Serviços Personalizados
Journal
Artigo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Acessos
Acessos
Links relacionados
-
 Similares em
SciELO
Similares em
SciELO
Compartilhar
Portuguese Journal of Nephrology & Hypertension
versão impressa ISSN 0872-0169
Port J Nephrol Hypert vol.29 no.2 Lisboa jun. 2015
ORIGINAL ARTICLE
Impact of pre-transplant anti-MICA sensitization in graft rejection and survival
Impacto da sensibilização anti-MICA pré–transplante na rejeição e sobrevida do enxerto
Rui Costa1, Jorge Malheiro2, Sandra Tafulo3, Clara Santos4, Manuela Almeida2, Sofia Pedroso2, La Salete Martins2, Leonídio Dias2, António Castro-Henriques2
1 Nephrology Department, CHTMAD, Vila Real, Portugal
2 Nephrology and Kidney Trasplant Department, CHP, Oporto, Portugal
3 Centro do Sangue e Transplantação do Porto, Oporto, Portugal
4 Nephrology Department, CHVNG, Vila Nova de Gaia, Portugal
ABSTRACT
Background: Evidence supporting deleterious effect of preformed major histocompatibility class I chain-related A (MICA) antibodies in rejection incidence and graft survival is still unclear. Methods: Retrospective analysis of 554 kidney transplanted patients. Comparison between positive or negative for MICA antibodies patients was performed to characterize sensitizing triggers. Further classification according to pre-transplant flow cytometry-recorded anti–MICA and/or anti-human leukocyte antigen (HLA) antibodies was made to determine first year rejection incidence and graft survival. Multivariate analysis was applied to determine predictors for acute rejection. Results: Pre-formed anti-MICA antibodies were detected in 41 patients (7.4%). HLA sensitization, blood transfusions and pregnancies were frequently found in anti-MICA+ patients but only pre-formed anti-HLA class I antibodies showed independent association (OR 2.67, p = 0.02). Comparing to MICA-/HLA–, MICA-/HLA+ group presented significantly lower first year rejection-free survival (78.6% vs. 89.3%, p < 0.01), mostly occurred in the first six months, while no difference was found in MICA+/HLA– (88.9% vs. 89.3%, p = ns). MICA-/HLA+ showed independent impact in rejection (OR 2.09, p = 0.03), while no evidence was found in MICA+/HLA- (OR 1.08, p = ns). At 4 years, MICA-/HLA+ group presented lower graft survival (85.8% vs. 95.3%, p = 0.03). Again, no difference was found in MICA+/HLA- group (95.1% vs. 95.3%, p = ns). Conclusion: Our results do not support HLA-independent deleterious pathogenic role of pre-formed MICA antibodies on first year rejection incidence and graft survival.
Key-Words: HLA; kidney; MICA; rejection; survival; transplant.
RESUMO
Introdução: O efeito deletério dos anticorpos para antigénios MICA (major histocompatibility class I chainrelated A) na incidência de rejeição aguda e sobrevida do enxerto ainda não está consensualmente estabelecido. Metódos: Estudo retrospetivo de 554 transplantados renais. A análise comparativa entre doentes positivos e negativos para anticorpos anti-MICA pré-formados foi realizada para avaliar eventos sensibilizadores. A incidência de rejeição aguda no primeiro ano pós transplante renal e a sobrevida do enxerto renal foram determinadas consoante o resultado da citometria de fluxo pré-transplante para anticorpos anti-MICA e/ou anti- HLA (anti-human leukocyte antigen). Aplicou-se um modelo de análise multivariada para identificação de preditores independentes para rejeição aguda. Resultados: Foram identificados 41 doentes (7.4%) com anticorpos anti-MICA pré formados. A sensibilização para HLA, as transfusões sanguíneas e gestações prévias foram mais frequentes nos doentes MICA + mas apenas a presença de anticorpos anti-HLA classe I apresentou uma associação independente (OR 2.67, p = 0.02). Comparativamente ao grupo MICA-/HLA–, o grupo MICA-/HLA+ apresentou menor sobrevida livre de rejeição ao 1º ano (78.6% vs. 89.3%, p < 0.01), maioritariamente ocorrida nos primeiros seis meses, enquanto que nenhuma diferença foi encontrada com o grupo MICA+/HLA– (88.9% vs. 89.3%, p = ns). Apenas o status MICA-/HLA+ teve impacto independente na incidência de rejeição (OR 2.09, p = 0.03), ao contrário do status MICA+/HLA- (OR 1.08, p = ns). O grupo MICA-/HLA+ apresentou menor sobrevida do enxerto censurada para a morte aos 4 anos (85.8% vs. 95.3%, p = 0.03), não se verificando diferenças no grupo MICA+/HLA- (95.1% vs. 95.3%, p = ns). Conclusão: Os nossos resultados não suportam um efeito deletério dos anticorpos pré-formados para MICA, independente da sensibilização HLA, na incidência de rejeição aguda no 1º ano pós transplante e na sobrevida do enxerto.
