Serviços Personalizados
Journal
Artigo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Acessos
Acessos
Links relacionados
-
 Similares em
SciELO
Similares em
SciELO
Compartilhar
Portuguese Journal of Nephrology & Hypertension
versão impressa ISSN 0872-0169
Port J Nephrol Hypert vol.26 no.4 Lisboa out. 2012
Chronic Q fever in two haemodialysis patients
Ricardo Vizinho1, Margarida Branco2, Tiago Amaral1, Célia Gil1, Margarida Gonçalves1, Ilídio Rodrigues1, Acácio P Negrão1, José D. Barata1
1 Hospital Santa Cruz, Department of Nephrology. Carnaxide, Portugal.
2 Hospital Egas Moniz, Department of Internal Medicine. Lisbon, Portugal.
ABSTRACT
Q fever is a zoonotic disease caused by the highly infectious agent Coxiella burnetii which may manifest as a fairly common acute benign disease, or a much rarer and potentially lethal chronic condition. To our knowledge, chronic Q fever has previously only been described in two dialysis patients. We report a further two cases of haemodialysis patients with chronic Q fever and hypothesise as to its possible underdiagnosis in this population. Both patients had a prosthetic heart valve and although no vegetations were found by transoesophagic echocardiography, they met the Duke criteria for possible infective endocarditis. One patient, diagnosed about 11 months after the initial clinical manifestations, died, while the other, started on doxycycline and chloroquine about 3 months after the initial clinical manifestations, survived. We underline some key points that should lead to suspicion of chronic Q fever, namely the combination of a fluctuant febrile syndrome of unknown origin, anaemia and prior heart valve disease, especially in an immunocompromised patient.
Key-Words: Anaemia; chronic Q fever; haemodialysis; heart valve.
INTRODUCTION
Q fever, a zoonotic infection, was first described by E. H. Derrick in 1937. The exact prevalence of Q fever is not known, but it has been seen worldwide1-3.
So far, to our knowledge, chronic Q fever has only been described in two dialysis patients4. We present two other haemodialysed patients with serological diagnosis of chronic Q fever. We hypothesise as to its possible underdiagnosis in this specific population and highlight some key points that should lead to a higher index of suspicion, namely a history of febrile syndrome of undetermined origin, anaemia and prior valve disease. We also stress the potential for cure, if treated, of an otherwise deadly disease.
CASE REPORTS
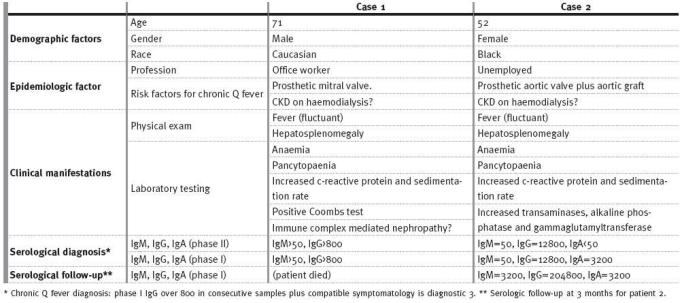
Both patients were admitted to the Nephrology ward of Hospital Santa Cruz. Some demographic, epidemiologic and clinical aspects of the cases are summarised in Table I.
Demographic, epidemiologic and clinical manifestations of the reported cases.
Case 1
This was a 71 -year -old Caucasian male retired office worker with controlled hypertension; longstanding type 2 diabetes mellitus, with adequate metabolic control; chronic kidney disease (CKD), with 24h proteinuria of 500 mg, assumed to be secondary to diabetic nephropathy; a mechanical prosthetic aortic valve placed in 1975 for rheumatic valve disease; coronary artery disease with percutaneous angioplasty and stenting of the left anterior descending artery in 1995; gastritis; intestinal angiodysplasia and several episodes of gastrointestinal bleeding. His regular medication included epoetin beta, iron, warfarin, folic acid, B-complex vitamins, carvedilol, perindopril, atorvastatin and pantoprazole.
