Serviços Personalizados
Journal
Artigo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Acessos
Acessos
Links relacionados
-
 Similares em
SciELO
Similares em
SciELO
Compartilhar
Portuguese Journal of Nephrology & Hypertension
versão impressa ISSN 0872-0169
Port J Nephrol Hypert vol.30 no.2 Lisboa jun. 2016
ORIGINAL ARTICLE
Clinicopathological profile and prognosis of idiopathic membranous nephropathy in adults: a developing country perspective
Syed Abbas Hyder Rizvi, MD, Senior Lecturer1,Rubina Naqvi, MD, Professor of Nephrology1, Ejaz Ahmed, FRCP, Professor of Nephrology1, Fazal Akhter, FRCP, Professor of Nephrology1, Khawar Abbas, FCPS, Assistant Professor2, Muhammed Mubarak, FCPS, Professor of Pathology3
1 Departments of Nephrology
2Immunology
3 Histopathology Sindh Institute of Urology and Transplantation, Karachi, Pakistan
ABSTRACT
Background: There is no study available from Pakistan on the longitudinal course of membranous nephropathy (MN). We aimed to analyse the clinicopathological profile, course, response to treatment and outcome of the disease in our setup. Methods: All consecutive adult patients (≥18 years) with MN on renal biopsy and on regular follow‑up were included. Relevant data items were retrieved from case files and biopsy reports. The outcome variables included doubling of baseline serum creatinine or the development of end‑stage renal disease (ESRD). Results: Of a total of 102 patients, 82 (80.4%) were males; mean age was 28.9±11.4 years. Peripheral oedema was present in 86 (84.3%), nephrotic‑range proteinuria in 80 (78.4%), microscopic haematuria in 25 (24.5%), hypertension in 42 (41.2%), and renal dysfunction in 13 (12.7%) patients at the time of presentation. The mean follow‑up period was 3±2 years. Overall, 22 (21.5%) subjects went into remission and a similar number experienced doubling of serum creatinine. Both conservative and immunosuppressive treatment groups had similar duration of follow‑up. The proportion of patients with remission and doubling of serum creatinine was similar between the two groups (p=0.70 and p=0.91 respectively), while the proportion of patients progressing to ESRD was significantly higher in the group treated conservatively (p=0.03). Baseline proteinuria and serum creatinine were predictive of remission (p=0.04 and p=0.05 respectively). Conclusions: In conclusion, untreated MN has a higher risk of progression to ESRD. Baseline proteinuria and serum creatinine predict response to treatment. Randomised controlled trials are needed to confirm the effects of immunosuppressive treatment.
Key words: Adults, end‑stage renal disease, membranous nephropathy, nephrotic syndrome, proteinuria.
INTRODUCTION
Membranous nephropathy (MN) has traditionally been considered the leading cause of idiopathic nephrotic syndrome (INS) in adults, which still holds true for Caucasian populations1. More recent studies, particularly from non‑Caucasian populations, however, have shown that focal segmental glomerulosclerosis (FSGS) has overtaken MN as the most common cause of INS in adults in many geographic areas2,3. In contrast to other primary glomerular diseases, such as IgA nephropathy (IgAN) and FSGS, the incidence rate of MN has remained constant over the past three decades1.
The presenting features, clinical course, response to treatment and outcome of MN have been extensively studied throughout the world4‑8.
At presentation, 60 to 70% of patients have NS. Microscopic haematuria is common (30 to 40%), but macroscopic haematuria and red cell casts are rare. The great majority of patients are normotensive, with hypertension present in 10 to 20% of the cases at initial presentation9‑12.
MN is a chronic disease, with spontaneous remissions and relapses. One third of patients progress to end‑stage renal disease (ESRD). Thus, in the United States and Europe, MN remains one of the leading causes of ESRD among the primary glomerulonephritides8.
Determining the prognosis is critical in making the decision regarding when and how to treat, i.e., conservative versus immunosuppressive treatment in patients with idiopathic MN13‑18.
