Serviços Personalizados
Journal
Artigo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Acessos
Acessos
Links relacionados
-
 Similares em
SciELO
Similares em
SciELO
Compartilhar
Portuguese Journal of Nephrology & Hypertension
versão impressa ISSN 0872-0169
Port J Nephrol Hypert vol.29 no.2 Lisboa jun. 2015
CASE REPORT
Mesalazine induced tubulointersticial nephritis
Nefrite tubulointersticial associada à mesalazina
Andreia Campos1, Sofia Santos1, Josefina Santos1,2, Jorge Malheiro1,2, Anabela Rodrigues1,2, Luísa Lobato1,2, J. Ramón Viscaíno3, António Cabrita1
1 Department of Nephrology, Centro Hospitalar do Porto – Hospital de Santo António. Porto, Portugal.
2 Multidisciplinary Unit for Biomedical Research UMIB, Instituto de Ciências Biomédicas Abel Salazar, University of Porto. Porto, Portugal.
3 Department of Pathology, Centro Hospitalar do Porto – Hospital de Santo António. Porto, Portugal.
ABSTRACT
Inflammatory bowel disease and its various treatments may affect the kidney in several ways. Tubulointersticial nephritis is a rare but serious complication of longer-term mesalazine use. There are few cases reported in the literature. We report the first two cases of mesalazine-induced tubulointersticial nephritis, recently diagnosed in our department. The first one refers to a patient with ulcerous colitis and the second one to a patient with Crohns disease. Then the authors present a review of literature about the renal involvement in the inflammatory bowel disease. New cases of mesalazine nephrotoxicity should be reported to allow more accurate incidence estimation of this serious adverse effect. Routine monitoring of renal function is simple, inexpensive and allows an early diagnosis of this complication.
Key-Words: Inflammatory bowel disease; kidney disease; mesalazine; tubulointersticial nephritis.RESUMO A doença inflamatória intestinal e as terapêuticas que lhe estão associadas podem afetar o rim de várias formas. A nefrite tubulointersticial é uma complicação rara, mas potencialmente grave inerente à terapêutica com mesalazina. Há alguns casos descritos na literatura. Os autores descrevem os dois primeiros casos de nefrite tubulointersticial associados ao uso de mesalazina, recentemente diagnosticados no nosso Serviço; um deles num doente com colite ulcerosa, outro num doente com doença de Crohn. Apresentam uma revisão da literatura sobre o atingimento renal na doença inflamatória intestinal. Os casos de nefrotoxicidade associada à mesalazina devem continuar a ser descritos para permitir estabelecer uma incidência mais precisa desde efeito adverso. A monitorização da função renal durante o tratamento é simples, barata e pode ajudar a diagnosticar precocemente esta complicação.
Palavras-Chave: Doença inflamatória intestinal; doença renal; mesalazina; nefrite tubulointersticial.
INTRODUCTION
Inflammatory bowel disease (IBD) is associated with a large spectrum of kidney disease. The kidney may be an extra-intestinal target of IBD. It could be affected by the secondary complications of bowel inflammation, such as stones and amyloidosis; there are some reported associations of glomerular disease1.
On the other hand, some drugs used in its treatment, including mesalazine, may be associated with tubulointersticial renal disease1-3. Acute and chronic interstitial nephritis are rare but serious complications of longer-term mesalazine use4. There are some reports in the literature1,5-7. This adverse drug reaction is likely idiosyncratic. The clinical presentation is usually non-specific and more frequent in men than in women8,9.
We report two cases of tubulointersticial nephritis (TIN) associated with mesalazine treatment, recently diagnosed in our department. The first refers to a patient with ulcerous colitis (UC) and the second one to a patient with Crohns disease (CD). Then the authors present a literature review about renal involvement in IBD.
CASE REPORTS
Case 1
A man with a past history of controlled hypertension and healed prostatic neoplasm had a diagnosis of UC at 59 years old (in 2011). At this time, he had 1.05 mg/dl serum creatinine and no urinalysis abnormalities, namely proteinuria, leukocyturia or erythrocyturia.