Palavras-Chave: HLA; MICA; rejeição; rim; sobrevida; transplante.
INTRODUCTION
In the past years, association between anti-human leukocyte antigen (HLA) sensitization and worse graft prognosis has been reinforced by several studies.
The basis for these findings consists on antibodymediated rejection (AMR), which has been recognized as a major predictor of acute and chronic graft loss1.
However, even in full matches at the HLA loci, graft rejection can still occur2, leading to an increasing belief that non-HLA antigens could be additional injury targets in transplant organs.
Major-histocompatibility-complex class (MHC) I-related chain A (MICA) has drawn major attention as a potential antigen target. The MICA gene is situated within the MHC region on chromosome 6p21.3 centromeric to HLA-B in the class I region and encodes polymorphic surface glycoproteins with functions related to innate immunity2. It has predominant expression on fibroblasts, epithelial, dendritic and endothelial cells, but can be upregulated and recognized by natural killer (NK) cells in virtually all tissues, including activated CD4+ and CD8+ cells, as a consequence of cell stress caused by cytomegalovirus (CMV) or other viral infections, tumours, autoimmune disorders or ischemia-reperfusion injury3.
Since evidence of anti-MICA antibodies capability in complement activation and endothelial injury was found, further investigations were directed to identify the anti-MICA sensitization mechanisms in waiting list patients and to discover its role in antibody-mediated rejection mechanism and effect in graft survival3-6.
Due to its polymorphic nature and endothelial presentation, exposure to allogenic MICA occurring in blood transfusion, pregnancies and during transplantation could elicit antibody formation, like allogenic HLA antigens. It has been suggested that pregnancy and retransplantion can induce MICA antibodies4-6 but the role of blood transfusions is still unclear since only one study showed this association6.
Several small reports suggested that preformed anti-MICA antibodies could have a role in antibodymediated rejection (AMR), since association to rejection events were found7. Additionally, preformed anti-MICA antibodies were found to have deleterious effect on graft survival in one multicentre study8. However, this effect was not found in more recent reports that did not show an independent pathogenic role of anti-MICA antibodies on graft survival6,9.
The purpose of this study relies on description of demographic, clinical and transplant features associated to MICA sensitizing events. Afterwards, we analyse the possible impact, independent or associated to anti-HLA antibodies, of pre-formed MICA antibodies in rejection incidence and graft survival.
MATERIAL AND METHODS
We retrospectively analysed 554 patients who received a kidney transplant between 2007 and 2013 in our unit. In all patients, pre-transplantation serum samples were collected and analysed for anti-MICA and anti-HLA antibodies. Data regarding demographic, clinical, induction and maintenance immunosuppressive therapy, delayed graft function, allograft survival and rejection episodes were collected from each kidney transplant recipient. Graft loss was considered to calculate death censored graft survival.
Allograft rejection was defined as biopsy proven rejection and classified according to the Banff Conference Report as updated in 200710.