There is very little data exclusively on MN from this part of the world, especially one delineating the longitudinal analysis of the disease. Occasional studies from Pakistan reported in the past were based on LM or at most IF examination in some cases and thus did not reflect the true picture of MN from this country19.
Moreover, all of the studies were cross‑sectional in nature and no outcome data was provided.
This study was designed to determine the clinical presentation, laboratory and pathological features, treatment protocols and long‑term renal prognosis of idiopathic MN in adults at our centre.
MATERIALS AND METHODS
All patients visiting the adult nephrotic outpatient department (OPD) of Sindh Institute of Urology and Transplantation (SIUT) from January 1994 to December 2004 with significant proteinuria and MN on renal biopsy and for whom regular follow‑up data was available were included in the study. Written informed consent was obtained from the participants and the research was conducted in accordance with the tenets of the Declaration of Helsinki. Detailed history and physical examination were carried out and recorded on initial visit and at each follow‑up visit. All patients underwent quantification of 24‑hour urinary protein excretion. The following baseline biochemical tests were performed in all patients; serum urea, creatinine, electrolytes, total proteins, and serum albumin.
All patients had complete blood count (CBC) done on the first visit as was the coagulation profile. Relevant serology was done at baseline including complement levels (C3 and C4) and immunoglobulins (Ig) including IgG, IgM and IgA. Hepatitis B surface antigen (HBsAg) and anti‑hepatitis C virus (HCV) antibody, anti‑nuclearantibody (ANA), anti‑ds DNA, rheumatoid arthritis (RA) factor and antistreptolysin titre (ASOT) were also done. Patients with any history or laboratory evidence suggestive of systemic diseases were excluded.
Hypertension was defined as systolic blood pressure (BP) measurements of >150 mmHg and diastolic of >90 mmHg on at least two consecutive occasions or the need for antihypertensive treatment. Anaemia was defined as haemoglobin (Hb) < 12 g/dl in males and < 11 g/dl in females. Renal dysfunction at presentation was defined as serum creatinine >1.5 mg/dl in males and >1.2 mg/dl in females. ESRD was defined as the need for permanent renal replacement therapy1,10.
Standard doses and duration of treatment of immunosuppressants were employed10. Treatment groups were divided into conservative and immunosuppressive groups. Complete remission (CR) was defined as proteinuria <0.2 g/day with normal renal functions, and partial remission (PR) as 50% reduction of proteinuria from the baseline but >0.2 g/day.
Renal biopsy analysis
Standardised procedure was used for procuring and processing the renal biopsies as described in detail in our previous studies20,21. At our centre, two cores of native tissue are routinely obtained for complete pathologic evaluation; one core is processed for light microscopy (LM) and is fixed in 10% neutral buffered formalin.
The other core is divided into two pieces under a dissection microscope such that both pieces contain glomeruli.
Of these, one piece is fixed in 4% glutaraldehyde and refrigerated for electron microscopy (EM) and the other piece is put into an OCT‑containing capsule and is snap frozen in liquid nitrogen for immunofluorescence (IF) study. For LM, routinely 10 serial sections are cut at a thickness of 2μm with levels 1, 5, and 10 stained by haematoxylin and eosin (H&E), level 7 with trichrome, level 8 with periodic acid‑Schiff (PAS) stain and level 9 with Jones methenamine silver (JMS) stain; levels 2, 3, 4 and 6 are left unstained for further tests as and when required.
Immunofluorescence (IF)
Tissue specimens for IF were snap‑frozen in liquid nitrogen and cut on cryotome. The sections were then stained by the direct or one‑step method using fluorescein isothiocyanate (FITC)‑conjugated antisera mono‑specific for IgG, IgA, IgM, C3 and C1q (Dako, Glosstrup, Demark). The slides were then visualized under the fluorescence microscope in dark and graded semiquantitatively as strong, moderate or weak and distribution described as membranous or mesangial in a granular or linear pattern. IF results were obtained from the original biopsy reports.