The patient had four hospital admissions with normocytic normochromic anaemia and fluctuant fever of unknown aetiology between August 2008 and April 2009. The anaemia did not respond to iron and increasing doses of epoetin, and frequent red cell transfusions were required. Reticulocyte count was 2.5 to 3.5%, lactate dehydrogenase was slightly elevated (700 to 800 U/L) and haptoglobin low (<8g/L). The presence of increased inflammatory markers (C -reactive protein and sedimentation rate ranging from 2 to 29 mg/dL and 30 to 102 mm, respectively) led to several courses of empirical antibiotic therapy (levofloxacin, ceftriaxone plus azithromycin, ciprofloxacin and vancomycin plus gentamicin) for presumed respiratory or urinary infection, with temporary improvements.
His arteriovenous fistula did not appear infected or inflamed. Microbiological testing (blood, urine, sputum, faeces cultures) was persistently negative. It was during his second hospital admission in November 2008 that he was started on haemodialysis because of an irreversible decline in renal functional (serum creatinine rose from 3.6 to 9 mg/dL).
During these nine months, the patient was submitted to a variety of time -consuming and costly investigations. Echocardiograms, including transoesophagic, excluded prosthetic valve leak or other valve dysfunction. Bone marrow examination was normal. Abdominal echocardiography followed by CT scan showed homogeneous splenomegaly (15 cm) with no other relevant findings. Upper gastrointestinal endoscopy revealed a hiatus hernia with no evidence of bleeding. Testing for hepatitis B and C viruses (HBV, HCV), human immunodeficiency virus (HIV), cytomegalovirus (CMV) and Epstein Barr virus (EBV) was negative. Mantoux testing was anergic. Tumour markers were normal.
The anaemia was considered to be multifactorial due to a combination of sequestration (hypersplenism) plus blood losses (anticoagulation, hiatus hernia and intestinal angiodysplasia) plus haemolysis (prosthetic heart valve and laboratory evidence of intravascular haemolysis) plus chronic disease (CKD5d, although the patient received iron and 10,000 units of beta epoetin three times per week) plus intercurrent infections. At this stage, in May 2009, the patient was referred to the Haematology unit. Based on a positive Coombs test, although performed after several red cell transfusions, he was started on corticosteroid therapy (beginning with prednisolone 80 mg per day) and had an initially favourable haematologic response.
Finally, in June 2009, the patient was readmitted with fever, anaemia and altered mental state. He had lost about 14% of his body weight in 10 months.
He developed generalised tonic-clonic seizures which were controlled with phenytoin and valproic acid. He was pancytopaenic and had elevated inflammatory markers. Again, cultures were negative, as was a CT brain scan and a lumbar puncture. Eventually, a positive phase I antibody titre for Coxiella burnetii revealed the diagnosis of chronic Q fever.
The patient was started on doxycycline and hydroxychloroquine but, within 48 hours, died of cardiorespiratory collapse despite advanced life support procedures.
Case 2
This was a 52-year-old unemployed black female of Guinean nationality with stage 5d chronic kidney disease of unknown aetiology who had been on haemodialysis since 1993. She had undergone implantation of an aortic graft for aortic aneurysm and prosthetic mitral valve placement in 2000, and also had intestinal angiodysplasia and polyps with prior intestinal bleeding episodes. Her regular medication included epoetin beta, iron, warfarin, folic acid, B-complex vitamins and pantoprazole.
She was admitted to hospital on three occasions between July and October 2010 with fluctuant febrile syndrome with persistently negative blood, urine, sputum and faeces cultures. Her arteriovenous dialysis fistula had never appeared infected or inflamed.
She had a normochromic normocytic anaemia (lowest haemoglobin 6.1 g/dL), with a reticulocyte count around 3.5%, elevated lactate dehydrogenase and normal haptoglobin. She received red cell transfusions on two occasions.
