INTRODUCTION
According to FAO, over than 3x106 t of olive oil (OO) were globally produced in 2019, of which almost 2/3 were generated in European Union, namely on Mediterranean countries such as Spain, Greece, Italy and Portugal. In the specific case of Portugal (1x105 t of OO; FAO, 2019), this industry contributed with 700 M€ for gross national income and is centred on inland areas where Trás-os-Montes e Alto Douro (TM) region is the second largest producer (15% of country’s production). Though significative to TM’s economy, industrial tissue is based on agri-food sector and characterized to be small-scale agriculture, without water drip irrigation and hardly automatized. After processed on olive mills, olive pomace (OP) it is generated that is transported to an oil extractor unity, where physical and chemical-based fat extractions are performed, for further refining, in order to be sold as edible oil. Nowadays, almost all of TM’s olive pomace is processed in a single outdated extractor factory (pilot area, PA). As result of the physical OO extraction (55-75% m:m), wastewaters are produced, olive mill wastewaters (OMWW) which are covered on Portuguese legislation and therefore, requires residues management. Nevertheless, given the inefficiency of conventional biological treatments, OMWW are stored in high volume and open-air lagoons, subjected to decantation, photolysis and evaporation processes. In fact, the existence of connective channels to rivers to pump water from near streams for machinery refrigeration, are typical in this kind of industry and widely used to drain excessive lagoons’ content in case of intense precipitation events or labour periods (olive harvest). Although it is known that olive pomace is phytotoxic due to its high organic load, scarce information is yet available on the impacts of OMWW on the ecological integrity of surrounding ecosystems, highlighting the importance of further research on this topic. The lack of information on this topic, arises the need to understand the real extension of ecological impacts of oleic industry, following the Water Directive Framework guidelines. Additionally, the research presents an alternative approach of this normative, in what concerns the sediments’ role within the biogeochemical balance and long-term “witness” of environmental stressors.
METHODOLOGY
In order to achieve the proposed aims, eight sites (five upstream PA, three downstream PA) along Tua River (Douro basin) and tributaries were seasonally monitored for a year (2021-2022), following standardized procedures defined by the Water Framework Directive. Environmental data was registered, considering water physicochemical (common metrics and specific pollutants), hydromorphological (River Habitat Survey) and biological (fish and invertebrates) factors.
Sediments were also assessed in percentage of organic carbon (SOC), ashes according to Almendros et al. (2018) and nutrients (NPK) content. Elemental analysis (Mg, Ca, N, P, K, Mg) were determined by Inductively coupled plasma mass spectrometry (ICP-MS).
Moreover, to understand the toxic role of OMWW, Daphnia magna and Unio delphinus were chosen as bioindicator organisms. Daphnia magna individuals were subjected to acute toxicity tests (DAPHTOXKIT F, 24 h Immobilisation test), according to ISO 6341 (BS EN ISO 6341, 2012) and OECD Guideline 202 (OECD, 2004), where lethal doses to 50% of the population was determined (24 h EC50). Individuals were exposed to seven different treatments, a control condition (CTL), and to a range of five concentrations of OMWW (2.00; 4.00; 8.00; 16.0; 32.0) (mg/L), as shown on Figure 1. Every treatment was constituted by four replicates containing five organisms each. Three consistent assays were performed using the same toxicant matrix (OMWW) collected on PA (February 2022).
In order to assess the impact in other trophic levels, a chronic exposure to OMWW was performed using native species of U. delphinus. Freshwater mussels and water were collected in Rabaçal River (41°33'52.10"N; 7°15'31.27"W), tributary of Tua River, and classified as good within ecological quality indexes. Organisms were placed in individual flasks containing sterilised sand and freshwater (1:3), with homogeneous aeration for 21 days (seven days acclimation + 14 days exposure). Individuals were exposed to three different treatments, a control condition (CTL), and two concentrations chosen considering the lowest and highest levels of oxidability/ chemical oxygen demand (mg/L) recorded on Tua River and PA’s discharge channel merging (8.77 mg/L and 21.31mg/L). As represented in Figure 2, every treatment was constituted by six replicates of one organism each. Toxicant used for contaminant solution was OMWW collected on PA (February 2022).
Ecotoxicological biomarkers were performed on gills and digestive glands to perform Potential metabolic assay (PMA), Lipid peroxidation (LPO) and enzymatic responses to potential oxidative stress occurrence (catalase CAT, superoxide dismutase SOD), as described in Costa et al. (2020).