Electron microscopy (EM)
Standardised procedures were used for the ultrastructural study of the renal biopsy. Briefly, for EM, the specimens were fixed in 3% buffered glutaraldehyde, postfixed in 1% osmium tetroxide and embedded in EPON resin. Ultrathin sections were cut on ultramicrotome.
Sections were stained on girds with lead citrate and uranyl acetate and examined under the EM.
Statistical Analysis
Data items were entered and analysed using the Statistical Package for the Social Sciences (SPSS) for Windows version 10.0 (SPSS, Chicago, IL, USA). Descriptive statistics such as mean ± standard deviation (SD) or median ± range for continuous variables such as age, serum creatinine and proteinuria and numbers (percentages) were used to describe the proportion of categorical variables such as sex and the responses to treatment. Chi‑squared and Fishers exact tests were used to compare the frequency of different parameters among the various groups. A p value <0.05 was considered significant. Survival curves were plotted by the Kaplan‑Meier method.
RESULTS
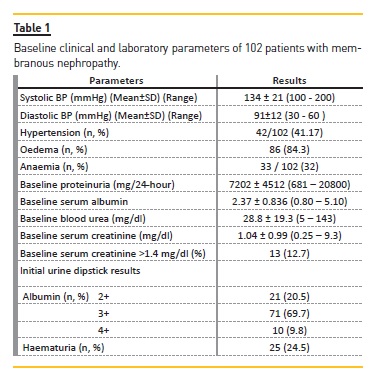
During the study period, a total of 102 patients from adult nephrotic OPD were diagnosed with MN with at least one year of follow‑up and these formed the study population. Of these, 82 (80.4%) were male; the majority of patients was young with a mean age of 28.9±11.4 years, and only five patients were aged between 50‑65 years. Their presenting features and initial clinical and laboratory parameters are shown in Table 1. As is apparent from this table, 42 (41.2%) patients were hypertensive on presentation with a mean systolic BP of 134±21 mmHg and diastolic of 91±12 mmHg. Peripheral oedema was present in 86 (84.3%) patients, while 33 (32%) were anaemic. Nephrotic‑range proteinuria was present in 80 (78.4%) subjects. Microscopic haematuria was detected in 25 (24.5%) patients. Thirteen patients (12.7%) had renal dysfunction at the time of presentation.
The mean duration of illness before presentation was 12.2±14.6 months. The mean follow‑up period was 3±2 years. At the end of follow‑up, overall, 22 (21.5%) subjects went into remission and a similar number experienced doubling of serum creatinine from the baseline.
During the study period, 50 (49%) patients were managed conservatively, while the remaining 52 (51%) were treated with steroids with or without cyclophosphamide.
The mean duration of steroid treatment was 18.53±16.077 weeks and that of cyclophosphamide was 13±3.52 weeks. Both conservative and immunosuppressive treatment groups had almost similar duration of follow‑up.
The proportion of patients with remission and doubling of serum creatinine from the baseline values was almost similar with no statistically significant difference between the two groups (p=0.70 and p=0.91 respectively), while the proportion of patients progressing to ESRD was significantly higher in the group treated conservatively (p=0.03), as shown in Table 2.
The immunosuppressive group was further divided into steroids alone and steroid plus cyclophosphamide groups. The frequency of remission was marginally better among patients in the steroids group than those treated with combined steroids and cyclophosphamide.
Remission was seen in 28% of steroid treated patients and 19% of steroid plus cyclophosphamide treated patients (p=0.70), as depicted in Table 3. When patients with deteriorating renal functions (n=22) were compared with those with stable renal functions (n=80), the only variable which showed marginal significance was the degree of baseline proteinuria (Table 4).
Similar parameters were compared among patients who achieved CR with those who did not achieve remission.
Baseline degree of proteinuria and serum creatinine emerged as the significant factors (p=0.04 and p=0.05 respectively) for predicting remission (Table 5).