Complement-dependent cytotoxicity panel-reactive antibodies test (CDC PRA) was performed in all patients and considered positive if cell lyses remained present after dithiothreitol (DTT) treatment, identifying only IgG anti-HLA isotypes positive cases. Pretransplantation screening of patients was performed for MICA and HLA antibodies using LABScreen Mixed assay (One Lambda, Inc. Canoga Park, CA, USA) and considered positive when the mean fluorescence intensity (MFI) of each bead was above the cutoff defined by the manufacturer. Microbeads were covered with the major HLA class I and Class II antigens and MICA antigens 01, 02, 04, 07, 09, 12, 17, 18, 19 and 27. HLA antibody specificity was confirmed by performing single antigen bead using LABScreen Single Antigen assay. Donor-specific antibody (DSA) positive reaction was defined above empirically determined MFI cutoff of 1000. However, this determination began in mid 2009, so almost half of our anti-HLA positive patients did not have DSA determination (47.6%). In order to avoid reducing statistical power of our analysis by removing these patients with unknown DSA identification, we considered only flow cytometry for anti-HLA antibodies results.
The association between demographic, clinical, immunological features and presence of pre-transplant MICA antibodies was compared using Pearsons chi-squared test or Fishers exact test for categorical data and students t-test for numerical data. Further binary logistic regression analysis was applied to determine independent effect on variables presenting p < 0.1 in univariate analysis. Survival analysis was performed using the Kaplan-Meier method and compared among groups by log rank analysis (global and pairwise comparisons). Logistic regression analysis was used to evaluate predictors for rejection during first year post-transplantation. Variables that reached p < 0.1 in univariate analysis were stepwise entered into multivariate model to access their independent effect. A p-value of less than 0.05 was considered significant. The SPSS 18.0 was used for analysis (SPSS, Inc. Chicago, IL, USA).
RESULTS
Pre-formed anti-MICA antibodies were detected in 41 patients (7.4%). Demographic and clinical characteristics stratified for MICA antibody are listed in Table I.. After univariate analysis, blood transfusion and pregnancy were associated to MICA antibodies, while no difference was noted regarding retransplantation, recipient/donor CMV status and positive panel-reactive antibody. Induction and maintenance immunosuppression was similar between both groups, although slightly less patients anti-MICA + received tacrolimus as part of their immunosuppressive regimens (not statistically significant) and only two were submitted to living donor transplant. In our series, 14 of 41 MICA + patients were also anti HLA+ (34.1%), as opposed to 95 MICA- patients (18.5%). This difference reached statistical significance (p = 0.017). However, this difference is mainly attributed to anti- HLA class I antibodies (31.7% vs. 15.8%, p = 0.01) and no difference was found in anti-HLA Class II antibodies between both groups. Given these results, binary logistic regression analysis including blood transfusion, pregnancies and class I or class II HLA antibodies, revealed that only anti HLA class I antibodies were associated to positive MICA (OR 2.77, 1.20 – 6.38). Tendency for lower MICA+ prevalence in living donor transplant was found (OR 0.25; 0.06 – 1.05).
A total of 86 patients (15.5%) experienced acute rejection during the first year post-transplant. In ten patients, histology data was not available. In the remaining, acute cellular rejection predominated (72.3%), mainly Banff 2 grade (Banff 1a/1b: 32.9%; Banff 2a/2b: 39.5%). Acute antibody-mediated rejection (AAMR) was responsible for the remaining cases (25%) and two patients presented evidence of chronic AMR (2.6%).
Regarding post-transplant outcomes, delayed graft function occurred equally in MICA positive or negative groups (17.7% and 24.4%, respectively). The same was true for acute rejection and histological characterization (acute cell rejection (ACR) / AAMR) was roughly similar in both groups. No differences were found in graft or patient survival during 30.3 ± 19.3 months of follow-up. In order to further investigate the independent impact of MICA antibodies, regardless of anti-HLA recipient status, patients were characterized into four categories: MICA-/HLA- (n = 413); MICA+/HLA- (n = 27); MICA-/HLA+ (n = 95) and MICA+/HLA+ (n = 14).
Rejection-free survival curves for each group during first year post-transplantation are showed in Fig.1. At first glance, anti-MICA status seems to mimic anti-HLA status. Anti-HLA positive groups, with or without MICA antibodies, presented lower first year rejection-free survival (76.8% and 78.6%, respectively) comparing to MICA-/HLA- (89.3%) but statistical significance was only obtained in HLA+/MICA- after stepwise analysis (p = 0.001). No differences were found in MICA+/HLA- when compared to MICA and HLA negative patients (88.9% vs. 89.3%, p = 0.948) and positive MICA status did not present addictive effect on anti-HLA positive group.
