Serviços Personalizados
Journal
Artigo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Acessos
Acessos
Links relacionados
-
 Similares em
SciELO
Similares em
SciELO
Compartilhar
Portuguese Journal of Nephrology & Hypertension
versão impressa ISSN 0872-0169
Port J Nephrol Hypert vol.32 no.1 Lisboa mar. 2018
ORIGINAL ARTICLE
Urine volume and residual renal function decline among patients on peritoneal dialysis – searching for associations
Ricardo A. Macau1, Joana R. Silva1, Andreia Leite2, Rita Magriço1, Pedro Bravo1, Fernando Teixeira e Costa1, Aura Ramos1
1 Hospital Garcia de Orta – Nephrology Department, Almada, Portugal
2 Public Health Unit, ACES Amadora, Portugal
ABSTRACT
Introduction: Preservation of urine volume and residual renal function in patients on peritoneal dialysis (PD) is a major concern. Some factors have been associated with better prognosis, such as the use of biocompatible solutions, furosemide, or renin-angiotensin-aldosterone system blockers. However, results from previous studies have not been consistent. We thus aimed to study the relation between baseline characteristics of incident patients on PD, treatment characteristics, glomerular filtration rate (GFR) and urine volume (UV) variation.
Subjects and methods: We retrospectively analyzed incident patients on PD (first option) in our unit in terms of variation of UV and GFR after 24 months of follow-up. We studied the association between GFR and UV decline and baseline characteristics (age, gender, diabetes mellitus or hypertension diagnoses, body mass index, CKD etiology, and use of beta-blockers, diuretics, renin-angiotensin-aldosterone system blockers) as well as PD treatment characteristics (PD modality, use of icodextrin, dialysis days per week, presence of peritonitis, membrane characteristics such as Ca125 peritoneal level and dialysate-to-plasma creatinine), dwell hours per day and glucose load) using Spearman correlation for numerical variables and differences of means for binomial variables.
Results: We analyzed 25 patients. Urine volume decreased on average 0.59 mL after 24 months and glomerular filtration rate declined from 7.9 to 7.03 mL/min/1.73m2. All patients used biocompatible solutions. We did not find any association between glucose-exposure, use of diuretics or renin-angiotensin-aldosterone system blockers and urine volume or glomerular filtration rate decline. There was a significant relation between diuresis and GFR changes.
Discussion: Our patients present a slower decline of residual renal function than that described in the literature. Strategies to preserve diuresis, including the use of biocompatible solutions, may explain these results.
Key-words: Diuresis, Glomerular Filtration Rate, Peritoneal Dialysis, Renal residual function
INTRODUCTION
Residual renal function (RRF) and urine volume (UV) preservation is associated with better clinical outcomes in dialysis patients, including improved dialysis adequacy, nutrition, quality of life, technique survival and patient survival1-3. Patients with chronic kidney disease (CKD) that start dialysis treatment usually do so before becoming anuric. Hemodialysis (HD) has been the main dialysis modality since its start in the early 1960s. Most patients under HD tend to become anuric4. The development of continuous ambulatory peritoneal dialysis (CAPD) in the late 1970s has shown a better preservation of both RRF and UV5. Studies evaluating patients under automated peritoneal dialysis (APD) have shown that RRF decline is similar to what happens in CAPD6. In patients on peritoneal dialysis (PD), UV might account for one half of the variance in glomerular filtration rate (GFR)7 and some studies have demonstrated a stronger association between clinical outcomes and urinary clearance than with peritoneal clearance markers4,8-10.
Previous studies have assessed a variety of factors as predictors of faster decline in RRF and UV. However, results are not always consistent. These studies identified females, non-Caucasian race, diabetes mellitus, and congestive cardiac failure as risk factors for rapid RRF decline in dialysis patients11. Higher baseline (GFR)7,12 and higher 4-hour dialysate-to-plasma creatinine ratio (DTPCR)7 were also associated with more rapid decline in RRF. Higher serum phosphate was also associated with faster decline12. Peritoneal effluent CA125 concentrations were also associated with peritoneal protein losses and with the increase in the usage of higher glucose dialysate to compensate for loss of residual renal function13. On the other hand, the use of biocompatible PD solutions, achievement of overall higher systolic blood pressure (SBP), lower peritoneal ultrafiltration (UF) and lower dialysate glucose exposure over time4 were identified as protective factors. Results regarding the use of furosemide, tolvaptan, angiotensin-converting enzyme inhibitors (ACEi) or angiotensin receptor blockers (ARB) have been inconsistent, with some studies showing a protective effect and others showing no effect1,4,14.