Serology was negative for HIV, EBV, CMV, HCV, HBV and Treponema pallidum. Bone marrow examination was suggestive of a reactive inflammatory process or anaemia of chronic disease, and marrow culture was negative. Inflammatory markers were elevated. Liver enzymes were elevated: transaminases 150 to 200 U/L, alkaline phosphatase 100U/L and gammaglutamyltransferase 107 U/L. Transoesophagic echocardiography showed a normally functioning prosthetic mitral valve with no evidence of vegetations. Tumour markers were negative. MR cholangiography and CT scan of chest and abdomen were normal, except for multiple mediastinal lymph nodes, the largest of 15 mm diameter. Bone scan showed multiple old rib fractures, without evidence of metastatic bone disease.
Although blood adenine deaminase (ADA) was positive (103 units/litre), Mantoux testing was anergic and fibre-optic bronchoscopy, and microscopy and culture of aspirations for tuberculosis, were negative, so she was not started on therapy for tuberculosis. During these months, because of suspicion of a respiratory or gastrointestinal source of infection, the patient was given empirical antibiotic therapy (amoxicillin-clavulanate, cefuroxime, ciprofloxacin, vancomycin and gentamicin) with transient improvement.
Three months later, the patient was readmitted with recurrence of the febrile syndrome. She had lost 8% of her body weight and had developed hepatosplenomegaly, later confirmed by ultrasound.
She had become pancytopaenic (Hb 6.4 g/dL, leucocytes 3000/mm3, neutrophils 58%, platelets 40000/uL) and had a C-reactive protein of 21mg/dL and a sedimentation rate of 92 mm. Cultures remained consistently negative as did echocardiographic evaluation and repeat bone marrow examination and culture.
She was initially started on empiric antibiotic therapy with ciprofloxacin and, later, piperacillintazobactam, but did not improve. Then, in view of the combination of unexplained fluctuant fever, anaemia/pancytopaenia, hepatosplenomegaly and prior heart valve disease, doxycycline plus hydoxychloroquine were started empirically, after blood sampling for Coxiella immunology. At the twentieth day of therapy, a positive titre for phase I antibodies was reported, confirming the diagnosis of chronic Q fever.
The patient was discharged from the hospital subjectively improved and afebrile, with improvement in haematologic and inflammatory parameters. A decision was made to continue therapy for a further 17 months with serologic monitoring. The patient was also referred for ophthalmological monitoring for hydroxychloroquine toxicity.
At 3 months follow-up, the phase I IgG titles were still increasing compared to the baseline determination; phase I IgA level remained unchanged at 3200 (Table I).
DISCUSSION
It is of note that our patients did not have professions classically associated with a higher risk of chronic Q fever, such as veterinary or slaughterhouse workers. This probably reflects a more universal exposure to Coxiella burnetii and also its highly infectious nature. Coxiella burnetii is so virulent that a single bacterium is sufficient to infect a human5, meaning the absence of a classic risk exposure profile should not exclude the diagnosis.
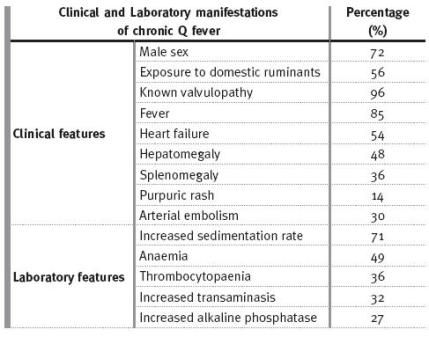
The prevalence of clinical and laboratory manifestations of chronic Q fever have been described in larger series of patients [Table II]. It is important to recognise that about 40 to 55% of patients with acute Q fever and pre-existing heart valve disease progress to chronic Q fever, and the percentage is even higher in patients with prosthetic valves. In fact, endocarditis seems to be the most frequent expression of the chronic form of disease, comprising 77% of the patients, and 88% if there was pre -existing valve disease5,6. Other risk factors for evolving to chronic Q fever are other anomalies or vascular prosthesis and immunocompromise, as in HIV or active neoplasia1,6-9. Thus, an unexplained fluctuant fever in the face of persistently negative cultures, anaemia, increased inflammatory markers and previous heart valve pathology are tell-tale signs that should trigger the serologic search for Q fever, especially in an immunocompromised patient [Table II]. The patients we report had these features [Table I].
