Serviços Personalizados
Journal
Artigo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Acessos
Acessos
Links relacionados
-
 Similares em
SciELO
Similares em
SciELO
Compartilhar
Portuguese Journal of Nephrology & Hypertension
versão impressa ISSN 0872-0169
Port J Nephrol Hypert vol.28 no.2 Lisboa jun. 2014
ORIGINAL ARTICLE
Predictive factors of dialysisdependent chronic kidney disease associated with ANCA vasculites
Fatores preditivos de doença renal crónica dependente de diálise nas vasculites ANCA
Ana L. Antunes1; Andreia Campos2; Sofia Santos2; Jorge Malheiro2,3; Pedro Aguiar2; Ramon Vizcaino3,4; Josefina Santos2,3; Luisa Lobato2,3; Antonio Cabrita2
1 Department of Internal Medicine, Hospital de Braga. Braga, Portugal.
2 Department of Nephrology, Centro Hospitalar do Porto – Hospital de Santo António. Porto, Portugal.
3 Multidisciplinary Unit for Biomedical Research UMIB, Instituto de Ciências Biomédicas Abel Salazar, University of Porto. Porto, Portugal.
4 Department of Pathology, Centro Hospitalar do Porto – Hospital de Santo António. Porto, Portugal.
ABSTRACT
Renal involvement in antineutrophil cytoplasmic antibodies (ANCA) associated vasculitis is frequent and, if there is no response to treatment, progression to end-stage renal disease is fast, leading to increased mortality. We evaluated several factors (clinical, analytical and histological) as predictors of progression to dialysis-dependent stage 5 chronic kidney disease within 2 years after diagnosis of kidney biopsy-proven ANCA-associated vasculitis (outcome), between 1997 and 2010. Twenty-seven patients (16 men, mean age 58 years) met the inclusion criteria. The most common extra-renal manifestations were haematological (93%) and systemic symptoms (70%). At the time of biopsy, mean creatinine and proteinuria were 5.11 ± 2.5mg/dL and 2.36 ± 2.1g/day, respectively. The majority of patients (81%) had ANCA against myeloperoxidase. The induction therapy was with corticosteroids and cyclophosphamide in 71%; 40% received maintenance treatment with azathioprine. At 2 years, 12 patients (27.44%) began renal replacement therapy (RRT). Only a higher serum creatinine at diagnosis, within the clinical and analytical variables analysed, was a significant predictor of renal outcome (odds ratio (OR) = 1.73, p = 0.046) in a logistic regression model adjusted for age and sex. We developed a histological index (0 to 1 point considering the absence or presence of: < 30% of normal glomeruli, > 50% cellular crescents, > 30% glomerulosclerosis, moderate-severe tubular atrophy and interstitial infiltrate), which was associated with renal prognosis at 2 years (OR = 2.07, p = 0.043). This means that for each 1-point increase in the created index the likelihood of needing to RRT to 2 years rises 2.1 times. We then stratified the histological variables into glomerular and tubulointerstitial findings. We found that only the glomerular findings (OR = 4.99, p = 0.049) were independent predictors of the outcome, with glomerulosclerosis (OR = 16.7, p = 0.04) being the most significant. We concluded that baseline serum creatinine and glomerular histological findings were independent predictors of the renal prognosis and may prove helpful in the management of these patients.
Key-Words: Chronic kidney disease; predictors; prognosis; ANCA vasculitis.