Table 6 shows that patients with heavy proteinuria i.e., > 10g/24‑hours had the lowest remission rate and highest rate of doubling of serum creatinine. None of the patients with proteinuria <3 g/24‑hours progressed to ESRD. Only two patients among those who eventually developed ESRD had elevated baseline creatinine levels. The remaining patients had normal serum creatinine at the time of presentation. On the other hand, the majority of patients with renal dysfunction at presentation did not progress to ESRD.
DISCUSSION
This is the first large and longitudinal study of idiopathic MN from Pakistan. Idiopathic MN remains the most common cause of NS in older adults in the world.
Because of its frequency, it occupies a higher position in the overall causes of ESRD. The natural history of idiopathic MN has been documented in many studies with wide variation in its outcome [22, 33]. In our study, majority of patients were young, which is in contrast to western studies. The reasons for this discrepancy in age are not known. Patients older than 60 years with MN have higher risk of progression towards chronic renal failure24. In the present study, no significant difference was found in the mean age of patients who exhibited doubling of serum creatinine at the end of the study. This observation might be related to the fact that the population studied comprised fewer patients of older age; in fact, none was older than 65 years.
Gender has been reported to influence the outcome in many reports25,26. In our study, there was significant male preponderance at all time periods and we were unable to see any advantage of female gender for remission or preservation of renal function. The ratio of male to female was the same at 4:1 in patients who progressed to ESRD as it was in the whole group of studied population.
The degree of proteinuria is another consistently reported factor associated with the rate of complete or partial remission. Some investigators have found that the amount of proteinuria at presentation could predict the outcome of MN27. Others have reported no impact of proteinuria on progression of the disease. Cattran and colleagues designed a model for risk of progression in IMN, based on the severity of proteinuria over a six‑month observation period28. This model incorporated a large number of studies of Canadian, Italian and Finnish patients and on the basis of these studies, Cattran et al. reported that patients with baseline normal serum creatinine and proteinuria less than 4 g/day over six months can be considered as low risk in comparison to normal serum creatinine and proteinuria 4‑8 g/day. Furthermore abnormal serum creatinine and >8 g/day of proteinuria carries a high risk of progression28.
In our study, the degree of proteinuria was found to have a significant effect on the rate of remission (p=0.04) but fell short of significance regarding worsening of renal function (p=0.91).
There is consensus that increased serum creatinine at presentation can predict progression towards ESRD [29‑33]; however, it should be kept in mind that in some patients, renal impairment could be due to haemodynamic factors. These factors are reversible and can change the overall results. In our observation, a lower initial serum creatinine was seen in patients who subsequently went into remission (p=0.05).
As we are a tertiary care unit, patients often consult our institute after a long delay and in the present studied population, the duration of symptoms varied from one month to 7.5 years, but we found no statistically significant difference in the rate of remission or worsening in renal function in terms of duration of disease before presentation (p=0.98 and 0.98 respectively).
It has been reported that patients who enter CR tend to maintain stable renal function in the long‑term.
In a review of literature, only 1 of 157 patients who achieved CR developed ESRD30‑33. Passerini et al. reported stable serum creatinine at 10 years in 33 patients with MN who had shown CR31. Some patients in this study developed relapse, however; at 10 years, 73% were still in remission.
PR with residual non‑nephrotic proteinuria also carries a favourable prognosis. Ponticelli in one report has shown a slower rate of progression towards ESRD in patients with PR with a fairly long duration of follow‑up in a group of 37 patients32. In contrast, persistent heavy proteinuria during follow‑up increases the likelihood of progressive renal function loss33‑38.
The treatment of idiopathic MN has been a topic of controversy over the past four decades. A high percentage of cases achieve spontaneous complete or partial remission in the absence of steroidal or any other immunosuppressive therapy. Both benign and aggressive forms of the disease are defined33‑35.
One area of controversy is the inability to predict patients who will progress to ESRD if a conservative approach is adopted.