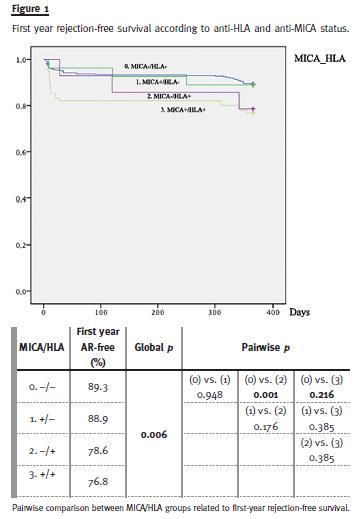
Further comparisons between these four groups and timing of first rejection (overall, 0-6 months or 6-12 months) were made (Table II). In the total 12 months period, rejection incidence was superior in MICA-/HLA+ versus MICA-/HLA- (23.2% vs. 10.6%, p < 0.01). Subsequent analysis for each period revealed that allograft rejection was higher in the first six months after transplantation in MICA-/HLA+ vs. MICA-/HLA- patients (17.9% vs. 6.7%, p = 0.001). No differences were found in the other groups comparisons.
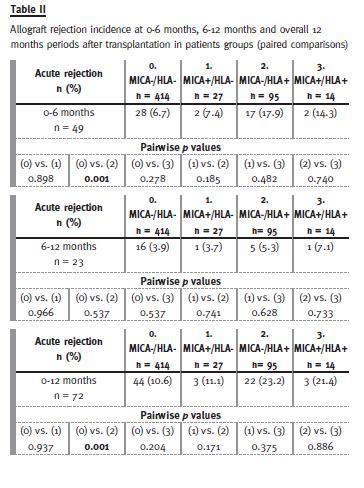
The same analysis for each type of rejection (cellular or antibody-mediated) showed similar results (data not shown).
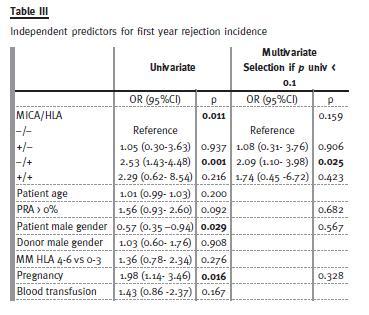
In order to evaluate the independent role of anti-MICA status in rejection occurrence, logistic regression analysis was applied considering anti-MICA and anti- HLA antibodies, positive PRA, donor and recipient gender, recipient age, HLA mismatch, previous blood transfusion or pregnancy (Table III). The production of anti-HLA antibodies was significantly associated with first-year rejection occurrence: MICA-/ HLA+ presented stronger association when compared with control group after multivariate analysis (OR 2.09: 1.10-3.98). Single production of anti-MICA antibodies had no effect in first year rejection incidence (OR 1.08: 0.31-3.76), neither did the concomitant presence of anti-MICA and anti-HLA antibodies present a stronger association when compared to the presence of anti-HLA antibodies alone (OR 1.74; 0.45-6.72).
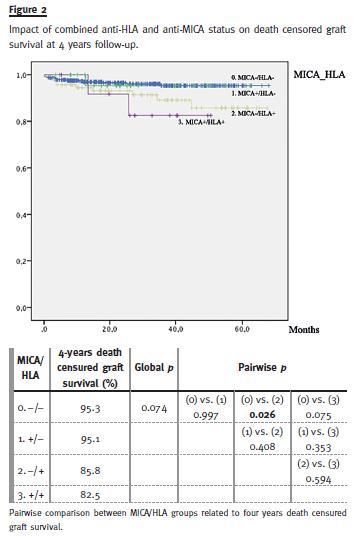
Among patients categorized according to anti-HLA and anti-MICA antibodies and considering a total of 4 years follow-up (Fig.2), tendency for lower deathcensured graft survival was found in MICA+/HLA+when compared to MICA-/HLA- patients (82.5% vs. 95.3%, p = 0.075). The lack of significance is justified by the small number of cases analysed in this group (n = 14). Worse graft prognosis was detected in MICA-/HLA+ (85.8% vs. 95.3%, p = 0.026), while similar graft survivals were shown between MICA+/HLA- and MICA-/HLA- groups (95.1% vs. 95.3%)
DISCUSSION
In our patients submitted to kidney transplant, we detected preformed anti-MICA antibodies in 7.4% of cases. This value is similar to the same pretransplantation anti-MICA antibodies prevalence reported by Sánchez-Zapardiel et al9, while in other studies anti-MICA antibodies determination varied from 11% to 25%8-11. The variation in these determinations may be explained by the heterogeneous populations analysed in each study regarding gender distribution, retransplanted patients proportions and methodological differences in MICA antibodies determination.