Hypertension and hypotension (as well as important variations in a single patient) were also identified as risk factors1. Finally, studies assessing peritonitis episodes have not shown a clear association between this variable and RRF decline15.
Improving our understanding of which factors are associated with RRF decline would help guide clinical practice. Therefore, further studies clarifying which variables determine RRF decline are needed. Even the definition of RRF decline is not consistent and this might explain some differences in terms of predicting factors. Some authors analyze the change slope while others have considered a rapid decline of RRF as either a decrease of GFR superior to 2 ml/min/1.73m2 in a period of 6 months (and confirmed after a second evaluation) or a UV reduction to less than 500 mL/day16.
We thus aimed to study the relation among baseline characteristics of incident patients on PD, treatment characteristics and GFR and UV variation.
SUBJECTS AND METHODS
Study design, inclusion and exclusion criteria, and data collection
We conducted a retrospective cohort study of the incident PD patients in our unit between January 1st, 2009 and June 30th, 2015. Patients were required to be 18 years or older and to have preserved diuresis (UV>500mL/24h) at baseline. Patients with (i) previous HD treatment; (ii) previous renal graft, or (iii) follow-up shorter than 24 months were excluded. We reviewed each patients medical record and collected demographic and clinical information. Baseline data included age, gender, BMI, diabetes or hypertension diagnoses, CKD etiology, and PD modality. We collected clinical data such as GFR, UF, peritoneal UF, DTPCR, peritoneal levels of Ca125, type of peritoneal solution, daily glucose-exposure, use of icodextrin, beta-blockers, loop diuretics, ACEi or ARB and episodes of peritonitis at baseline and then at 6, 12, and 24 months (± 1 month).
Net peritoneal ultrafiltration (UF, L/day), was defined as the mean difference between the total volume of dialysate inflow and the total volume of dialysate outflow.
Daily dialysate glucose exposure (g/day) was calculated as the mean of the daily volume of dialysate (L/day) multiplied by the dialysate glucose concentration (g/L). Residual GFR was calculated as the average of 24-hour urinary creatinine clearance. When data was missing (4 total missing-values related to 4 patients) we estimated GFR using CKD-EPI formula. We have considered the use of icodextrin, beta-blockers, loop diuretics, ACEi or ARB when they were present during more than 6 months of follow-up. We have considered a patient with significant episodes of peritonitis when they have been more than one episode during the follow-up (24 months).
Statistical analysis
We analyzed data descriptively at baseline, using absolute numbers and proportions for categorical variables and central tendency and variation for numerical variables. We assumed a linear association between GFR and UV decline, and time from baseline at the individual level. We then defined this decline as the slope of linear regression (minimum squares method) between each one of these variables and time from baseline until end of follow-up. We have calculated and analyzed both GFR and UV slopes from baseline to 24 months (mean square slopes). We studied the association between GFR and UV decline and baseline Spearman correlation for numerical variables and differences of means for binomial variables. We were particularly interested in evaluating the effect of glucose daily exposure with GFR variation. Stata(R) software v. 15 was used.
RESULTS
We consulted our clinical registries and identified 43 adult patients that started PD in our unit between January 1st, 2009 and June 30th, 2015. Eighteen patients were excluded as 4 had been previously on HD; one had a UV less than 500mL/24h at baseline and 14 did not accomplish 24 months of follow-up (two of them died, three of them received a renal transplant and eight were transferred to HD).
Twenty-five patients were included; sample characteristics are presented in Table 1. Patients were mainly male (19, 76%), with a mean age when starting PD of 59 years-old. The majority was hypertensive (83%) and overweighed (60%), and only 20% were diabetic.
Regarding etiology, only 3 patients had diabetic nephropathy. Other etiologies included chronic glomerulonephritis (n=3), ANCA vasculitis (n=2), and autosomal dominant polycystic kidney disease (n=2). The vast majority of the patients were treated with diuretics (92%), renin-angiotensin-aldosterone system blockers (RAASB) (72%). All patients used biocompatible solutions and 36% also used icodextrin solution. APD was the modality of choice in 15 patients. In our sample, 10 patients had more than one episode of peritonitis during 24 months. Average number of dialysis days per week was 6.8, with an average period of 9.6 hours per day. The average amount of glucose-exposure was calculated as 117 ± 35 g/day. In this period, 6 patients were hospital admitted (length of hospitalization of 8 days in average).
GFR mean at the beginning of PD was 7.93 ± 3.25 mL/min/1.73m2 and after 24 months was 7.03 ± 4.49 mL/min/1.73m2. The GFR change overall after 24 months was -0.06 mL/min/1.73m2. The diuresis change overall after 24 months was -0.59 mL. UV mean at the beginning was 1544 mL and after 24 months was 1670mL. Mean peritoneal levels of Ca125 were 24 U/L and mean DTPCR was 0.69. Mean UF was 189 mL (Table 1).