Schieppati et al., reported the results of patients who did not receive any form of immunosuppressants including steroids at any time8. The probability that ESRD would not develop was 88% after five years and 73% after eight years. The results of this study and a few other published reports that allow the calculation of survival curves in untreated patients indicate that the probability of maintaining renal function five years after the onset of disease ranges from 70‑92% 8,34‑38.
In our series, the five‑year probability of stable renal function i.e. without doubling of serum creatinine was 62% in the whole group (data not shown). No difference was found in the proportion of subjects experiencing doubling of creatinine between the untreated group and those treated with steroids with or without cyclophosphamide.
The use of angiotensin converting enzyme inhibitors (ACEis) and/or angiotensin receptor blockers (ARBs) has been reported from various groups with favourable results in terms of reduction in the degree of proteinuria and achieving remission. In our study, as a policy during the initial years (between 1994 and 1998), we treated our patients conservatively, with ACEi and/or ARBs along with diuretics and /or stains as and when indicated. Later on (from 1998 onwards), with the published results of the benefits of using immunosuppressants, we started using them in our patients. This led to the creation of two groups of patients and provided us with an opportunity to compare the parameters between the two groups. We found insignificant difference in remission rates and doubling of serum creatinine at almost equal follow‑up duration in the two groups. However, the number of patients developing ESRD was significantly higher in patients treated conservatively.
Our group of patients treated with immunosuppressants was again divided into those who received steroids only and those who received steroids in combination with cyclophosphamide and there was no significant difference in baseline proteinuria and serum creatinine among these two groups.
In all, ten patients in our series developed ESRD; eight of them had normal initial serum creatinine values and their duration of progression to ESRD varied between 26‑168 months, and all except one had heavy proteinuria at the time of presentation. The two patients who had decreased renal function at baseline progressed to ESRD in 12 and 26 months; for one of them initial 24‑hours protein excretion could not be quantified while the other had 19.8 g/day excretion of protein. This finding is not in agreement with other published reports of a significant association of baseline serum creatinine with progression towards ESRD33,35,38.
Furthermore, of these 10 patients, only one acquired CR; two PR and the rest never acquired remission.
This study has certain limitations too. These include it being a retrospective study with no clearly demarcated randomisation among patients, and relatively small sample size. The study also originates from a single centre and the pathological features of renal biopsies were not analysed in the predication of remission or progression to ESRD.
CONCLUSION
In conclusion, untreated idiopathic MN is associated with a high risk of progression to ESRD. Baseline serum creatinine plays a significant role in predicting remission but no significant role in predicting progression towards ESRD. Randomised controlled trials are needed to confirm the beneficial effects of immunosuppressive treatment in our patients.
References