RESUMO
O envolvimento renal nas vasculites associadas aos anticorpos anticitoplasma de neutrofilos (ANCA) e frequente e, na ausencia de resposta ao tratamento, evoluem rapidamente para doenca renal terminal, com aumento da mortalidade. Avaliamos varios fatores (clinicos, analiticos e histologicos) como preditores da evolucao para Doenca Renal Cronica Estadio 5 com necessidade de dialise aos 2 anos em doentes com envolvimento renal por vasculite ANCA confirmado por biopsia (outcome) entre 1997 e 2010. Vinte e sete doentes (16 do sexo masculino, idade media de 58 anos) obedeceram aos criterios de inclusao. As manifestações extra-renais mais frequentes foram hematologicas (93%) e sintomas sistemicos (70%). A data da biopsia a creatinina e a proteinuria media eram 5,11 ± 2,5mg/dL e 2,36 ± 2,1g/dia, respectivamente. A maioria dos doentes (81%) tinha ANCA contra mieloperoxidase. A terapeutica de inducao foi em 71% com corticoides e ciclofosfamida; 40% receberam tratamento de manutencao com azatioprina. Aos 2 anos, 12 (27,44%) iniciaram tecnicas de substituicao renal (TSR). Apenas a creatinina mais elevada a apresentacao, dentro das variaveis clinicas e analiticas, foi preditor significativo de outcome (odds ratio (OR) = 1,73; p = 0,046), num modelo de regressao logistica ajustado para a idade e sexo. Desenvolvemos um indice histológico (0 ou 1 ponto considerando a presenca de: < 30% de glomerulos normais, > 50% crescentes celulares, > 30% glomerulosclerose, infiltrado intersticial e atrofia tubular moderados-graves), que se associava com o prognostico renal aos 2 anos (OR = 2,07, p = 0,043). Isto significa que, por cada aumento de 1 ponto no indice criado, a probabilidade de necessidade de TSR aos 2 anos torna-se 2,1 vezes maior. Depois estratificamos as variaveis histologicas em achados glomerulares e tubulointersticiais: verificamos que apenas os primeiros eram preditivos do prognostico (OR = 4,99, p =0,049), sendo a glomerulosclerose a mais significativa (OR = 16,07, p = 0,04). Concluiu-se que a creatinina e os achados histologicos glomerulares sao preditores independentes do prognostico renal, podendo mostrar-se uteis no acompanhamento destes doentes.
Palavras-chave: Doenca renal cronica; preditivos; prognostico; vasculite ANCA.
INTRODUCTION
Primary small-vessel vasculitis have the common histological hallmarks of inflammation and fibrinoid necrosis of small-vessel walls with few or no immune deposits. With unknown aetiology, they are also called ANCA-associated systemic vasculitis (AAV), because of their frequent association with antineutrophil cytoplasm antibodies (ANCA)1. The most common types of ANCA are antibodies directed against proteinase-3 (PR3) and myeloperoxidase (MPO)2.
The incidence of these diseases is increasing, and occurring more often in an elderly population, with a peak age between 55 and 70 years old3. The clinical manifestations are diverse and may be limited to the kidney or compromise upper airway, lungs, skin and other organs in different combinations.
However, renal and respiratory manifestations are more common. The introduction of glucocorticoids and cyclophosphamide (Cyc), has transformed overall survival4. The 5-year survival rates are approaching 80%, but relapse and low-grade persistent disease result in survival with high degree of morbidity5.
Worse prognosis is related to diagnosis delay, drugrelated toxicity, refractory disease and propensity to relapse6. In approximately 10 to 15% of patients, renal function is inadequately restored7. If immunosuppressant therapy fails, regular dialysis is the only treatment option, apart from kidney transplantation.
In the meantime, these patients have been exposed to the potentially lethal adverse effects of the immunosuppressant drugs. It is a clinical challenge to distinguish at onset patients who will benefit from immunosuppressant therapy and those who will not, because in irreversible kidney damage it may be justifiable to opt for the renal replacement technique alone. If the latter ones could be identified at the beginning, they would be protected from possible adverse effects of this therapy. To this date, several studies aimed to identify clinical and histological predictors of renal outcome provided contradictory results8.
SUBJECTS AND METHODS
Review of demographic characteristics, clinical presentation, immunological, analytical and histological data in the population of patients with biopsyproven AAV in the Department of Nephrology of Centro Hospitalar do Porto – Hospital de Santo Antonio, between 1997-2010. We defined as the main outcome of interest the need of renal replacement therapy (RRT) within 2 years after vasculitis diagnosis.
