at 5 V/cm. Amplifications were visualized using the VersaDoc Gel Imaging System (Bio-Rad, USA). The expected length of the PCR products was 265 bp for G. pallida and 434 bp for G. rostochiensis. Possible contamination was checked by including negative controls (no DNA) in all amplifications.
RESULTS & DISCUSSION
Portuguese PCN populations present in potato fields are morphologically variable. The identification of Globodera species based on morphology is difficult, when cysts and J2 are individually analysed, due to the variability of the main morphological characters, with some features outside the expected ranges for G. pallida and/or G. rostochiensis described in the literature (Marks & Brodie, 1998; EPPO, 2017b), and the overlapping of the standard measures in the two species.
An initial approach to discriminate PCN species is the cyst colour. Females of G. rostochiensis change from white to yellow, and then to brown cysts (chromogenesis), while cysts of G. pallida go directly from white to brown. In the present study, all cysts presented only brown colour so that this character was not taken into account.
Using morphometric and morphologic parameters (Figure 1) 23 cysts were identified as G. rostochiensis and 17 cysts as G. pallida. In some cases, the characterisation of the knobs shape was ambiguous and the cyst wall pattern did not always allow species identification.
PCR amplified a product of the same size as G. rostochiensis in 9 samples and of the same size as G. pallida in 31 samples, meaning this molecular analysis only confirmed the morphological results for 26 cysts out of the 40 analysed (65% match) (Figure 2).
According to the obtained morphometric data, the morphological characterisation does not always match the molecular identification, due to the overlapping of the standard measures between the two PCN species (Figure 3; Table 1). The same result was observed in measurements performed in other studies around the world (Canada, United States, Serbia and Slovakia). In these studies the nematodes’ measurements were in agreement with the published standard values, but the amplitude between the individuals was outside the expected intervals (Yu et al., 2010; Skantar et al., 2011; Bačić et al., 2013; Douda et al., 2014).
PCN identification through morphometric studies is time consuming and requires a high level of expertise for species differentiation. It did not detect consistently low frequency mixtures on both species. An initial morphological approach followed by molecular methods enables the reliable identification of PCN species. This study concluded that it is highly recommended to combine both methods described and others if available. These results are supported by other researches (Bačić et al., 2013; Tirchi et al., 2016; Seesao et al., 2017).
Conclusion
The morphological study of PCN is crucial as a previous step of the PCR preventing cross-reactivity with closely related species, namely other Globodera species and some Heterodera spp.. In the present work, the limit of detection in PCR was just a half cyst with eggs and juveniles, since it worked with only half of each cyst used for DNA extraction (the other half being used in the morphological characterisation).
The morphological identification based on second stage-juveniles characteristics and cyst’s terminal zone has been successful, but always with some uncertainty, since the values of nematodes’ measurements overlap in many cases. Although this is useful, only highly specialised and trained technicians in morphological identification equipped with high resolution optical equipment can overcome this difficulty, becoming necessary to complement the morphological identification with molecular methods.
The multiplex PCR test with primers ITS5/PITSp4+PITSr3, allows the distinction between both species of PCN, and can be used in routine analysis.
It should be emphasized that to morphologically characterise a PCN population a minimum of 20 cysts and 20 juveniles from each cyst should be used. However, frequently not enough cysts are recovered from the soil samples and the detection of cyst nematodes must be mainly achieved through PCR analysis. Despite the development of molecular identification methods, the use of the morphological approach remains useful, hence being two complementary approaches.
References
Bačić, J.; Gerič Stare, B.; irca, S. & Urek, G. (2013) - Morphometric and molecular analysis of potato cyst nematodes from Serbia. Zbornik Predavanj in Referatov, n. 11, p. 369-372. [ Links ]
Bulman, S. & Marshall, J. (1997) - Differentiation of Australasian potato cyst nematode (PCN) populations using the polymerase chain reaction (PCR). New Zealand Journal of Crop and Horticultural Science, vol. 25, n. 2, p. 123-129. http://dx.doi.org/10.1080/01140671.1997.9513998 [ Links ]
