Serviços Personalizados
Journal
Artigo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Acessos
Acessos
Links relacionados
-
 Similares em
SciELO
Similares em
SciELO
Compartilhar
Arquivos de Medicina
versão On-line ISSN 2183-2447
Arq Med v.23 n.6 Porto nov. 2009
Amikacin Induces Renal Morphohistological Alterations in Wistar Rats
Vanessa Barboza de Sousa*, Ilana Jozi Pereira Dutra†, Hévio Freitas de Lucena†, Hilkéia Carla de Souza Medeiros†, Gerlane Coelho Bernardo Guerra‡, Raimundo F. de Araújo Júnior†, Aurigena A. Araújo‡, Maria do Socorro Costa Feitosa Alves*
*Universidade Federal do Rio Grande do Norte, Brasil; Departementos de †Morfologia e ‡Farmacologia,Universidade Federal do Rio Grande do Norte, Brasil
The aim of this experimental study was to investigate the morphohistological effects of amikacin in rats. Male Wistar rats aged 8-12 weeks and weighing 200-250g were placed in a temperature (23±2°C) controlled room in which a 12/12h light: dark cycle was maintained. Twenty-five rats were randomly assigned to five experimental groups of five animals each that received daily treatment with 15 mg/kg of amikacin intraperitoneally. The control group was treated with 0.9% saline for 28 days. Groups I, II, III and IV were injected intaperitoneally with amikacin for 7, 14, 21 and 28 days, respectively. Renal tissues slides were stained with PAS reagent and examined light microscopy. The morphology of the PAS stained structures of the renal glomeruli was evaluated in the different experimental groups using scores. The renal morphohistology and the morphologic analysis of PAS staining were analysed statistically with the Kruskal-Wallis Test and Dunn’s Multiple Comparisons Test (5% statistical significance). The results show microscopically that kidneys in the groups treated with amikacin for 3 and 4 weeks showed marked cellular necrosis (p< 0.0001), tubular obstruction, and basal membrane interruption. This result clearly shows that amikacin administration induces renal morphohistological alterations.
Key-words: amikacin; aminoglycosides; nephrotoxicity; tubular injury.
Alterações Morfohistológicas Renais Induzidas pela Amicacina em Ratos Wistar
O objetivo deste estudo experimental foi investigar as alterações morfohistológicas da amicacina em ratos. Ratos Wistar machos com idade de 8-12 meses e peso 200-250 g foram mantidos em temperatura (23±2°C) ambiente controlada com ciclos claro/escuro 12/12h. 25 ratos foram distribuídos em 5 grupos experimentais, cada grupo com 5 animais. 4 grupos receberam uma dose diária de amicacina de 15 mg/Kg pela via intraperitoneal. O grupo-controle foi tratado com 0,9% solução salina por 28 dias. Aos grupos I, II, III e IV foram administrados pela via intraperitoneal amicacina por 7, 14, 21 e 28 dias, respectivamente. Lâminas de tecido renal foram coradas com o reagente PAS e examinadas pela microscopia de luz. A morfologia das estruturas coradas pelo PAS do glomérulo renal foi avaliada nos diferentes grupos experimentais através de scores aplicando-se análise estatística de Kruskal-Wallis e Teste de comparação múltipla de Dunn’s com nível de significância de 5%. Os resultados da microscopia mostraram que os rins dos grupos tratados com amicacina por 3 a 4 semanas apresentaram necrose celular (p< 0,0001), obstrução tubular e interrupção da membrana basal. Estes resultados mostraram que a administração crônica da amicacina induz alterações morfohistológicas no tecido renal.
Palavras-chave: amicacina; aminoglicosídeos; nefrotoxicidade; injúria tubular.
INTRODUCTION
Due to its prominent role in major excretory pathways, the kidney is particularly sensitive to toxicity from antimicrobials drugs, especially the aminoglycosides. Storage of these drugs in the renal cortex with their effect on renal cells has consequences for renal function, and when reabsorbed by renal tubules induces nephrotoxicity (1,2).