We created a global histological index, attributing 0 to 1 point considering the absence or presence of the following: < 30% of normal glomeruli, > 50% cellular crescents, > 30% glomerulosclerosis, moderate to severe tubular atrophy and interstitial infiltrate.
Twenty-seven patients with renal involvement by AAV confirmed by biopsy between 2007 and 2010 were included. The student t-test was used for continuous data and the chi-square test for categorical data. We performed a comparative analysis of clinical variables between 2 subgroups defined by the presence or absence of the defined outcome. We explored possible factors associated with the outcome through different multivariate logistic models; firstly, we constructed a model including age, sex, creatinine at presentation and histology score (model 1). A second model was then evaluated considering age, sex, glomerular and tubulointersticial scores (model 2).
Finally, model 3 was developed to include only the 3 glomerular scores (normal glomeruli < 30%, cellular crescents > 50% and glomerulosclerosis > 50%), age and sex. All statistical data were calculated using SPSS 20.0 software, and results were considered statistically significant at p < 0.05.
RESULTS
Patients had a mean age of 58 ± 17 (range: 30-85 years) and were predominantly male (16 vs. 11). The main clinical presentation was rapidly progressive renal insufficiency (n = 21) or acute kidney injury (n = 6).
Five patients had a history of diabetes mellitus and nine of hypertension. There were five patients with a previous history of chronic kidney disease (CKD) of unknown aetiology prior to the diagnosis of vasculitis, which led us to wonder if this would not be a manifestation of the disease.
Haematological changes were present in 93% (Table I) followed, in order of frequency, by systemic symptoms (70%; of these, the most frequent were anorexia – n = 16). Respiratory symptoms developed in 48% of the patients and were highly variable.
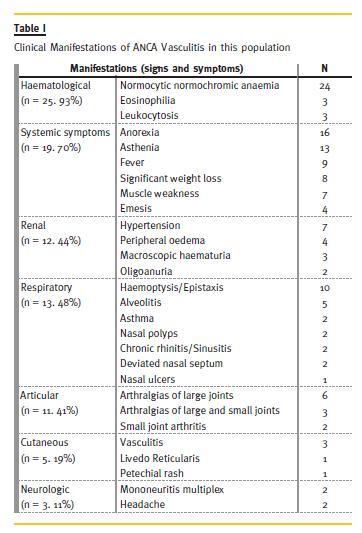
Since renal biopsy was one of the inclusion criteria, all patient had acute kidney injury. Seven had hypertension of recent onset and four presented with peripheral oedema. Approximately 40% of patients had inflammatory arthralgia. Skin involvement was present in 19%. Neurological manifestations were rare (11%), and manifested as headache or mononeuritis multiplex.
Antineutrophil cytoplasmic antibodies (ANCA) against myeloperoxidase (MPO) was the most frequent (81%). In 10 patients antinuclear antibodies (ANA) were positive, with no consumption of complement or anti-double stranded DNA antigen detectable.
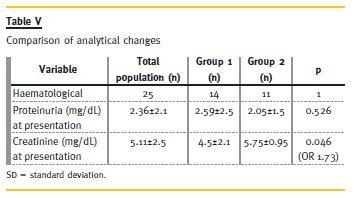
At the time of biopsy, mean serum creatinine was 5.1 ± 2.5 mg/dL (range: 1.6-13mg/dL) and proteinuria average was 2.36 ± 2.1g/day (range: 0.23-7.9 g/day), reaching the nephrotic level in three patients.
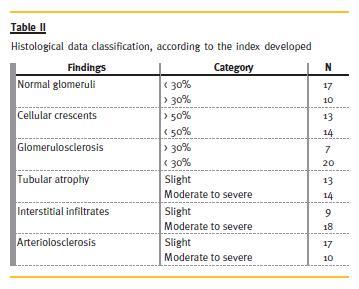
Histological data was evaluated according to the established index (Table II): 17 patients had less than 30% of normal glomeruli, 13 had more than 50% of cellular crescents, seven had more than 30% of glomerulosclerosis, and moderate to severe tubular atrophy, interstitial infiltrates and arteriolosclerosis was present in 14, 18 and 10 patients, respectively.