The number of different MICA specificities used for Anti-MICA antibodies in flow cytometrybased reagents differs in all studies, varying from five to twenty three in recent ones6,8,9. Logically, higher number of known specificities will increase anti-MICA prevalence. Additionally, cytometry-flow beads bound to single recombinant MICA antigens used for IgG anti-MICA determinations varied between LSA-MIC kit (Gen-Probe Transplant Diagnostics) or LABScreen MICA Single Antigen kit (One Lambda), which invariably accentuates methodological differences.
Real estimate of true anti MICA antibodies prevalence before transplantation will be difficult until uniformity and consensual methodology in MICA antibodies determination is achieved.
During the present study, we tried to determine the factors associated to anti MICA sensitization in patients submitted to kidney transplant, notwithstanding our small number of MICA positive cases.
In addition, we analysed if classic anti-HLA sensitizing events also induced anti-MICA sensitization. History of previous transplantation was not more frequent in the MICA+ group, similarly to previous reports8,9.
No differences were found regarding recipients or donors gender. Previous reports found tendency for higher association between male recipient and MICA positivity despite the potential role of pregnancy as a sensitizing event6,8. In these studies, the reason for the influence of male gender on the presence of MICA antibodies was unclear and further investigation was required. Nevertheless, this association was not verified in other studies5,9.
Blood transfusions or pregnancy history occurred more frequently in MICA+ patients and concomitant higher detection of anti-HLA antibodies in these patients would make us consider that those events are causative for both antibodies formation. However, this hypothesis dissipates when only anti-HLA formation showed independent role on MICA sensitization.
Lemy et al.6 identified previous transfusion and pregnancy as independent risk factors for development of MICA antibodies. The absence of this effect in our sample may be related to our lesser number of patients submitted to blood transfusions when compared to Lemys study (34.3% vs. 48.1%) and in different pregnancy variable categorization (we included any episode as positive, while Lemy considered positive more than two episodes).
Patients sensitized against MICA antigens were more likely to have antibodies against HLA as well.
Interestingly, only HLA class I sensitization showed independent association with MICA antibodies formation, suggesting that similar sensitizing events can produce anti-MICA and anti-HLA antibodies simultaneously.
Endothelial cell injury caused by inflammation, ischaemia or viral infections induces higher expression of both HLA Class I and MICA antigens in cells. One example is CMV infection, which has been showed to upregulate the expression of MICA antigens forms and causes a possible anti-MICA antibody response12,13. We analysed whether anti-CMV positivity was more frequent in MICA+ patients, but no difference was found, like previously reported9.
In our unit, living donor transplant is mostly performed in pre-emptive manner, although some patients may start dialysis while waiting for conclusive donor evaluation. Interestingly, living donor patients tended to present less frequently anti MICA antibodies (OR 0.25, p = 0.059). In one previous study, chronic kidney disease (CKD) stage 5 was found to present increased prevalence of MICA immunization when compared to controls, regardless of transfusions and transplantation events6. One possible explanation for these results is uraemia induced endothelial cells stress with increased MICA antigen or cumulative exposure to dialysis filters and dialysate fluids. Lower cumulative time on CKD stage 5 and under dialysis treatment may explain our lower MICA sensitization on living donor transplanted patients.