Aminoglycosides are natural or semi-synthetic antibiotics with a heterocyclic structure formed by two or more aminosugarslinked by glycoside bonds to an aminocyclitol ring.Aminoglycoside antibiotics are clinically used for treatment of Gram-positive and Gram-negative infections by inhibiting bacterial protein synthesis. However, they show clinical side effects with long-term administration, such as nephrotoxicity and ototoxicity (3). It is considered that aminoglycoside nephrotoxicityis caused by accumulation of aminoglycosides in proximal tubular cells, therefore cellular membrane structure and some transport functions would be affected (4).
Amikacin has become an antibiotic used especially for the early treatment of bacterial Gram-negative infections in hospitals, since bacterial resistance to gentamicin and tobramycin has become a significant problem. Adverse effects are ototoxicity and nephrotoxicity (5). Thus, the purpose of the present article is to investigate morpho-histological kidney alterations due to chronic treatment with amikacin, by means of PAS staining.
MATERIALS AND METHODS
Male Wistar rats 8-12 weeks old, weighing 200-250g, were placed in a temperature (23±2°C) controlled room in which a 12/12h light: dark cycle was maintained. Twenty-five rats were randomly assigned to five experimental groups of five animals each that received daily amikacin treatment, 15mg/kg, intraperitoneally. The control group was treated with 0.9% saline for 28 days. Groups I, II, III and IV were injected intaperitoneally with amikacin for 7, 14, 21 and 28 days, respectively. After treatment, the animals in all groups were sacrificed and the kidneys were quickly removed, decapsulated and divided longitudinally into two equally sized pieces. One piece was placed in formaldehyde solution to be embedded in paraffin and 5 µm-thick sections were cut for histological examination by light microscopy.
Histological evaluation
Histochemistry
Renal tissues slides were stained with PAS reagent and examined under light microscopy. The PAS reaction stains carbohydrates and carbohydrate-rich macromolecules. It is used to visualize the basal membrane that underlies epithelia. Three coded slides from each kidney were examined in a blinded manner.
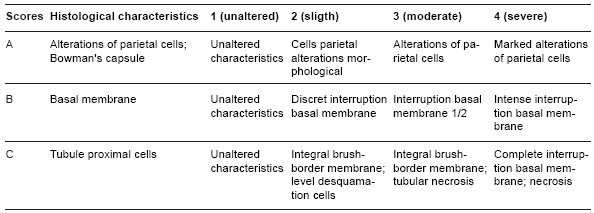
Qualitative and quantitative evaluation of morphology
The morphology of the PAS-stained structures of the renal glomeruli was evaluated in different experimental groups using scores (Table 1) to classify the histological alterations with respect to the state of the basal membrane of the glomerulari capilaris, and the continuity of the basal membrane of the renal glomeruli and parietal cells. The proximal and distal tubules were evaluated for the morphology of the revestment cells (necrosis). Kidney sample sections were analyzed with a microscope (with camera) connected to a microcomputer.
Table 1 - Scores of renal histological alterations in Wistar rats.
Statistical analysis
The renal morphohistology and the morphologic analysis for PAS staining were analysed statistically with the Kruskal-Wallis Test and Dunn’s Multiple Comparisons Test (5% statistical significance).
RESULTS
Figure 1 shows the results of renal morphohistological alterations found after histochemical PAS staining to the different groups treated with amikacin. Scores to histochemical alterations were classified according to table 1.

Fig. 1 -Morphological comparisons between control group and all treated amikacin groups (PAS). Group 01 vs. Control Group (p>0.05); Group 02 vs. Control Group (p<0.001); Group 03 vs. Control Group (p<0.001); Group 04 vs. Control Group (p<0.001); Group 01 vs. Group 02 (p>0.01); Group 01 vs. Group 03 (p<0.001); Group 01 vs. Group 04 (p<0.001); Group 02 vs. Group 03 (p>0.05); Group 02 vs. Group 04 (p<0.001); Group 03 vs. Group 04 (p>0.05).