Five patients had, at presentation, a transient need for dialysis; the creatinine average was 7.5 ± 2.1 mg/dl (range: 1.89-11 mg/dl); all of them recovered after induction therapy, without needing long-term dialysis.
The treatment instituted (Table III) and its duration was highly variable, according to the clinical status of the patient, response to treatment, associated complications, and the dependence of RRT.
Regarding induction therapy, all patients received corticotherapy, alone or combined with intravenous Cyc (n = 14, three in association with plasmapheresis, three with plasmapheresis and intravenous immunoglobulins/i.v. Ig) or oral cyc (n = 11). Maintenance therapy was instituted with Azathioprine/AZA (n = 11), Cyclosporine/CsA (n = 2) and Mycophenolate Mofetil/MMF (n = 1).
As side-effects of therapy, one patient developed iatrogenic diabetes mellitus, three had significant cytopenias and eight had infectious complications requiring hospitalization (respiratory – n = 3; catheter – n = 3; urologic – n = 2).
After 2 years of follow-up, 15 patients did not need RRT (Group 1), while 12 became dependent on haemodialysis (Group 2). At presentation, the creatinine average was 4.5 ± 2.1mg/dl (range: 1.6-9mg/dl) and 5.75 ± 0.95 (range: 1.7-12.6mg/dl), respectively.
Among the first group there was good response of extra-renal manifestations to treatment, without relapses in the first two years. Among patients in Group 2, there was recurrence of respiratory manifestations in two cases, with alveolar haemorrhage, justifying maintenance immunosuppressive therapy.
As regard the renal function in Group 1, mean serum creatinine at 2 years was 1.58 ± 0.6 mg/dL (range: 0.7-2.67mg/dL). The maximum proteinuria was 1.5 g/day, with eight patients having less than 0.5 g/day.
There were no significant differences in respect to demographic data, clinical manifestations, of antibodies between Groups 1 and 2 (Table IV and Table V), and these variables showed no predictive value for RRT need in 2 years. From clinical and analytical variables, creatinine at presentation had a p = 0.046 and an odds ratio (OR) of 1.73. The remaining did not show significant predictive value to our outcome. The histological index we created (Table VI) proved to be a good predictor of outcome (p = 0.043, OR = 2.07). This means that for each 1-point increase in the created index the likelihood of needing RRT to 2 years rises 2.1 times. Glomerular findings (Table VII) were the most important (p = 0.049, OR = 3.99), and within these, glomerulosclerosis (Table VIII) was the main predictor factor (p = 0.046, OR = 16.07).
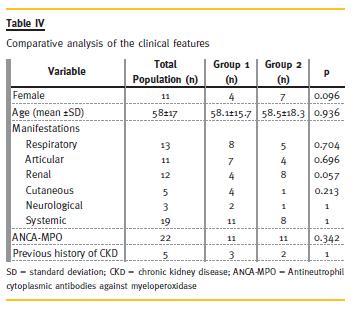
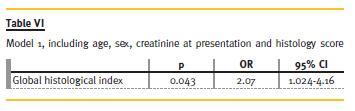
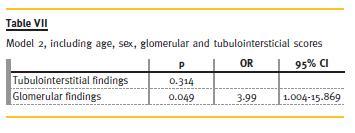
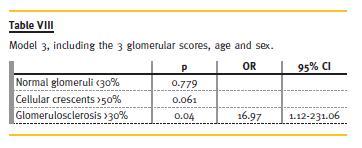
Regarding the evolution (Table IX) until now (and some patients reached a follow-up time of 14 years), from Group 1, two patients died: one for unknown reasons (3rd year) and the other after septic shock following a respiratory infection (8th year). Two patients were lost to follow-up. One patient, 3 years after biopsy, had recurrence of respiratory symptoms, requiring immunosuppressive therapy with AZA and Prednisolone, with clinical improvement. Two patients had deterioration of renal function, due to recurrence of vasculitis, with increased titers of ANCA – one patient recovered (cyc orally for 6 months, followed by AZA), but the other required RRT.