1. Fervenza FC, Sethi S, Specks U. Idiopathic membranous nephropathy: diagnosis and treatment. Clin J Am Soc Nephrol. 2008;3(3):905‑19. [ Links ]
2. Swaminathan S, Leung N, Lager DJ, et al. Changing incidence of glomerular disease in Olmsted County, Minnesota: a 30‑year renal biopsy study. Clin J Am Soc Nephrol. 2006;1(3):483‑7. [ Links ]
3. Haas M, Meehan SM, Karrison TG, Spargo BH. Changing etiologies of unexplained adult nephrotic syndrome: a comparison of renal biopsy findings from 1976‑1979 and 1995‑1997. Am J Kidney Dis. 1997;30(5):621‑31. [ Links ]
4. Glassock RJ. Secondary membranous glomerulonephritis. Nephrol Dial Transplant. 1992;7(1):64‑71. [ Links ]
5. Couser WG. Membranous nephropathy: a long road but well traveled. J Am Soc Nephrol. 2005;16(5):1184‑7. [ Links ]
6. Zucchelli P, Ponticelli C, Cagnoli L, Passerini P. Long‑term outcome of idiopathic membranous nephropathy with nephrotic syndrome. Nephrol Dial Transplant. 1987;2(2):73‑8. [ Links ]
7. Donadio JV Jr, Torres VE, Velosa JA, et al. Idiopathic membranous nephropathy: the natural history of untreated patients. Kidney Int. 1988;33(3):708‑15. [ Links ]
8. Schieppati A, Mosconi L, Perna A, et al. Prognosis of untreated patients with idiopathic membranous nephropathy. N Engl J Med. 1993;329(2):85‑9. [ Links ]
9. du Buf‑Vereijken PW, Branten AJ, Wetzels JF. Idiopathic membranous nephropathy: outline and rationale of a treatment strategy. Am J Kidney Dis. 2005;46(6):1012‑29. [ Links ]
10. Cattran D. Management of membranous nephropathy: when and what for treatment. J Am Soc Nephrol. 2005;16(5):1188‑94. [ Links ]
11. Glassock RJ. The treatment of idiopathic membranous nephropathy: a dilemma or a conundrum? Am J Kidney Dis. 2004;44(3):562‑6. [ Links ]
12. Branten AJ, du Buf‑Vereijken PW, Klasen IS, et al. Urinary excretion of beta2‑microglobulin and IgG predict prognosis in idiopathic membranous nephropathy: a validation study. J Am Soc Nephrol. 2005;16(1):169‑74. [ Links ]
13. Bazzi C, Petrini C, Rizza V, et al. Urinary excretion of IgG and alpha(1)‑microglobulin predicts clinical course better than extent of proteinuria in membranous nephropathy. Am J Kidney Dis. 2001;38(2):240‑8. [ Links ]
14. Troyanov S, Wall CA, Miller JA, Scholey JW, Cattran DC; Toronto Glomerulonephritis Registry Group. Idiopathic membranous nephropathy: definition and relevance of a partial remission. Kidney Int. 2004;66(3):1199‑205. [ Links ]
15. Dumoulin A, Hill GS, Montseny JJ, Meyrier A. Clinical and morphological prognostic factors in membranous nephropathy: significance of focal segmental glomerulosclerosis. Am J Kidney Dis. 2003;41(1):38‑48. [ Links ]
16. Gansevoort RT, Heeg JE, Vriesendorp R, de Zeeuw D, de Jong PE. Antiproteinuric drugs in patients with idiopathic membranous glomerulopathy. Nephrol Dial Transplant. 1992;7(1):91‑6. [ Links ]
17. Ambalavanan S, Fauvel JP, Sibley RK, Myers BD. Mechanism of the antiproteinuric effect of cyclosporine in membranous nephropathy. J Am Soc Nephrol. 1996;7(2):290‑8. [ Links ]
18. Rostoker G, Ben Maadi A, Remy P, Lang P, Lagrue G, Weil B. Low‑dose angiotensin‑converting‑enzyme inhibitor captopril to reduce proteinuria in adult idiopathic membranous nephropathy: a prospective study of long‑term treatment. Nephrol Dial Transplant. 1995;10(1):25‑9. [ Links ]
19. Azad NS, Muzaffar S, Pervez S, Ahmed A, Rabbani A, Ali A. Demographic and Clinicopathologic features of membranous glomerulonephritis. J Coll Physicians Surg Pak. 2004;14(1):35‑8. [ Links ]
20. Mubarak M, Kazi JI, Naqvi R, et al. Pattern of renal diseases in native renal biopsies in adults from Pakistan. Nephrology. 2011;16:87‑92. [ Links ]