Due to disease activity, he began treatment with oral mesalazine (1600 mg/day) and prednisolone.
One year later, he had a 1.9 mg/dl serum creatinine. At that moment, the UC was in activity and mesalazine dose was increased (1.6 to 3.6 g/day). Three months later, the serum analysis showed an increase in creatinine (2.1 mg/dl) and urinalysis showed proteinuria (100 mg/dl), leukocyturia (25-50 WBC/field) and erythrocyturia (25-50 RBC/field). At Nephrology evaluation, IBD was in clinical remission. The patient had no symptoms and no abnormalities on physical examination. Initial laboratory results were: normal haemoglobin, normal leucocyte, eosinophils and platelets count; blood urea nitrogen 80 mg/dl, serum creatinine 2.1mg/dl; normal serum sodium, potassium, calcium and phosphorus. Urinalysis revealed: an albumin/creatinine ratio 1420 mg/g, a proteincreatinine ratio 1.5g/g, erythrocyturia and leukocyturia.
The urine culture was negative. Serology revealed normal C3, C4, IgG, IgM, IgG; negative anti-nuclear antibody (ANA); positive anti-saccharomyces cerevisiae (ASCA) test. Renal ultrasound did not reveal any hydronephrosis, nephrolithiasis or cortical atrophy; both kidneys were of normal size. Kidney biopsy showed an acute interstitial nephritis with linfomononuclear cells and a granulomatous reaction without central necrosis (Figs. 1 and 2). The vessels were normal; there was 40% fibrosis and 20% tubular atrophy. Immunofluorescence with auto-antibodies to albumin, C3, C4, C1q, IgA, IgM and IgG was positive to mesangial and peripheral deposits for C3 and linear IgA(+).
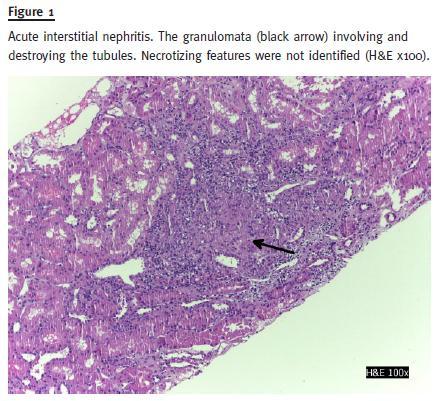
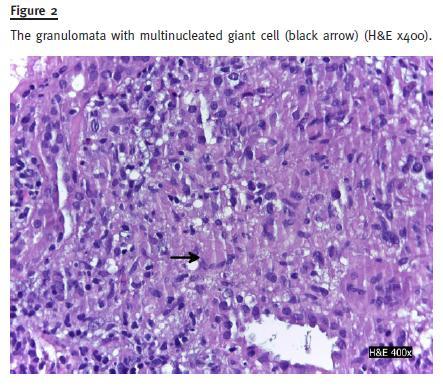
Mesalazine was discontinued and azathioprine was introduced (2 mg/Kg) in combination with prednisolone (5mg/day). Two months later, serum creatinine decreased to 1.5 mg/dl and urinalysis revealed no white blood cells/field and 2-5 red blood cells/field; proteinuria/creatinine ratio decreased to 0.3 g/g.
Case 2
A man with Crohns disease and no other relevant medical history was sent to Nephrology consultation for chronic kidney disease. Inflammatory bowel disease had been diagnosed when he was 35 years old (in 2009). At that moment, he had a serum creatinine of 0.8mg/dl, with no abnormalities in urinalysis. He started oral mesalazine (4g/day) and prednisolone (60 mg/day) with a gradual decrease of doses and clinical remission. One year later he had 1.48 mg/dl serum creatinine. After two years, he had 1.80 mg/dl serum creatinine and leukocyturia (25-50 WBC/field) in urinalysis. Mesalazine was discontinued and he started azathioprine (AZA) (1mg – 2mg/kg/day) with poor control of IBD activity and need to increase the corticosteroids (CT) dose (1mg/kg/day) with a gradual decrease and clinical remission. At Nephrology evaluation, the patient had only gastro-intestinal symptoms (abdominal discomfort and diarrhoea).