Histological examination of the kidneys from animals in the control group revealed, as expected, entirely normal histological features, as illustrated in figure 1. However, there was tubular necrosis in kidneys from animals in the amikacin treated group, initially in group 2 and particularly in group 4, as illustrated in figures 2 and 3, respectively.
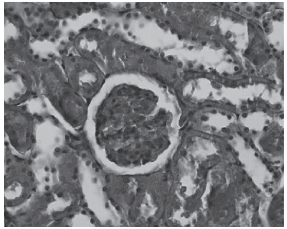
Fig. 2 - Control Groups (PAS).
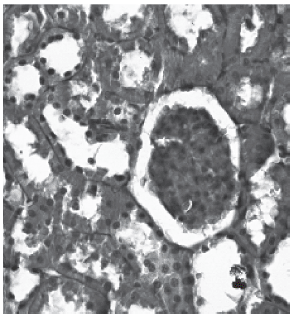
Fig. 3 - Group 2 (PAS).
The lumen of the tubules was filled with degenerate and desquamated epithelial desquamated epithelial cells. Furthermore interruptions in the basal membrane are present.
DISCUSSION
Most of the aminoglycoside injected into the body is excreted into the urine without being metabolized. However, the remainder of the injected dose accumulates selectively and abundantly in the renal cortex. Aminoglycoside taken up by renal proximal tubular cells remains there for an extended period, leading to renal damage, such as structural changes and functional impairment of the plasma membrane, mitochondria and lysosome (6). Consequently, there has been great interest in the mechanism by which aminoglycosides are absorbed into the renal proximal tubular cells across the brush-border membrane. So far, results have indicated that aminoglycosides are taken up by receptor-mediated endocytosis following the binding of aminoglycosides to the brush-border membrane. There is also evidence that aminoglycosides directly disturb glomerular physiology, reducing SNGFR by lowering both the Kf and the afferent glomerular arteriolar output (7).
Perazella, (8) showed experimental data suggesting that both vascular, glomerular and tubular targets are involved in drug-induced nephrotoxicity, as a result of mechanisms that disrupt normal cellular structures and functions (mitochondria, membraneintegrity, etc.),induce renal injury through intratubular obstruction (crystal deposition), and promote cellular swelling and tubular luminal occlusion (through osmotic effects). These effects were observed in our work, as seen in group 2 treated with amikacin, where we observed discrete basal membrane interruptions.
Studies of nephrotoxicity involving animal models have found that once-daily administration of gentamicin or to bramycin was significantly less toxic than more frequent (i.e., twice or three times daily) dosages or continuous infusion (9-12).Animal studies have further elucidated the probable mechanism involved in aminoglycoside nephrotoxicity, which involves an absorptive influx at proximal convoluted tubule cells and is mediated by a low-affinity, high-capacity mechanism that can be saturable, linear, or a mixture of the two (13-14). The exact mechanism is not known, but speculation has led primarily to the following hypotheses: a lysosomal mechanism, with aminoglycosides causing an accumulation of myeloid bodies within lysosomes and thereby inhibiting lysosomal phospholipases with a subsequent decrease in sphingomyelinase activity, or an extralysosomal mechanism, with inhibition of Na, K -ATPase and phospholipase C, leading to unopposed angiotensin II activity, or a combination of these mechanisms. In these experiments amikacin was administered once-daily, therefore significantly less toxic effects were observed than when traditional dosing regimens are applied.
Aminoglycoside-induced nephrotoxicity is characterized by tubular necrosis, basal membrane disruption, mesangial cell contraction, proliferation and apoptosis, indicated by a decrease in glomerular filtration and alteration in intraglomerular dynamics (15).
Our results clearly show that adverse effects observed in rats induced by chronic treatment with amikacin occur mainly in group 4, where morphological alterations were marked. Our results suggesting that amikacin is a potent factor are consistent with the clinical literature regarding nephrotoxicity. Our morphohistologicalfindings include cellular necrosis, tubular obstruction, and basal membrane interruption.