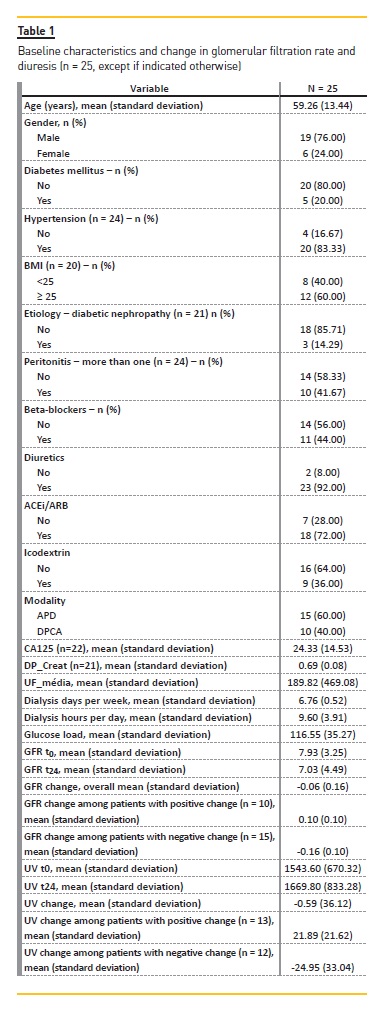
Table 2 and Table 3 present the results of our analyses assessing association between the variables under study and GFR and UV variation, respectively.
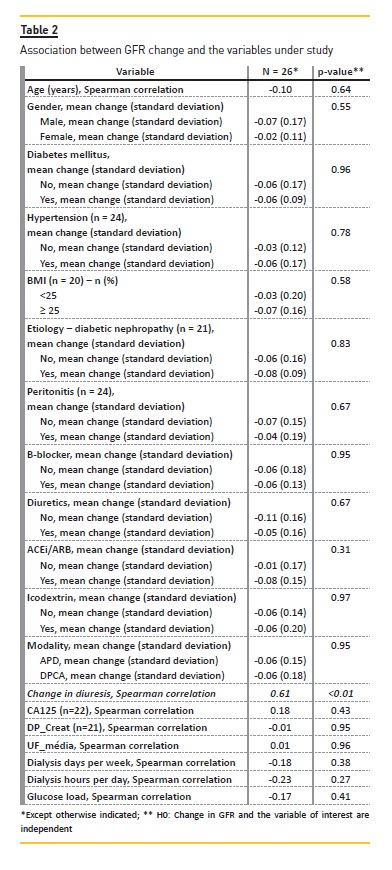
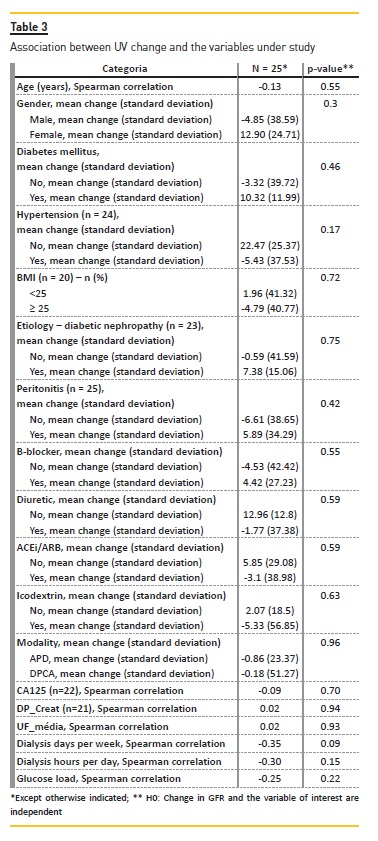
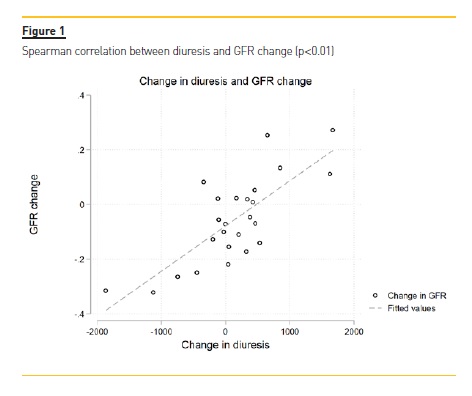
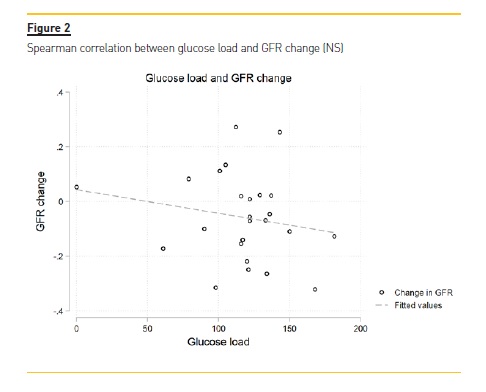
There was a significant correlation between UV reduction and GFR reduction (p-value = 0.01) (Figure 1). We did not observe any clear correlation between UV/GFR variation and age, gender, presence of diabetes mellitus or hypertension, BMI categories, CKD etiology, frequent episodes of peritonitis, use of beta-blockers, diuretics, ACEi or ARB, use of icodextrin, PD modality, Ca125, DTPCR,UF, dialysis days per week, dialysis hours per day or glucose load. There was no association with glucose load and GFR variation (Figure 2).