Abdomen examination revealed increased bowel sounds and diffuse tenderness to palpation with no other abnormalities. Initial laboratory results were: haemoglobin 13g/dl; normal leucocyte, eosinophils and platelets count; blood urea nitrogen 120, serum creatinine 2,6mg/dl and a normal serum sodium, potassium, calcium and phosphorus. Urinalysis revealed: albumin, red blood cells (10 RBC/field) and white blood cells (25-50 WBC/field). The urine culture was negative. Serology revealed normal C3, C4, IgG, IgM, IgG; negative anti-nuclear antibody (ANA); positive ASCA test. Renal ultrasound was normal. Kidney biopsy showed glomerulosclerosis (30%) and a marked interstitial inflammatory infiltrate with eosinophils in 30% of cortex extension and mononuclear cells (Fig. 3) Vessels were normal. There was 50% cortical atrophy (Fig. 4). Immunofluorescence with auto-antibodies to albumin, C3, C4, C1q, IgA, IgM and IgG was negative.
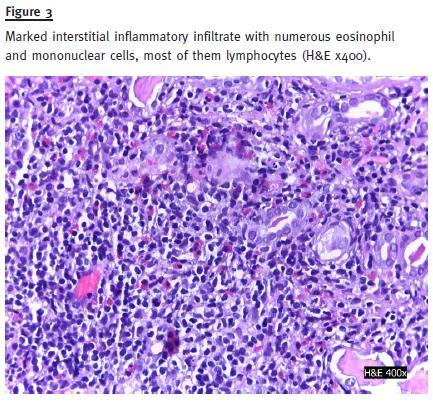
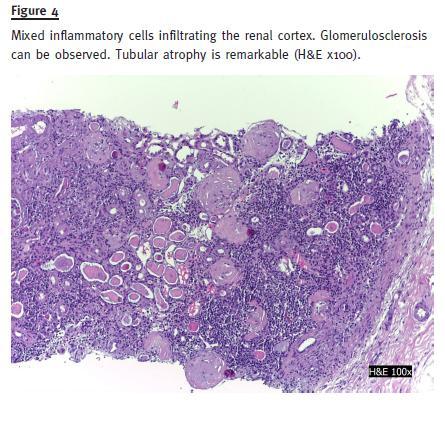
Three months after stopping mesalazine, serum creatinine decreased to 1.5 mg/dl and urinalysis revealed no white or red blood cells. Six months after treatment with AZA and lower doses of prednisolone (5 mg/day), the IBD became active. AZA was increased to 2 mg/kg/day and CT to 1 mg/kg/day with clinical remission. AZA was stopped two months later due to pancytopenia. The patient was proposed for further treatment with anti-TNF due to refractory IBD.
DISCUSSION
Aminosalicylates compounds are widely used in acute phase and remission maintenance therapy of IBD10. Mesalazine also known as 5-aminosalicylic acid (5-ASA), is a derivative of salicylic acid and it is the active moiety of sulfasalazine (without the adverse effects attributed to the latter one). A number of reports have linked oral mesalazine therapy to tubulointersticial nephritis. Although this association is currently well established there are no guidelines for monitoring renal function in patients who are receiving this treatment11.
The first reports were published in 1989, and in 1990 the Committee on Safety of Medicines (CSM) issued a warning of nephrotoxic reactions to mesalazine.
From 1985 to 1997, the CSM had received 35 reports implicating the drug in interstitial nephritis.
In recent editions of nephrology textbooks, mesalazine is one of the drugs that could be associated with acute and chronic interstitial nephritis9,12,13.
The true incidence of this problem is not really known. Primas et al. showed that renal impairment, defined as any increase in serum creatinine, may occur in up to one in 100 patients treated with this drug14. The incidence of clinically significant interstitial nephritis was estimated to be less than one in every 500 patients treated, when serum creatinine is monitored regularly. In a larger study of 223 outpatients with IBD, Schreiber et al. found an increased prevalence of tubular proteinuria in patients who were receiving high dosages of 5-ASA11. All reports of interstitial nephritis in IBD patients are associated with drugs used in its treatment9.