Data analysis was performed by PERMANOVA + add-on in PRIMER v7 using multivariate analysis of variance. Statistical differences, represented using lower case letters, considered the following null hypotheses: i) no significant differences existed between contaminated (CTL) and non-contaminated organisms (8.77 mg/L and 21.31 mg/L); ii) no significant differences existed between different OMWW concentrations (8.77 mg/L and 21.31 mg/L).
RESULTS AND DISCUSSION
When comparing the obtained results, sites located downstream of oil extraction industries showed a significant decrease on the water quality (higher values of oxygen demand and particularly of phenols). Ecological metrics (taxonomic richness, EPT taxa), and multivariate analyses detected the ecological impact of the pollutant effluent. Moreover, it was noticed that all three sampling sites downstream the extractor (sampling site PB6) were classified as Bad, Poor or Reasonable, for biological quality (according to Shannon-Wiener H’, invertebrate IptIN- Table 1- and fish F-IBIP indexes- Table 2) indicating diffuse contamination along water streams. Ecological quality was assessed using normative criteria defined by the National and European Water Authorities (INAG, 2008, 2009; EC, 2011, 2013; EPA, 2012; INAG and AFN, 2012).
Table 1 Classification of biological quality results for the Northern Portuguese invertebrates (IPtIN) index. Three samplings (Summer, Autumn and Winter) were performed per each site (PB1- Miradeses; PB2- Eixes (upstream); PB3- Eixes (downstream); PB4- Chelas; PB5- Upstream Extractor; PB6- Downstream Extractor; PB7- Frechas; PB8- Vilarinho das Azenhas). Results are expressed as Excellent, Good, Reasonable, Poor or Bad, according to INAG (2008)
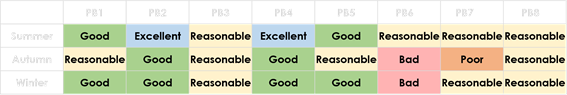
Table 2 Classification of biological quality results for the Fish Index of Biotic Integrity (F-IBIP). Two samplings (Autumn and Winter) were performed per each site (PB1- Miradeses; PB2- Eixes (upstream); PB3- Eixes (downstream); PB4- Chelas; PB5- Upstream Extractor; PB6- Downstream Extractor; PB7- Frechas; PB8- Vilarinho das Azenhas). Results are expressed as Excellent, Good, Reasonable, Poor or Bad, according to INAG & AFN (2012)

The continuous discharging, despite the far-reaching on aquatic compartment, it is very targeted to the static sediments surrounding the industrial discharge channel. In fact, higher organic matter (ashes) and carbon levels were recorded for sampling sites closer to the industry (Figure 3). Even though decreasing trend was observed, both results were much higher when comparing with the control sampling site (upstream the extractor) (Figure 3). The effects are indeed a consequence of the oil extraction processes considering that sediments’ macronutrients concentration (P and K) are remarkably higher for sampling sites closer to the industry (Table 3). Olives are, besides water, greatly constituted by K but also P, suggesting that discharges present a high content of these elements, forming more stable forms and thus, enriching river sediments (Maestri et al., 2019).
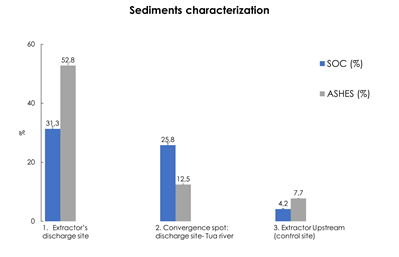
Figure 3 Organic content: percentage of carbon (SOC) and organic matter (ashes) of sediments sampled on (1) Extractor discharge site, the (2) convergence point between industrial discharge site and Tua river and a (3) control site (upstream the Extractor).