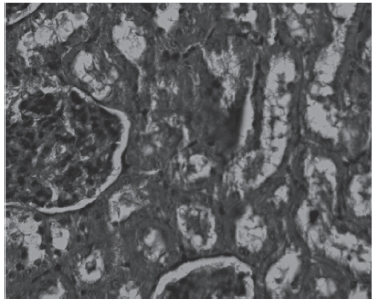
Fig. 4 - Group 4 (PAS).
REFERENCES
1 -Souza VB, Oliveira RFL, Lucena HF, Antunes AA, Guerra GCB, Freitas ML et al. Gentamicin induces renal morpho-pathology in Wistar rats. Int J of Morphol 2009;27:59-63.
2 -Souza VB, Oliveira RFL, Antunes AA, Araújo Júnior RF. Alterações renais por aminoglicosídeos. Arquimed 2008; 22:131-5.
[ Links ]3 -Kaloyanides GJ, Pastoriza-Munoz E.Aminoglycoside nephrotoxicity. Kidney Int 1980;18:571-82.
4 -Inui K, Saito H, Hori R. The use of kidney epithelial cell line (LLC-PK1) to study aminoglycoside nephrotoxicity. Dev Toxicol Environ Sci 1986;14:217-26.
5 -Chambers HF. In: Goodman e Gilman (Org.): As bases farmacológicas da terapêutica, editores. 10ed. Rio de Janeiro: McGraw-Hill, pp.913-24, 2003.
6 -Mingeot-Leclerq MP, Tulkens PM.Aminoglycosides: nephrotoxicity, Antimicrob. Agents Chemother 1999;43:1003-12.
7 -Baylis C, Renke HR, Brenner BM. Mechanisms of the defect in glomerular ultrafiltration associated with gentamicin administration. Kidney Int 1977;12:344-53.
8 -Perazella MA, 2003. Drug-induced renal failure: update on new medications and uinique mechanisms of nephrotoxicity. Am J Med Sci 2003;325:349-62.
9 -Herscovici L, Grise G, Thauyin C, Lemeland JF, Fillastre JP. Efficacy and safety of once daily versus intermittent dosing of tobramycin in rabbits with acute pyelonephritis. Scandinavian J of Inf Dis 1988;20:205-12.
10 -Frame PT, Phair JP, Watanakunakorn C, Bannister TWP. Pharmacologic factors associated with gentamicin nephrotoxicity in rabbits. J of Inf Dis 1977;135:952-6.
11 -Bennett WM, Plamp CE, Gilbert DN, Parker RA, Porter GA. The influence of dosage régimen on experimental gentamicin nephrotoxicity: dissociation of peak serumlevels from renal failure. J of infect Dis 1979;140:576-80.
12 -Tran Ba Huy P, Bernard P, Schacht J. Kinetics of gentamicin uptake and release in the rat. Comparison of inner ear tissues and fluids with other organs. J of Clinic Investig 1986;77:1492-500.
13 - Kaloyanides GJ. Renal pharmacology of aminoglycoside antibiotics. Cont to Nephrol 1984;42:148-67.
14 -Bennett WM, Wood CA, Houghton DC, Gilbert DN. Modification of experimental aminoglycoside nephrotoxicity. American J of Kidney Dis 1986;8:292-6.
15 -Martínez-Salgado C, Henández-López FJ, Novoa-López JM. Glomerular nephrotoxicity of aminoglycosides. Toxicol and applied pharmacol 2007;223:86-98.
Correspondência:
Dr.ª Aurigena A. A. Ferreira
Departamento de Farmacologia
Universidade Federal Rio Grande do Norte
Centro de Biociências
Campus Universitário/UFRGN
CEP: 59072-970
Natal/RN, Brazil
e-mail: aurigena@ufrnet.br