The nephrotoxicity associated with mesalazine takes the form of an acute or an indolent chronic and progressive interstitial nephritis. It can be severe and lead to chronic kidney disease14,15. The development of tubulointersticial nephritis is independent of treatment duration. Fifty per cent of patients present within the first 12 months of starting therapy but some are diagnosed within 5 years after beginning treatment1. It has been suggested that undercurrent steroid therapy used in the IBD treatment may delay presentation.
Signs and symptoms are scanty and non-specific. Patients may be asymptomatic, such as those who presented. A minority of patients have systemic symptoms like fever, myalgia, anorexia and weight loss16. An increase in serum creatinine is a late event but may be the first evidence of nephrotoxicity.
Reagent strip urinalysis can be normal, or positive to blood and protein. Proteinuria can be mild to heavy and can be influenced by IBD activity. Sterile pyuria may be present. Eosinophilia has been recorded but it is not a specific finding16-18.
The pathogenic basis of mesalazine associated interstitial nephritis is not completely known. In the acute stage of the disease, there is a mixed inflammatory cell infiltrate composed of B and T lymphocytes and macrophages, with interstitial oedema, which is comparable to that seen in renal allograft acute cellular rejection. It has been suggested that this represents a delayed cell-mediated hypersensitivity response16-20. Only a minority of patients manifest a systemic type-I hypersensitivity reaction with fever and eosinophilia8.
In the chronic stage, the cellular infiltrate is accompanied by interstitial fibrosis and tubular atrophy, as it happened in our second case. The extent of these hallmarks means an irreversible renal impairment and is predictive of progression to chronic renal failure21.
Mesalazine is structurally related to salicylic acid; salicylates inhibit the synthesis of intra-renal prostaglandins, which are vasoactive mediators of intra-renal blood flow, uncouple oxidative phosphorylation in mitochondria and inhibit the pentose phosphate shunt rendering the kidney susceptible to oxidative damage22-25. All of these effects cause a disturbance in intra-renal blood flow and promote local tissue hypoxia.
Direct tubular toxicity in addition to tissue hypoxia is also suggested20,21. After reperfusion injury, there is an inflammatory response that may contribute to chronic inflammation and interstitial fibrosis23-26.
Due to the small number of cases described and the inability to determine the degree of systemic absorption, is impossible to say if nephrotoxicity is dose-related. Tubulointersticial nephritis has been reported in patients taking doses of mesalazine as low as 400 mg/day. The systemic bioavailability is determined by the route of administration and by the location and extent of the IBD. Systemic absorption of rectal mesalazine is approximately 50% of oral mesalazine; there is no report of significant nephrotoxicity in patients using only rectal formulation; the nephrotoxicity could be an idiosyncratic effect20,24.
In the Dutch Pentasa Study, over 150 subjects were screened prospectively for renal toxicity. They were randomized to receive two dosages of oral slow-release mesalazine (1.5 vs. 3.0 g per day), and followed-up for 12 months. There were no differences in toxicity or compliance rate between the high and low dose groups. Two patients (1.3%) developed modest, reversible renal impairment, but only one of them had biopsy proven interstitial nephritis21.
Mesalazine should be withdrawn when the renal impairment is not explained by other factors. A renal biopsy provides diagnostic and prognostic information.
It is indicated in subjects with impaired renal function that is not explained by other factors, in which renal function does not recover to baseline following withdrawal of the drug, or in those who present with atypical features such as nephrotic range proteinuria, which may suggest an alternative diagnosis.
The response to drug withdrawal is variable. It leads to recovery of renal function in 85% of cases where the diagnosis is made within 10 months of starting treatment. When the diagnosis is delayed beyond 18 months from the start of treatment only one-third of cases show some recovery of renal function and this is usually only partial20-23. Active chronic tubulointersticial nephritis was described in biopsies from patients in whom mesalazine has been withdrawn several months prior to biopsy19,22,26.
Steroids and azathioprine have been used for IBD treatment with a good response, but this depends of level on the established chronicity lesions in renal biopsy. Nevertheless, a trial of high dose steroid (1 mg/day/kg, maximum 60 mg/day for up to 3 months) is recommended in patients whose renal function does not respond to drug withdrawal alone. This may be preceded by pulsed intravenous methylprednisolone in severe cases (0.5 to 1 g on three successive days).