Among patients in Group 2, three underwent renal transplantation; of these, two had a good response, but one had recurrence of renal vasculitis with graft loss and started dialysis again. All of them remained without extrarenal symptoms.
DISCUSSION
Demographic characteristics of this population are similar to those described in the literature, with an average age at diagnosis of 58 and with male predominance9.
Since one of the selection criteria was renal biopsy, renal injury was common to all patients.
Kidney and lung manifestations were the most frequent causes for seeking medical attention. We found a very high incidence of systemic symptoms, often alone and of difficult appreciation, as described in other studies10.
Introduction of Cyc in the AAV treatment has improved prognosis, but the side-effects of long-term treatment are serious11. Maintenance treatment can be performed with AZA, although its intensity and duration is still under discussion. Generalized disease can be treated with pulses of intravenous Cyc or with MMF.
A study evaluating remission induction with MMF was performed in Chinese patients with new-onset renal AAV. Adverse effects and renal function at 6 months were similar between the MMF and Cyc groups, but the dose of Cyc was low (0.5-1 g/m2) compared with conventional European schemes12.
The IMPROVE study compared the efficacy of the 2 drugs in maintenance therapy – of 156 patients, 80 received AZA (2mg/kg/day) and 76 were treated with MMF (2g/day); the authors verified the same rate of adverse effects, but more relapses in patients treated with MMF13.
Some studies demonstrate the efficacy of Rituximab (RTX) in inducing remission without the concomitant use of classical immunosuppressants. A recent report enrolling 197 patients, compared 18-month efficacy of a single course of RTX and conventional immunosupression14. The authors reported no significant difference between the groups in any efficacy measure, including the duration of complete remission and the frequency or severity of relapses. They also found no differences regarding adverse events. In our population, no patient was treated with RTX. Corticosteroids are part of treatment in all regimens but the intensity and duration of steroid treatment is still under discussion. In lifethreatening disease, the adjunctive efficacy of plasma exchange has been demonstrated. In the MEPEX trial, 151 patients were randomized to adjunctive therapy with either 7 plasma exchange treatments or 3 pulses of intravenous methylprednisolone. The mortality in both arms was the same, but renal survival was much better in patients treated with plasma exchange15. Small studies found a benefit of i.v. Ig in persistent disease. In our population, choice and duration of therapy was dependent on the previous condition of the patients and their response to treatment, although, in most cases, we initiated therapy with corticoids and Cyc followed by AZA. It is also important to consider the evolution of the therapeutic schemes over the study period which, by itself, has conditioned the different choices.
Two years after biopsy, 12 patients (44%) needed RRT. Since medical treatment had different time duration and patients had different characteristics, we could not evaluate its potential effect as a predictor of outcome. It is, however, important to refer that all patients who received plasmapheresis needed dialysis at presentation; only three of these were on RRT after 2 years.
Although some studies in the past correlated clinical variables (such as gender and advanced age) with poorer prognosis, there was no statistically significant relationship between those and the need for RRT at 2 years in our population. The ANCA pattern showed no significant relationship with the outcome in our study, although others linked ANCA pattern with the likelihood of relapse16. Similarly to what was found in some published studies17-19, we found that higher values of creatinine at presentation correlated with the clinical outcome in a model adjusted to patient gender and age.
Many authors have evaluated the role of the histological findings in the prediction of renal prognosis of ANCA vasculitis. The findings have often been inconsistent18, with some studies advocating best performance of glomerular findings, while others report best performances of tubulointerstitial findings.
Even within each group, the studies are not consistent about which injury has higher predictive value. Meta-analyses are difficult to conduct: the studies were very heterogeneous9, regarding inclusion criteria, scoring methods, treatment strategies and end-points.