Evidence pointing to a direct pathogenicity of MICA antibodies in rejection events is essentially circumstantial, mainly based on higher frequency of MICA antibodies detection among patients that experienced rejection4. However, concomitant anti-HLA antibodies presence in these cases makes difficult to infer a causative role of anti-MICA antibodies. In our study, allograft rejection was observed more frequently only in MICA-/HLA+ and most episodes occurred in the early six months period (Table II), a typical feature of rejection mediated by pre-formed antibodies. Furthermore, preformed MICA antibodies showed no independent role in allograft rejection occurrence, regardless of anti HLA antibodies detection.
Stepwise analysis has not shown lower rejection-free survival of MICA positive patients without HLA sensitization when compared to the control group. In fact, only three patients experienced rejection in the MICA+/HLA- group. The small number of events may have underpowered any potential causative role of anti MICA antibodies. Nevertheless, previous studies were also not able to show increased risk for rejection in MICA+ patients reporting similar first year cumulative rejection episodes comparing to MICA- patients6. Only one study showed borderline association between MICA+/HLA– patients and rejection incidence during the first 3 months when compared to the control group (8% vs. 2%, p = 0.049)9.
One immunohistochemistry study14 has showed increased MICA antigen expression in podocytes within the glomeruli during rejection events, as well within infiltrating mononuclear cell, B cells, CD8+ T cells and NK cells. Logically, AMR mechanisms could be present especially because MICA antibodies are capable of complement-dependent cell injury. Additionally, direct CD8+ T and NK cells activation via NKG2D ligands engaging MICA antigen could make possible to increase direct cellular-mediated lyses mechanisms during the rejection episode14. Previous reports failed to show an independent association of MICA antibodies in antibody-mediated rejection episodes, while higher frequency of cellular mediated rejection (CMR) in MICA+ patients has been described5.
This finding was not achieved in our study since similar proportion of CMR episodes between overall MICA+ and MICA- patients and after MICA/HLA characterization was found. Lack of independent pathologic role of anti MICA antibodies in AMR events may be related to the already know stronger effect of anti-HLA antibodies also found in these patients, masking any potential independent pathogenicity of MICA sensitization.
The present data argues against a significant effect of preformed MICA antibodies on graft survival. Possible addictive deleterious effect of MICA sensitization on already HLA sensitized patients is suggested after graft survival curves analysis but statistical significance was not achieved, probably due to the small number of cases. In two major studies that evaluated the effect of pre-transplant MICA sensitization on graft survival, considering overall and only negative PRA cases, showed broadly dissimilar results. The first one, a multicentre cohort of 1910 kidney transplant recipients8 showed not only association between preformed anti MICA antibodies and lower graft survival but also a stronger deleterious effect on first graft transplants, lower HLA mismatches and in absence of panel-reactive HLA antibodies, suggesting an independent role of MICA antibodies.
However, another cohort study6 with 10 years followup showed similar first year rejection incidence and overall graft survival in MICA patients, comparing to MICA negative group. Further analysis considering only primary grafts revealed the same outcome. The conflicting results between these studies were explained by differences in immunosuppressive protocols, since lower induction therapy and lower prevalence in tacrolimus and MMF use were found in the one that showed lower first year graft survival.
Therefore, the impact of MICA antibodies could be attenuated under heavier immunosuppression. In our case, about 90% of patients performed induction therapy, with antithymocyte globulin (32.1%) or IL-2 receptor inhibitor (58.3%), and practically all used tacrolimus (95.5%) in maintenance therapy. More recently, the Sanchez-Zapardiel group9 also failed to show lower graft survival in MICA+/HLA- patients and justified their results because of the small sample size (n = 25) and higher efficiency of modern drugs, allowing better resolution of rejection episodes.
In order to achieve true impact of MICA antibodies on kidney transplantation it is necessary to determine the pathogenic role of MICA donor-specific antibodies (MICA DSA) instead of just taking into account the presence or absence of antibodies regardless of donor specificity. Therefore, MICA alleles typing of patients and donors would allow MICA mismatching determination and identification of pre-formed MICA DSA. Cox et al5 have shown that mismatching MICA alleles could lead to posttransplant MICA antibodies and these antibodies were independently associated with decreased glomerular filtration rate and poorer graft outcome in the first two years after transplantation. These findings suggest that MICA DSA antibodies could have distinct pathologic role to HLA DSA antibodies, contributing to deleterious graft outcome.