Table II
Clinical and laboratory manifestations of chronic Q fever, from Can J Cardiol 2006; 22(9):781 -78511, based on references 15,16,17.
The two cases of chronic Q fever in haemodialysis patients published in the literature were diagnosed in the context of unexplained fever and an unexplained shoulder arthritis4, a rare case of osteoarticular Q fever infection1,10.
It is possible that our first patient had an additional insult to the baseline diabetic nephropathy, in the form of an immune complex-mediated glomerulonephritis, a rare nephrologic expression of chronic Q fever, which contributed to the rapid decline of previously stable renal function (without major proteinuria) in a patient with adequate metabolic and blood pressure control.
Neither of our cases had evidence of vegetations even at transoesophagic echocardiography. It is important to note that in Coxiella burnetii endocarditis, the vegetations are usually small and thin or nodular, making them difficult to recognise by echocardiography, even to the trained eye10,11. The diagnosis of cardiac involvement is typically determined by the modified Duke criteria for endocarditis, and our patients met those criteria8.
Coxiella burnetii does not grow on routine culture substrates12. It is also considered to be an agent of bioterrorism and therefore may only be cultured in special, high-biosafety level laboratories. Serology is the usual way to diagnose Coxiella burnetii infection.
The accepted paradigm is a first seroconversion in acute disease, with antibody positivity to phase II antigens, and, with evolution into chronic Q fever, a second seroconversion, with positivity of antibodies to phase I antigens. The double seroconversion reflects an antigenic shift occurring at the Coxiella surface5. The patients we report were diagnosed accordingly (Table I).Serologic follow-up at 3 months for Patient 2 revealed a still increasing IgG phase I titre and a stable IgA phase I (3200) titre, when compared to baseline. The increasing IgG phase I at follow-up is commonly found, despite appropriate antibiotic treatment, revealing a persisting immunologic stimulus and, perhaps, in this case, a more vigorous immune response of a now healthier, recovering, patient. The already stabilised IgA titles indicate the cure process (Table I).
Both patients were treated with a combination of doxycycline and hydoxychloroquine. Coxiella burnetii seems to have similar susceptibility to drugs such as doxycycline, chloramphenicol, rifampin and quinolones, but monotherapy, although able to reduce symptoms, rarely results in cure11,13. Doxycycline plus hydoxychloroquine for at least 18 months is widely accepted as the first -line regimen1,10,11,13. Some authors recommend serologic monitoring to determine recurrence or cure1,14.
The patients we present were diagnosed at approximately eleven and three months after the first symptoms. The first died but the second was treated successfully. This confirms the poor prognosis of the disease, particularly if treatment is delayed11. The average time to diagnosis in published cases appears to be about six months after the start of symptoms11.
Haemodialysis patients are immunocompromised and have a high incidence of cardiac valve pathology and other comorbidities. They are at high risk for a wide range of cardiovascular and infectious complications, which may potentially confound the diagnosis of chronic Q fever and contribute to the risk of mortality. Use of antibiotics. such as macrolides and quinolones, which have a bacteriostatic, but not bactericidal, effect against Coxiella burnetii, for other indications may also delay diagnosis. For all the above reasons, we hypothesise that chronic Q fever may currently be underdiagnosed in this population.
CONCLUSION
Coxiella burnetii may cause disease in patients without obvious classic risk factors and the chronic form is potentially lethal. Diagnosis is difficult because of the rarity of the infection and its non-specific presentation.
The key is probably to keep in mind the possibility of chronic Q fever when there is an unexplained febrile syndrome plus anaemia and elevated inflammatory markers in a patient with prior heart valve pathology, especially if immunocompromised.
The diagnosis is serologic, since echocardiographic findings are frequently deceptive and isolation of the agent very difficult. Q fever may be underdiagnosed in the dialysis population. Finally, the death of one of the patients we report illustrates the poor prognosis associated with the often late diagnosis of chronic Q fever, while the survival of the other patient emphasizes the potentially treatable nature of the disease.