We developed a histological index that considered both glomerular and tubulointerstitial findings, adjusted for age and sex, and found that it was related to the outcome in our population. Comparing tubulointerstitial and glomerular findings, we found the latter one to be a better predictor, with glomerulosclerosis as the most important. Several studies have found similar results. In 2010, an international group of pathologists established an index of 4 histological classes of ANCA vasculitis: focal, crescentic, mixed and sclerosis21. These classes seemed to relate, by the order listed, with the renal prognosis, which was worse in the sclerotic. Patients with sclerotic class also had a higher risk of mortality in the first year.
Two validation studies were published immediately after the initial publication. One of these was in the Chinese population, comprising 121 patients, and showed increased likelihood of progression to end stage renal disease (ESRD) with ascending categories of focal, mixed, crescentic and sclerotic classes. The other study involved 87 Japanese patients and found that renal survival in focal, mixed, crescentic and sclerotic classes at 5 year follow-up was 100%, 96%, 86%, and 29% respectively. Thereafter, there were further validation studies confirming a worse renal prognosis for the sclerotic histological category. In all but the original study, patients with crescentic class biopsies showed better outcome than those with mixed class biopsies. These findings related to the regression of active lesions with therapy, whereas chronic lesions were not reversible22.
However, despite interesting results, our study has some limitations, resulting from the following main reasons: [1] small population sample, since this is a disease with a low incidence; [2] retrospective design, which often complicates access to the data; [3] the heterogeneity of therapeutic regimens instituted, which does not allow us to compare their role in the prevention of progression to ESRD.
Therefore, confirmation of its applicability in a Portuguese population throughout a multicentre study is necessary.
CONCLUSION
In our population, renal function at presentation and histologic findings were important predictors of the need for renal replacement therapy at two years.
Among the histologic factors, the glomerulosclerosis arose as the most important predictor. The evaluation of these factors could prove to be very important in clinical practice, allowing for the selection of patients with poor renal prognosis in whom the introduction of immunosuppression would be more deleterious than advantageous.
References
1. PavoneL, Grasselli C, Chierici E, et al. Outcome and prongnostic factors during the course of primary small-vessel vasculitides. J Rheumatol 2006;33(7):1299-1306. [ Links ]
2. Tesar V, Rihova Z, Jancova E, Rysava R, Merta M. Current treatment strategies in ANCA-positive renal vasculites – lessons from European randomized trials. Nephrol Dial Transplant 2003;18(Suppl 5): v2-v4. [ Links ]
3. PaoliniMV, Ruffino JP, Fernandez Romero DS. Anti-neutrophil cytoplasmic antibodyassociated vasculitis. Clinical aspects and treatment. Medicina (B Aires) 2013;73(2):119-126. [ Links ]
4. Smith RM, Jones RB, Jayne DR. Progress in treatment of ANCA-associated vasculitis. Arthritis Res Ther 2012; 14(2):210. [ Links ]
5. Mukhtyar C, Flossman O, Hellmich B, et al. Outcomes from studies of antineutrophil cytoplasm antibody associated vasculitis: a systematic review by the European League Against Rheumatism systemic vasculitis task force. Ann Rheum Dis 2008;67(7):1004-1010. [ Links ]
6. Vanhille P, Vrigneaud L, Quemeneur T. Renal disease in ANCA-associated vasculites. Presse Med 2012; 41(3 Pt 1):247-253. [ Links ]