Table 3 Macronutrients profile of sediments sampled on: Extractor discharge site (1), the convergence point between industrial discharge site and Tua river (2) and a control site (upstream the Extractor, 3)
| P mg/kg | Mg mg/kg | Ca mg/kg | K mg/kg | |
| 1. Extractor discharge site | 732.4 | 472.1 | 746.3 | 3689.7 |
| 2. Convergence spot: discharge site - Tua river | 283.3 | 171.7 | 355.0 | 553.8 |
| 3. Extractor upstream (control site) | 103.0 | 287.7 | 964.8 | 172.4 |
Additionally, the conducted 24 h EC50 assays, revealed that the minimum doses required to cause the death, of at least 50% of the tested population (D. magna), is equivalent to 8.88 mg/L of OMWW (Figure 4). Results are in accordance with similar research, using other crustaceans’ species to perform 24 h EC50 assays (Danellakis et al., 2011; Babić et al., 2019; Domingues et al., 2020). Though slightly, 24 h EC50 levels were lower, when compared with the mentioned studies which may be explained by the oil extraction method (authors used OMWW obtained by three-phase system and discontinuous traditional process).
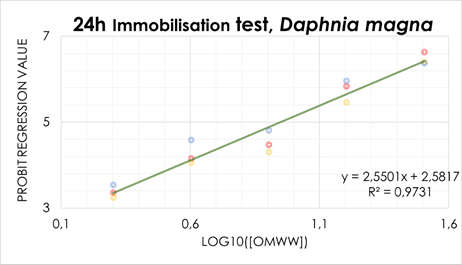
Figure 4 Acute toxicity (24 h EC50) for D. magna exposed to OMWW ([0.00, 2.00, 4.00, 8.00, 16.0, 32.0] mg/L). Results consider data obtained from three consistent assays and EC50 is obtained by x value on y=5 (expressed in percentage).
Chronic exposures to OMWW revealed the toxic role of OMWW to non-target organisms (such as filter freshwater mussels) when available on aquatic ecosystems. Olive mill wastewaters induced cellular membranes’ disruption, confirmed by the higher content of thiobarbituric acid reactive substances (TBARs), suggesting lipid peroxidation occurrence (Figure 5A). In fact, the increase of reactive oxygen species, triggered the antioxidant enzymes (SOD) specially on Gills (Figure 5C). This observation is justified by the fact this species is a filter-feeding organism, therefore, gills are the primary barrier to avoid OMWW uptake. Though detoxification mechanisms were activated, oxidative stress was not prevented, considering the results on TBARs (Figure 5A), reason why, neuronal system activity was inhibited, as seen on AChE results (Figure 5D). Though ineffective in oxidative damage prevention, OMWW presence induced the activation of primary detoxification machinery, as confirmed in other studies performed with both marine and freshwater bivalves using the same toxicant (Danellakis et al., 2011; Babić et al., 2019; Domingues et al., 2020).
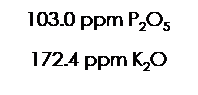
Figure 5 Results of Thiobarbituric acid reactive substances (TBARS) as byproduct of lipid peroxidation (A); Superoxide dismutase activities (SOD) on Gills (B) and Digestive Glands (C); Acetylcholinesterase (AChE) activity on Haemolimph (D) for Unio delphinus exposed for 14 days to OMWW ([0.0; 8.77; 21.31] mg/L). Results are expressed by obtained mean values (n=6) + standard errors.
Reported data clearly pinpoints the need to develop and implement alternatives and sustainable solutions to treat OMWW, reducing its toxicity, thus, promoting ecological conservation, not only for endangered species, such as Unio delphinus but also fostering soils/sediments health and biotic balance. As future perspectives, it is intended to develop a remediation procedure for OMWW, using carbonaceous materials and re-evaluate the toxicity of treated OMWW. Activated carbon is not only a very promising strategy for water remediation but also as soil amender. The correct application on soils may also be an important step to restore soils health and consequentially, ecosystems conservation.
CONCLUSIONS
The present research shows that despite the fact that the contaminant (OMWW) is discharged into water, this brings effects not only to inhabiting biota but also to sediments bioaccumulation. The evidences presented suggest a multi-level and diffuse source of contamination, when considering the expected water quality decay and consequential degradation on downstream extractor sampling sites. Nevertheless, data also emphasizes the continuous discharging of this contaminant into the environment. The results obtained from the sediments’ characterization and considering the timeframe needed for elemental stabilization on non-mobile phase, the research suggests that the surrounding ecosystems are not exposed only when severe precipitation events lead to leaking, but to constant and persistent exposure to OMWW. Thus, it became so important to understand not only acute but chronic effects of this toxic upon aquatic biota. In fact, the mussels showed a clear antioxidant mechanisms activation as result of higher levels of reactive oxidative species, when exposed to environmentally relevant concentrations (quantified while monitoring sampling sites).
