Both our patients had improvement in renal function after discontinuation of the drug and none of them made an intensive course of corticosteroid therapy. In both cases, the low-dose of corticosteroids may have contributed to the improvement of the renal function.
Other manifestations of renal disease in patients with inflammatory bowel disease (IBD)
Extraintestinal manifestations (EIM) of IBD are common. These manifestations can involve nearly any organ system – including the renal but also the musculoskeletal, dermatologic, hepatopancreatobiliary, ocular, and pulmonary systems9. Symptoms of renal impairment are not always specific and since the underlying bowel disease is preponderant, renal function deterioration may be underestimated10.
Renal or urinary complications can occur in 4%-23% of patients; the most common manifestations are kidney stones, enterovesical fistulas and ureteral obstruction. Patients with IBD have a 10 to 100 time higher risk of nephrolithiasis compared with the general population. The risk is higher in adults than in children and in patients with CD. Kidney stones in IBD are composed primarily of calcium oxalate or uric acid26.
Fistulas between the gastrointestinal tract and the urinary system are uncommon, occuring in 1%-8%; they are more common in patients with ileal or ileocecal disease than in patients with colonic disease27.
Morphological changes in the glomeruli – podocyte effacement, mesangial deposition of immunoglobulin and complement have been well described. There are also reported associations with membranous glomerulonephritis, rapidly progressive glomerulonephritis, mesangiocapillary glomerulonephritis, IgA nephropathy (as in our first case), and thin basement membrane disease1,9,26-31. Glomerulonephritis presenting as nephrotic syndrome in patients with IBD is rare16. Tubular damage was seen in 31% of CD and in 23% of UC patients who were not on ASA therapy. Granulomatous interstitial nephritis, interstitial nephritis with hyperoxalouria and renal tubular acidosis have also been reported32-35.
Secondary amyloidosis (AA amyloidosis) is a rare but serious complication that may occur in chronic inflammation diseases, including IBD, mainly longstanding Crohns disease. The reported prevalence of secondary amyloidosis in IBD patients varies from 0.5 to 29% in CD and from 0 to 0.4% in UC with a higher prevalence at autopsy16,29.
The majority of patients have renal involvement with asymptomatic proteinuria or manifestations of nephrotic syndrome. Differential diagnosis of primary systemic amyloidosis presenting as regional enteritis should always be considered. The response of IBD-related secondary amyloidosis to treatment is variable. It is reported that medical or surgical treatment has stabilized or improved renal disease but there exist reports suggesting no benefit with them30.
There is only a retrospective review study to date that examined the histopathology of 83 renal biopsies performed in IBD patients. The authors present 45 and 38 cases CD and UC, respectively. IgA nephropathy was the most common finding (24%), followed by interstitial nephritis (19%), arterionephrosclerosis (12%), acute tubular injury (8%), proliferative GN (7%), and minimal-change disease (5%). Of the 16 cases of interstitial nephritis, nine (56%) had current or recent past exposure to aminosalicylates, including all cases of granulomatous interstitial nephritis17.
There are no specific guidelines for surveillance of renal function. Frandsen et al. recommend measurement of serum creatinine prior to the start of mesalazine treatment and every 6 months thereafter4.
CONCLUSIONS
These are the first two cases of TIN associated with mesalazine identified by renal biopsy in our Hospital.
Management of mesalazine-induced interstitial nephritis includes discontinuation of drug and sometimes a trial of corticosteroid therapy. In both cases, the history of inflammatory bowel disease, mesalazine use and the histopathology, support the diagnosis of a drug-associated renal injury.
It is very important to recognize renal impairment early, to prevent the progression to an end-stage renal disease. We suggest regular monitoring of renal function (serum creatinine and urinalysis) in patients treated with mesalazine; it is simple, inexpensive and it is the only way to diagnose new cases early.