7. de Lind van Wijngaarden RA, Hauer HA, Wolterbeek R, et al. Chances of Renal Recovery for Dialysis-Dependent ANCA-associated glomerulonephritis. J Am Soc Nephrol 2007;18(7):2189-2197. [ Links ]
8. de Lind van Wijngaarden RA, Hauer H A, Wolterbeek R, et al. Clinical and histologic determinants of renal outcome in ANCA-associated vasculitis: A prospective analysis of 100 patients with severe renal involvement. J Am Soc Nephrol. 2006;17(8):2264-2274. [ Links ]
9. Seck SM, Dussol B, Brunet P, Burtey S. Clinical features and outcomes of ANCAassociated renal vasculitis. Saudi J Kidney Dis Transpl 2012;23(2):301-305. [ Links ]
10. Eisenberger U, Fakhouri F, Vanhille P, et al. ANCA-negative pauci-immune renal vasculitis: histology and outcome. Nephrol Dial Transplant 2005;20(7):1392–1399. [ Links ]
11. Kallenberg CG. Treatment of ANCA-associated vasculitis, where to go? Clin Rev Allergy Immunol 2012;43(3):242-248. [ Links ]
12. Ntatsaki E, Mooney J, Watts RA. ANCA vasculitis: time for a change in treatment paradigm? Not yet. Rheumatology (Oxford) 2011;50(6):1019-1024. [ Links ]
13. Silva F, Cisternas M. Anti-neutrophil citoplasmic antibody (ANCA)-associated vasculitis: advances in pathogenesis and treatment. Rev Med Chile 2013;141(6):765-773. [ Links ]
14. Specks U, Merkel PA, Seo P, et al. Efficacy of remission-indution regimens for ANCAassociated vasculitis. N Engl J Med 2013;396(5):417-427. [ Links ]
15. Tesar V, Rihova Z, Jancova E, Rysava R, Merta M; European randomized trials. Current treatment strategies in ANCA-positive renal vasculites – lessons from European randomized trials. Nephrol Dial Transplant. 2003;18 (Suppl 5):v2-v4. [ Links ]
16. Lionaki S, Blyth ER, Hogan SL, et al. Classification of antineutrophil cytoplasmic autoantibody vasculitides: the role of antineutrophil cytoplasmic autoantibody specificity for myeloperoxidase or proteinase 3 in disease recognition and prognosis. Arthritis Rheum 2012;64(10):3452-3462. [ Links ]
17. Vergunst CE, van Gurp E, Hagen CE, et al. An index for renal outcome in ANCAassociated . Am J Kidney Dis 2003;41(3):532-538. [ Links ]
18. Kaplan-Pavlovcic S, Cerk K, Kveder R, Lindic J, Vizjak A. Clinical prognostic factors of renal outcome in anti-neutrophil cytoplasmic autoantibody (ANCA)-associated glomerulonephritis in eldery patients. Nephrol Dial Transplant 2003;18 Suppl 5:v5-v7. [ Links ]
19. Rupcic´ V, Jakic´ M, Rupcic´ V, Vizjak V, Fijacko M. Clinical course and outcome predictors in pauci-immune ANCA-positive renal-limited vasculitis. Acta Clin Croat, 2011;50(4):475-483. [ Links ]
20. Hauer HA, Bajema IM, van Houwelingen HC, et al. Determinants of outcome in ANCAassociated glomerulonephritis: A prospective clinico-histopathological analysis of 96 patients. Kidney Int 2002;62(5):1732–1742. [ Links ]
21. Rahmattulla C, Berden AE, Bajema IM. Kidneys in ANCA-associated vasculites: what to learn from biopsies?. Presse Med 2013;42(4 Pt 2):563-565. [ Links ]
22. Neumann I, Kain R, Regele H, Soleiman A, Kandutsch S, Meisl FT. Histological and clinical predictors of early and late renal outcome in ANCA-associated vasculitis. Nephrol Dial Transplant 2005;20(1):96-104. [ Links ]
Dra Ana Luisa Antunes
Department of Internal Medicine
Hospital de Braga. Sete Fontes, 4710-243 Sao Victor.
Braga, Portugal
E-mail: ana_luisa_antunes@hotmail.com
Conflict of interest statement: This work was partially supported by the Multidisciplinary Unit for Biomedical Research UMIB, which is funded by grants from the Foundation for Science and Technology (FComp-01-0124-FEDER-015896).
Received for publication: 22/11/2013
Accepted in revised form: 09/05/2014