21. Kazi JI, Mubarak M, Ahmed E, Akhter F, Naqvi SA, Rizvi SA. Pattern of glomerulonephritides in adult nephrotic syndrome from Pakistan. Clin Experiment Nephrol. 2009;13:38‑43. [ Links ]
22. Davison AM, Cameron JS, Kerr DN, Ogg CS, Wilkinson RW. The natural history of renal function in untreated idiopathic membranous glomerulonephritis in adults. Clin Nephrol. 1984;22(2):61‑7. [ Links ]
23. Marx BE, Marx M. Prediction in idiopathic membranous nephropathy. Kidney Int. 1999;56(2):666‑73. [ Links ]
24. Zent R, Nagai R, Cattran DC. Idiopathic membranous nephropathy in the elderly: a comparative study. Am J Kidney Dis. 1997;29(2):200‑6. [ Links ]
25. Neugarten J, Acharya A, Silbiger SR. Effect of gender on the progression of nondiabetic renal disease: a meta‑analysis. J Am Soc Nephrol. 2000;11(2):319‑29. [ Links ]
26. Silbiger SR, Neugarten J. The impact of gender on the progression of chronic renal disease. Am J Kidney Dis. 1995;25(4):515‑33. [ Links ]
27. Pei Y, Cattran D, Greenwood C. Predicting chronic renal insufficiency in idiopathic membranous glomerulonephritis. Kidney Int. 1992;42(4):960‑6. [ Links ]
28. Cattran DC, Pei Y, Greenwood CM, Ponticelli C, Passerini P, Honkanen E. Validation of a predictive model of idiopathic membranous nephropathy: its clinical and research implications. Kidney Int. 1997;51(3):901‑7. [ Links ]
29. Davison AM, Cameron JS, Kerr DN, Ogg CS, Wilkinson RW. The natural history of renal function in untreated idiopathic membranous glomerulonephritis in adults. Clin Nephrol. 1984;22(2):61‑7. [ Links ]
30. Laluck BJ Jr, Cattran DC. Prognosis after a complete remission in adult patients with idiopathic membranous nephropathy. Am J Kidney Dis. 1999;33(6):1026‑32. [ Links ]
31. Passerini P, Pasquali S, Cesana B, Zucchelli P, Ponticelli C. Long‑term outcome of patients with membranous nephropathy after complete remission of proteinuria. Nephrol Dial Transplant. 1989;4(6):525‑9. [ Links ]
32. Ponticelli C, Zucchelli P, Passerini P, et al. A randomized trial of methylprednisolone and chlorambucil in idiopathic membranous nephropathy. N Engl J Med. 1989;320(1):8‑13. [ Links ]
33. Reichert LJ, Koena RA, Wetzels JF. Prognostic factors in idiopathic membranous nephropathy. Am J Kidney Dis. 1998; 31:1‑11. [ Links ]
34. Perna A, Schieppati A, Zamora J, Giuliano GA, Braun N, Remuzzi G. Immunosuppressive treatment for idiopathic membranous nephropathy: a systematic review. Am J Kidney Dis. 2004;44(3):385‑401. [ Links ]
35. Honkanen E, Törnroth T, Gronhagen‑Riska C. Natural history, clinical course and morphological evolution of membranous nephropathy. Nephrol Dial Transplant. 1992;7(1):35‑41. [ Links ]
36. Kida H, Asamoto T, Yokoyama H, Tomosugi N, Hattori N. Long‑term prognosis of membranous nephropathy. Clin Nephrol. 1986;25(2):64‑9. [ Links ]
37. Honkanen E. Survival in idiopathic membranous glomerulonephritis. Clin Nephrol. 1986;25(3):122‑8. [ Links ]
38. Torres A, Dominguez‑Gil B, Carreño A, et al. Conservative versus immunosuppressive treatment of patients with idiopathic membranous nephropathy. Kidney Int. 2002;61(1):219‑27. [ Links ]
Prof. Dr. Muhammed Mubarak
Professor of Pathology
Histopathology Department,
Sindh Institute of Urology and Transplantation,
Civil Hospital, Karachi, Pakistan
Phone No. 9221 94989172
Fax No. 9221 32726165
Email address: drmubaraksiut@yahoo.com
Disclosure of Potential Conflicts of Interest: None declared
Received for publication: Jun 16, 2015
Accepted in revised form: Oct 30, 2015