DISCUSSION
We analyzed the evolution of UV and GFR and we attempted to identify factors associated with RRF decline among incident peritoneal dialysis. We had a better GFR decline profile than that identified in previous studies. In particular, we observed a mean GFR of 7.93 ± 3.25 mL/min/1.73m2 at baseline and a mean GFR of 7.03 ± 4.49 mL/min/1.73m2 at 24 months while in the balANZ trial1, GFR declined from 7.5 ± 2.9 mL/min/1.73 m2 at baseline to 3.3 ± 2.8 mL/min/1.73 m2 at 24 months.
While mean UV at end of follow-up was slightly higher than at baseline, we observed an overall decline in UV. This apparent discrepancy can be explained by the method used to assess change in UV. For this calculation, we considered all the data points available (0, 6, 12 and 24 months) and not only two points. It is thus possible to have a higher UV at 24-months than at baseline and still a decreasing UV. It is noteworthy that we observed a decline but it is minimal. The high variation in UV at the end of follow-up is also consistent with this result.
Our analyses did not identify any factor associated with RRF decline. This might be explained by several factors. First, there was not a significant decline of RRF in our sample. Second, our sample had very limited variation in a number of factors: (i) we only used biocompatible PD solutions, which have been identified as a protective factor; (ii) there was a PD prescription consistency among the group and there were only slight differences in terms of glucose load, number of hours and number of days of dialysis, (iii) most of the patients were under diuretics and RAASB, (iv) and finally, we had a small sample, limiting power to detect a relationship if it exists and we did not have enough patients to split the sample into a fast versus a slow GFR decline groups.
We were not able to apply multivariate models due to sample size. In a PD population using only biocompatible solutions, other factors might have a modest impact that was not detectable due to the sample size.
Most of our patients were hypertensive and were under anti-hypertensive agents. We were not able to perform an evaluation regarding blood pressure variation based only on single values of office blood pressure registries. The association of hypotensive episodes regarding GFR and UV decline might be further explored in further studies. The relationship between this episodes and hospitalization might also be assessed. RRF might be associated with decrease hospitalization length17 and the impact of hospitalization in RRF decline might be analyzed in further studies.
Our study includes patients with a comprehensive follow-up and clinical incidents were identifiable in our clinical database. Our sample is mostly composed of Portuguese-origin patients.
As limitations to our study, we neither analyzed the effect of blood pressure or phosphorous control, nor the effect of using nephrotoxics (such as iodated contrasts).
Some patients might have some degree of GFR and diuresis recovery if they started PD after an acute on chronic renal injury which is sometimes difficult to assess. In this study, we only selected incident patients that had not been previously on hemodialysis nor received a kidney transplant. Results with patients that have not primarily opted for PD might have been different. We were not able to track any important change regarding CA125 peritoneal levels or DTPCR variation due to sample size and available data. We did not find any association concerning baseline levels and RRF or UV decline.
We have seen a low increment in diuresis and the fact that diuresis was associated with GFR might indicate that strategies to increment diuresis, including use of diuretics and use of biocompatible solutions, might have a beneficial effect upon GFR preservation. As mentioned above, UV preservation might have an important association with GFR preservation and clinical outcomes7.
We believe that these results are important to show that using biocompatible solutions, RRF might decline more slowly and that strategies to preserve UV are also important. In the future, analyzing the rest of our PD patients that were excluded might be challenging because they tend to present a lesser volume of diuresis at baseline, but it might be interesting to look for factors associated with some degree of recuperation of diuresis and GFR in this group. At present, the predictors of preserved RRF and its impact in terms of technique and patient survival in the global cohort of PD patients (first-choice of renal replacement therapy and the others) are yet to be defined. Studies assessing this issue with larger samples and the use of multivariate models might help to identify other associations.