References
1. Hartzell JD, Wood -Morris RN, Martinez LJ, Trotta RF. Q fever: Epidemiology, Diagnosis, and Treatment. Mayo Clin Proc 2008;83:574-9 [ Links ]
2. Tissot Dupont H, Raoult D, Brouqui P, et al. Epidemiologic features and clinical presentation of acute Q fever in hospitalized patients: 323 French cases. Am J Med 1992;93:427-34 [ Links ]
3. Maurin M, Raoult D. Q fever. Clin Microbiol Rev 1999;12:518-53 [ Links ]
4. Leonetti F, Raoult D, Dusol B, Brunet P, Berland Y. Chronic Q fever in hemodialysis patients. Nephron 1994;67:231-3 [ Links ]
5. Rault D. Microbiology and epidemiology of Q fever. UptoDate online. Section Editor: Sexton DJ, Deputy Editor: McGover BH. Last update Apr 2009
6. Gardon J, Héraud JM, Laventure S, et al. Suburban transmission of Q fever in French Guiana: evidence of a wild reservoir. J Infect Dis 2001;184:278-84 [ Links ]
7. Rault D. Clinical features, diagnosis, treatment, and prevention of Q fever. Uptodate online. Section Editor: Sexton DJ, Deputy Editor: McGover BH. Last update Sept 2008 [ Links ]
8. Rault D. Q fever endocarditis. Uptodate online. Section Editor: Sexton DJ, Deputy Editor: McGover BH. Last update Apr 2009 [ Links ]
9. Raoult D, Fenollar, Stein A. Q fever during pregnancy: diagnosis, treatment, and follow-up. Arch Intern Med 2002;162:701-4 [ Links ]
10. Siciliano RF, Ribeiro HB, Furtado RH, et al. Endocardite por Coxiella burnetii (febre Q). Doença rara ou pouco diagnosticada? Relato de caso. Rev Soc Bras Med Trop 2008;41:409-12 [ Links ]
11. Deyell MW, Chiu B, Ross DB, Nanette Alvarez. Q fever endocarditis: A case report and review of the literature. Can J Cardiol 2006;22:781-5 [ Links ]
12. Rolain JM, Lecam C, Raoult D. Simplified Serological Diagnosis of Endocarditis Due to Coxiella burnetii and Bartonella. Clin Diagn Lab Immunol 2003;10:1147-8 [ Links ]
13. Levy PY, Drancourt M, Etienne J, et al. Comparison of Different Antibiotic Regimens for Therapy of 32 cases of Q fever endocarditis. Antimicrob Agents Chemother 1991;35:533-7 [ Links ]
14. Torley H, Capell H, Timbury M, McCartney C. Chronic Q fever with mixed cryoglobulinaemia. Ann Rheum Dis 1989;48:254-5 [ Links ]
15. Houpikian P, Habib G, Mesana T, Raoult D. Changing clinical presentation of Q fever endocarditis. Clin Infect Dis 2002;34:E28-31 [ Links ]
16. Duroux-Vouilloz C, Praz G, Francioli P, Peter O. Clinical presentation and serologic follow-up of 21 patients. Schweiz Med Wochenschr 1998;128:521-7 [ Links ]
17. Sanchez-Recalde A, Mate I, Lopez E, et al. Coxiella burnetii endocarditis: Long term clinical course in 20 patients. Rev Esp Cardiol 2000;53:940-6 [ Links ]
Dr Ricardo Vizinho
Department of Nephrology
Hospital de Santa Cruz
Carnaxide, Portugal
E-mail: rsenosviz@gmail.com
Conflict of interest statement: The authors have no conflict of interest of any type (personal, commercial, political, academic or financial) in the elaboration and presentation of this work.
Acknowledgment
The authors thank Ana Sofia Santos, PhD (from the National Institute of Health – Dr. Ricardo Jorge) for her useful advice in the development of the manuscript regarding serologic diagnosis and follow-up.
Received for publication: 24/03/2012
Accepted in revised form: 02/09/2012