Further epidemiological data are needed to clarify the causal mechanisms of nephrotoxicity. Similar data should be reported to estimate the true incidence of this serious adverse effect. These cases show us the need to create some protocols for routine renal function surveillance in IBD patients.
References
1. Co ML, Gorospe EC. Pediatric case of mesalazine-induced interstitial nephritis with literature review. Pediatr Int 2013;55(3):385-387. [ Links ]
2. World MJ, Stevens PE, Ashton MA, Rainford DJ. Mesalazine-associated interstitial nephritis. Nephrol Dial Transplant 1996;11(4):614-621. [ Links ]
3. Tadic M, Grgurevic I, Scukanec-Spoljar M, et al. Acute interstitial nephritis due to mesalazine. Nephrology (Carlton) 2005;10(2):103-105. [ Links ]
4. Frandsen NE, Saugmann S, Marcussen N. Acute interstitial nephritis associated with the use of mesalazine in inflammatory bowel disease. Nephron 2002;92(1):200-202. [ Links ]
5. Bonet J, Vaquero M, Bayés B, Romero R. Renal involvement in patients with chronic inflammatory intestinal disease treated with mesalazine. How to prevent its nephrotoxicity?] Med Clin (Barc) 1999;113(5):199. [ Links ]
6. Gorospe EC, Leggett CL. Mesalazine-induced interstitial nephritis in a patient with ulcerative colitis. BMJ Case Rep 2012. doi: 10.1136/bcr.12.2011.5351. [ Links ]
8. Bosque López MJ, Garrido Durán C, Ginard Vicens D, et al. [Severe tubulo-interstitial nephritis secondary to 5-ASA]. Gastroenterol Hepatol 2009;32(1):29-31. [ Links ]
9. Calviño J, Romero R, Pintos E, et al. Mesalazine-associated tubulo-interstitial nephritis in inflammatory bowel disease. Clin Nephrol 1998;494:265-267. [ Links ]
10. Gisbert JP, González-Lama Y, Maté J. 5-Aminosalicylates and renal function in inflammatory bowel disease: a systematic review. Inflamm Bowel Dis 2007;13(5):629-638. [ Links ]
11. Gisbert JP, Luna M, González-Lama Y, et al. Effect of 5-aminosalicylates on renal function in patients with inflammatory bowel disease: 4-year follow-up study. Gastroenterol Hepatol 2008;31(8):477-484. [ Links ]
12. Corrigan G, Stevens PE. Review article: interstitial nephritis associated with the use of mesalazine in inflammatory bowel disease. Aliment Pharmacol Ther 2000;14(1):1-6. [ Links ]
13. De Broe ME, Stolear JC, Nouwen EJ, Elseviers MM. 5-Aminosalicylic acid (5-ASA) and chronic tubulointerstitial nephritis in patients with chronic inflammatory bowel disease: is there a link? Nephrol Dial Transplant 1997;12(9):1839-1841. [ Links ]
14. Brouillard M, Gheerbrant JD, Gheysens Y, et al. [Chronic interstitial nephritis and mesalazine: 3 new cases?] Gastroenterol Clin Biol 1998;22(8-9):724-726. [ Links ]
15. Primas C, Novacek G, Schweiger K, et al. Renal insufficiency in IBD-prevalence and possible pathogenetic aspects. J Crohns Colitis 2013;7(12):e630-e634. [ Links ]
16. Skalova S, Dedek P, Pozler O, Podhola M. Mesalazine-induced interstitial nephritis. Ren Fail 2009;31(2):159-161. [ Links ]
17. Oikonomou K, Kapsoritakis A, Eleftheriadis T, Stefanidis I, Potamianos S. Renal manifestations and complications of inflammatory bowel disease. Inflamm Bowel Dis 2011;17(4):1034-1045. [ Links ]
18. Ambruzs JM, Walker PD, Larsen CP. The histopathologic spectrum of kidney biopsies in patients with inflammatory bowel disease. Clin J Am Soc Nephrol 2014;9(2): 265-270. [ Links ]
19. Colvin RB, Traum AZ, Taheri D, Jafari M, Dolatkhah S. Granulomatous interstitial nephritis as a manifestation of Crohn disease. Arch Pathol Lab Med 2014;138(1):125-127. [ Links ]
20. Cury DB, Moss AC, Schor N. Nephrolithiasis in patients with inflammatory bowel disease in the community. Int J Nephrol Renovasc Dis 2013;29;6:139-142. [ Links ]
21. Fockens P, Mulder CJ, Tytgat GN, et al. Comparison of the efficacy and safety of 1.5 compared with 3.0 g oral slow-release mesalazine (Pentasa) in the maintenance treatment of ulcerative colitis. Dutch Pentasa Study Group. Eur J Gastroenterol Hepatol 1995;7(11):1025-1030. [ Links ]
22. Lewis B, Mukewar S, Lopez R, Brzezinski A, Hall P, Shen B. Frequency and risk factors of renal insufficiency in inflammatory bowel disease inpatients. Inflamm Bowel Dis 2013;19(9):1846-1851. [ Links ]
23. Sattianayagam PT, Gillmore JD, Pinney JH, et al. Inflammatory bowel disease and systemic AA amyloidosis. Dig Dis Sci 2013;58(6):1689-1697.