References
1. Htay H, Cho Y, Pascoe EM, Darssan D, Hawley C, Johnson D. Predictors of residual renal function decline in peritoneal dialysis patients: the balANZ trial. Perit Dial Int 2017;37(3):283-89. [ Links ]
2. Perl J, Bargman JM. The importance of residual kidney function for patients on dialysis: a critical review. Am J Kidney Dis 2009;53(6):1068-81. [ Links ]
3. Wang AYM, Brimble KS, Brunier G, et al. ISPD cardiovascular and metabolic guidelines in adult peritoneal dialysis patients part I – assessment and management of various cardiovascular risk factors. Perit Dial Int 2015;35(4):379-87. [ Links ]
4. Krediet RT. Preservation of residual kidney function and urine volume in patients on dialysis. Clin J Am Soc Nephrol 2017;12(3):377-379. [ Links ]
5. Rottembourg J, Issad B, Gallego JL, et al. Evolution of residual renal function in patients undergoing maintenance haemodialysis or continuous ambulatory peritoneal dialysis. Proc Eur Dial Transplant Assoc 1983;19:397-403. [ Links ]
6. Bieber SD, Burkart J, Golper TA, Teitelbaum I, Mehrotra R. Comparative outcomes between continuous ambulatory and automated peritoneal dialysis: a narrative review. Am J Kidney Dis 2014;63(6):1027-37. [ Links ]
7. Johnson DW, Mudge DW, Sturtevant JM, et al. Predictors of decline of residual renal function in new peritoneal dialysis patients. Perit Dial Int 2003;23(3):276-83. [ Links ]
8. Bargman JM, Thorpe KE, Churchill DN, CANUSA Peritoneal Dialysis Study Group. Relative contribution of residual renal function and peritoneal clearance to adequacy of dialysis: a reanalysis of the CANUSA study. J Am Soc Nephrol 200112(10):2158-62. [ Links ]
9. Termorshuizen F, Korevaar JC, Dekker FW, et al. The relative importance of residual renal function compared with peritoneal clearance for patient survival and quality of life: an analysis of the Netherlands Cooperative Study on the Adequacy of Dialysis (NECOSAD)-2. Am J Kidney Dis 2003;41(6):1293-302. [ Links ]
10. Diaz-Buxo JA, Lowrie EG, Lew NL, Zhang SM, Zhu X, Lazarus JM. Associates of mortality among peritoneal dialysis patients with special reference to peritoneal transport rates and solute clearance. Am J Kidney Dis 1999;33(3):523-34. [ Links ]
11. Moist LM, Port FK, Orzol SM, et al. Predictors of loss of residual renal function among new dialysis patients. J Am Soc Nephrol 2000;11(3):556-64. [ Links ]
12. He L, Liu X, Li Z, et al. Rate of decline of residual kidney function before and after the start of peritoneal dialysis. Perit Dial Int 2016;36(3):334-9. [ Links ]
13. Redahan L, Davenport A. Peritoneal dialysate effluent and serum CA125 concentrations in stable peritoneal dialysis patients. J Nephrol 2016;29(3):427–34. [ Links ]
14. Szeto CC, Kwan BCH, Chow KM, et al. Predictors of residual renal function decline in patients undergoing continuous ambulatory peritoneal dialysis. Perit Dial Int 2015;35(2):180-8. [ Links ]
15. Rivera-Gorrin M, Teruel-Briones JL, Rodríguez-Mendiola N, et al. Residual renal function in patients on peritoneal dialysis: effect of peritonitis episodes. Nefrologia (English Version) 2014;34:802-4. [ Links ]
16. Herget-Rosenthal S, von Ostrowski M, Kribben A. Definition and risk factors of rapidly declining residual renal function in peritoneal dialysis: an observational study. Kidney Blood Press Res 2012;35(4):233-41. [ Links ]
17. Curran, SP, Bargman, JM. The importance of residual renal function in peritoneal dialysis. Dial. Transplant 2011;40: 349-55. [ Links ]
Ricardo A. Macau, MD
Hospital Garcia de Orta – Nephrology Department, Almada, Portugal
E-mail: ricardoandremacau@gmail.com
Acknowledgements
Authors would like to acknowledge the help of the Peritoneal Dialysis unit staff
Disclosure of potential conflicts of interest: None to declare.
Received for publication: 13 Jan 2018
Accepted in revised form: 1 Mar, 2018