In summary, our data did not show a meaningful pathogenic role of preformed MICA on first year rejection incidence or allograft survival in our centre.
It further shows that the study of pre-transplant MICA sensitization and impact on kidney transplantation is severely hampered by the low prevalence of MICA detection and their frequent association with HLA antibodies. Therefore, further studies enclosing higher number of patients analysed, methodological uniformity in MICA antibodies identification and donor specificity determination will be needed to assess the possible independent role of preformed MICA in post-transplant outcomes.
References
1. Otten HG, Verhaar MC, Borst HP, Hené RJ, van Zuilen AD. Pretransplant donor-specific HLA class-I and class-II antibodies are associated with an increased risk for kidney graft failure. Am J Transplant 2012; 12(6):1618-1623. [ Links ]
2. Opelz G with the Collaborative Transplant Study. Non-HLA transplantation immunity revealed by lymphocytotoxic antibodies. Lancet 2005;365(9470):1570-1576. [ Links ]
3. Sumitran-Holgersson S, Wilczek HE, Holgersson J, Söderström K.. Identification of the nonclassical HLA molecules, MICA, as targets for humoral immunity associated with irreversible rejection of kidney allografts. Transplantation 2002;74(2):268-277. [ Links ]
4. Mizutani K, Terasaki PI, Shih RN, Pei R, Ozawa M, Lee J. Frequency of MIC antibody in rejected transplant patients without HLA antibody. Hum Immunol 2006;67(3): 223-229. [ Links ]
5. Cox ST, Stephens HA, Fernando R, et al. Major histocompatibility complex class I-related chain A allele mismatching, antibodies, and rejection in renal transplantation. Hum Immunol 2011;72(10):827-834 [ Links ]
6. Lemy A, Andrien M, Wissing KM, et al. Major histocompatibility complex class 1 chainrelated antigen A antibodies: sensitizing events and impact on renal graft outcomes. Transplantation 2010;90(2):168-174. [ Links ]
7. Zou Y, Mirbaha F, Lazaro A, Zhang Y, Lavingia B, Stastny P. MICA is a target for complement-dependent cytotoxicity with mouse monoclonal antibodies and human alloantibodies. Hum Immunol 2002;63(1):30-39. [ Links ]
8. Zou Y, Stastny P, Süsal C, Döhler B, Opelz G. Antibodies against MICA antigens and kidney-transplant rejection. N Engl J Med 2007;357(13):1293-1300. [ Links ]
9. Sánchez-Zapardiel E, Castro-Panete MJ, Castillo-Rama M, et al. Harmful effect of preformed anti-MICA antibodies on renal allograft evolution in early posttransplantation period. Transplantation 2013;96(1):70-78. [ Links ]
10. Solez K, Colvin RB, Racusen LC, et al. Banff 07 classification of renal allograft pathology: updates and future directions. Am J Transplant 2008;8(4):753-760 [ Links ]
11. Zou Y, Heinemann FM, Grosse-Wilde H, et al. Detection of anti MICA antibodies in patients awaiting kidney transplantation, during the post-transplant course, and in eluates from rejected kidney allografts by Luminex flow cytometry. Hum Immunol 2006;67(3):230-237. [ Links ]
12. Zou Y, Bresnahan W, Taylor RT, Stastny P. Effect of human cytomegalovirus on expression of MHC class I-related chains A. J Immunol 2005;174(5):3098-3104. [ Links ]
13. Groh V, Rhinehart R, Randolph-Habecker J, Topp MS, Riddell SR, Spies T. Costimulation of CD8alphabeta T cells by NKG2D via engagement by MIC induced on virus-infected cells. Nat Immunol 2001;2(3):255-260. [ Links ]
14. Li L, Chen A, Chaudhuri A, et al. Compartmental localization and clinical relevance of MICA antibodies after renal transplantation. Transplantation 2010;89(3):312-319. [ Links ]
Dr. Rui Costa
Nephrology Department, HTMAD
Avenida da Noruega, 5000-508 Lordelo, Vila Real, Portugal.
E-mail: ruimiguelccosta@gmail.com
Conflict of interest statement: None declared.
Received for publication: 14/10/2014
Accepted in revised form: 10/02/2015