Camacho, M.J.; Nóbrega, F.; Lima, A.; Mota, M. & Inácio, M.L. (2017a) - Morphological and molecular identification of the potato cyst nematodes (Globodera rostochiensis and G. pallida) in Portuguese potato fields. Nematology, vol. 19, n. 8, p. 883-889. http://dx.doi.org/10.1163/15685411-00003094 [ Links ]
Camacho, M.J.; Nóbrega, F.; Andrade, E.; Serra C. & Inácio M.L. (2017b) - Os nemátodes de quisto da batateira: ameaça crescente para os campos de batata em Portugal. V Jornadas Técnicas da Batata, COTHN, Escola Superior Agrária de Santarém. Abstracts: p. 19. [ Links ]
Conceição, I.; Santos, M.; Abrantes, I. & Santos, S. (2003) - Using RAPD markers to analyse genetic diversity in Portuguese potato cyst nematode populations. Nematology, vol. 5, n. 1, p. 137-143. https://doi.org/10.1163/156854102765216759 [ Links ]
Cunha, M.; Conceição, I.; Abrantes, I.; Evans, K. & Santos, S. (2004) - Characterisation of potato cyst nematode populations from Portugal. Nematology, vol. 6, n. 1, p. 55-58. https://doi.org/10.1163/156854104323072928 [ Links ]
Cunha, M.; Conceição, I.; Abrantes, I. & Santos, S. (2006) - Assessment of the use of high-performance capillary gel electrophoresis to differentiate isolates of Globodera spp. Nematology, vol. 8, n. 1, p. 139-146. https://doi.org/10.1163/156854106776179980 [ Links ]
Cunha, M.; Cunha, M.; Conceição, I.; Abrantes, I. & Santos, S. (2012) - Virulence assessment of Portuguese isolates of potato cyst nematodes (Globodera spp.). Phytopathologia Mediterranea, vol. 51, p. 51-68. http://dx.doi.org/10.14601/Phytopathol_Mediterr-9517 [ Links ]
Douda, O.; Zouhar, M.; Renco, M. & Marek, M. (2014) - Molecular and morphological exploration of a mixed population of two potato-parasiting nematode species, Globodera rostochiensis and G. pallida. Helminthologia, vol. 51, n. 1, p. 3–6. http://dx.doi.org/10.2478/s11687-014-0201-3 [ Links ]
EPPO (2017a) - EPPO Alert List. [cit. 2016-09-13]. <https://www.eppo.int/QUARANTINE/Alert_List/alert_list. [ Links ]htm>.
EPPO (2017b) - Globodera rostochiensis and Globodera pallida PM 7/40 (4). European and Mediterranean Plant Protection Organization. EPPO Bulletin, vol. 47, n. 2, p. 174–197. http://dx.doi.org/10.1111/epp.12391 [ Links ]
Fenwick, D. (1940) - Methods for the recovery and counting of cysts of Heterodera schachtii from soil. Journal of Helminthology, vol. 18, n. 4, p. 155-172. https://doi.org/10.1017/S0022149X00031485 [ Links ]
Macara, A. (1963) – Aspectos sobre a importância dos nemátodos de interesse agrícola em Portugal e no ultramar português. Agros, n. 46, p. 367-384. [ Links ]
Marks, B. & Brodie, B. (1998) - Potato cyst nematodes: biology, distribution and control. Wallingford, UK, CAB International., 408 p. [ Links ]
Martins, F.; Santos, S.; Abreu, C.; Abrantes, I. & Evans, K. (1996) - Potato Cyst nematodes – The situation in the North East Part of Portugal. 13th Triennial Conference of EAPR, Veldhoven, 14-19 Julho, Abstracts, pp. 369-370. [ Links ]
Oostenbrink, M. (1950) - Het aardappelaaltje (H. rostochiensis Wollenweber) een gevarlijke parasiet voor de eenzijdige aardappelcultuur. Versl. PlZiekt. Diensf. Wageningen, 115: 230. [ Links ]
Santos, S. & Fernandes, M. (1988) - The occurrence of Globodera rostochiensis and G. pallida in Portugal. Nematologia Mediterranea, vol. 16, n. 1, p. 145. [ Links ]
Santos, S.; Evans, K.; Abreu, C.; Martins, F. & Abrantes, I. (1995) - A review of potato cyst nematodes in Portugal. Nematologia Mediterranea, vol. 23, p. 35-42. [ Links ]
Seesao, Y.; Gay, M.; Merlin, S.; Viscogliosi, E.; Aliouat-Denis, C. & Audebet, C. (2017) - A review of methods for nematode identification. Journal of Microbiological Methods, 138, p. 37-49. http://dx.doi.org/10.1016/j.mimet.2016.05.030 [ Links ]
Skantar, A.; Handoo, Z.; Zasada, I.; Inghram, R.; Carta, L. & Chitwood, D. (2011) - Morphological and molecular characterization of Globodera populations from Oregon and Idaho. Phytopathology, vol. 101, n. 4, p. 480-491. http://dx.doi.org/10.1094/PHYTO-01-10-0010 [ Links ]
Tirchi, N.; Troccoli, A.; Fanelli, E.; Mokabli, A.; Mouhouche, F. & De Luca, F. (2016) - Morphological and molecular identification of potato and cereal cyst nematode isolates from Algeria and their phylogenetic relationships with other populations from distant their geographical areas. European Journal of Plant Pathology, vol. 146, n. 4, p. 861-880. https://doi.org/10.1007/s10658-016-0965-z [ Links ]
Yu, Q.; Ye, W.; Sum, F. & Miller, S. (2010) - Characterization of Globodera rostochiensis (Tylenchida: Heteroderidae) associated with potato in Quebec, Canada. Canadian Journal of Plant Pathology, vol. 32, n. 2, p. 264–271. http://dx.doi.org/10.1080/07060661003740322 [ Links ]
Received/recebido: 2017.12.28
Accepted/aceite: 2018.06.19