24. Huang V, Mishra R, Thanabalan R, Nguyen GC. Patient awareness of extraintestinal manifestations of inflammatory bowel disease. J Crohns Colitis 2013;1; 7(8):e318-e324. [ Links ]
25. Firwana BM, Hasan R, Chalhoub W, et al. Nephrotic syndrome after treatment of Crohns disease with mesalamine: Case report and literature review. Avicenna J Med 2012;2(1):9-11.
26. Zallot C, Billioud V, Frimat L, Faure P, Peyrin-Biroulet L, with the CREGG (Club de Reflexion des cabinets et Groupes dHépato-Gastroentérologie). [ Links ]
27. 5-Aminosalicylates and renal function monitoring in inflammatory bowel disease: a nationwidesurvey. J Crohns Colitis 2013;7(7):551-555. [ Links ]
28. Katsanos KH, Tsianos EV. The kidneys in inflammatory bowel disease. Ann Gastroenterol 2002;15(1):41-52. [ Links ]
29. Pardi DS, Tremain WJ, Sandborn WJ, Mc Carthy JT. Renal and urologic complications of inflammatory bowel disease. Am J Gastroenterol 1998;93(4):504-514. [ Links ]
30. Polci R, Mangeri M, Faggiani R, Cristi E, Ranalli TV, Feriozzi S. Granulomatous interstitial nephritis in a patient with Crohns disease. Ren Fail 2012; 34(9):1156-1159. [ Links ]
31. Choi JY, Yu CH, Jung HY, et al. A case of rapidly progressive IgA nephropathy in a patient with exacerbation of Crohns disease. BMC Nephrol 2012;13:84. [ Links ]
32. Basturk T, Ozagari A, Ozturk T, Kusaslan R, Unsal A. Crohns disease and secondary amyloidosis: early complication? A case report and review of the literature. J Ren Care 2009;35(3):147-150. [ Links ]
33. Levine JS, Burakoff R. Extraintestinal manifestations of inflammatory bowel disease. Gastroenterol Hepatol (NY) 2011;7(4):235-241. [ Links ]
34. Wilcox GM, Aretz TH, Roy MA, Roche JK. Glomerulonephritis associated with inflammatory bowel disease. Report of a patient with chronic ulcerative colitis, sclerosing cholangitis, and acute glomerulonephritis. Gastroenterology 1990;98(3):786-791. [ Links ]
35. Kreisel W, Wolf LM, Grotz W, Grieshaber M. Renal tubular damage: an extraintestinal manifestation of chronic inflammatory bowel disease. Eur J Gastroenterol Hepatol 1996;8(5):461-468. [ Links ]
Drª Andreia Campos
Department of Nephrology, Centro Hospitalar do Porto – Hospital Geral de Santo António
Largo Prof. Abel Salazar 4099-001 PORTO – Portugal
E-mail: andcriscampos@hotmail.com
Conflicts of Interest: None declared.
Received for publication: 10/03/2015
Accepted in revised form: 22/05/2015














