INTRODUCTION
Atrial fibrillation (AF) is the most common cardiac arrhythmia, with an estimated prevalence between 2 and 4%(1), worldwide, and a tendency for the incidence rate to increase2,3. End‑stage kidney disease (ESKD) patients have higher risk of developing AF1,2,4,5,6,7,8,9.
The prevalence of AF in this population ranges from 7 to 27%, a frequency 10‑to 20‑fold higher than in the general population, and it seems to be due to specific issues related with this condition, such as hypertension, left ventricular hypertrophy, ischemic heart disease, electrolyte abnormalities, and modulation of sympathetic nervous and renin‑angiotensin systems10. There is an established relationship between the increased incidence of AF and worsening kidney function9. Furthermore, we are observing a parallel rise in incidence of AF and chronic kidney disease (CKD), probably due to the increase in the elderly population and comorbidities such as diabetes mellitus and hypertension9.
At this point, the management of AF is controversial. In individuals without kidney disease, anticoagulant treatment should be instituted based on risk scores, such as the CHA2DS2‑VASc score, to prevent thromboembolism. However, CKD stages 3‑5 and 5D patients have higher hemorrhagic risk per se that must be weighted by the physician when prescribing anticoagulant treatment, and the usual risk scores seem to have worse predictive value in this population5,6. Also, guidelines from various societies are contradictory in which is the best approach in these situations1,11‑13.
The aim of this review is to understand the state of art on this topic, specifically the recommendations of the newest guidelines, the various risk scores and their pitfalls, and the possible benefits of non‑vitamin K oral anticoagulants (NOAC) versus the well‑known vitamin K antagonists (VKA).
INDICATIONS FOR ANTICOAGULANT TREATMENT IN AF
One of the key points in the treatment of AF is the prevention of thromboembolic events. Patients with AF have increased risk of stroke (up to five‑fold) and transient ischemic attack, due to the abnormal blood stasis in the atria, endotelial dysfunction and abnormalities of blood coagulation, conferring a prothrombotic state1,14,15.
However, this risk is not homogeneous, and it is modulated by the presence of specific factors, including age, gender, hypertension, diabetes mellitus, heart failure, history of stroke or thromboembolism, and vascular disease1,14,15. For this reason, it is mandatory to assess the stroke risk of each individual. The most used clinical risk score is the CHA2DS2‑VASc score, which summarizes the common stroke risk factors (Table I). It is a good predictor of low‑risk patients, and has moderate power identifying high‑risk patients1,14.
Table I The CHA2DS2‑VASc scoring system and characteristics of bleeding risk scores.
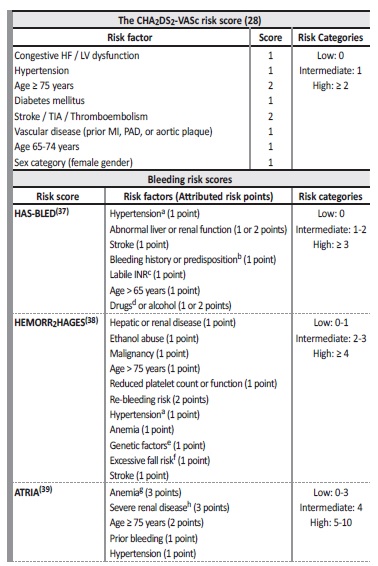
HF = heart failure; LV = left ventricular; TIA = transient ischemic attack; MI = myocardial infarction; PAD = peripheral artery disease.
a Uncontrolled; b Includes anemia; c Defined as the time in therapeutic range < 60%; d Includes antiplatelet or nonsteroidal anti‑inflammatory drugs; e CYP 2C9 single nucleotide polymorphism; f Based on high risk of falling, dementia, Parkinson’s disease or psychiatric disease; g Defined as hemoglobin < 13 g/dL in men and < 12 g/dL in women; h Glomerular filtration rate < 30 mL/min or dialysis dependent
Following the evidence‑based treatment guidelines from the European Society of Cardiology (ESC) and the American Heart Association/American College of Cardiology/Heart Rhythm Society (AHA/ACC/HRS), anticoagulation is recommended for prevention of stroke in AF patients except those with low risk, therefore oral anticoagulants (OAC) should be considered in patients with a CHA2DS2‑VASc score of 2 or greater in men or 3 or greater in women1,11.
Despite the advantages of prescribing anticoagulants to prevent thromboembolic events, it is important to take into account the bleeding risk associated with the use of OAC. Based on this premise, when initiating antithrombotic therapy, the clinician must assess the patient’s bleeding risk, and balance the risk/benefit of these drugs. The most used score and the better at predicting bleeding risk is the HAS‑BLED score. The European Cardiology Society recommends the assessment of bleeding risk, not to withhold the use of OAC in high‑risk patients (evidence shows a net clinical benefit of OAC even in high‑risk group), but to draw attention and manage the modifiable risk factors, for instance hypertension, excessive alcohol intake, concomitant use of antiplatelet or non‑steroidal anti‑inflammatory drugs (NSAIDs), and labile international normalized ratio (INR), and flag up the high‑risk patients for earlier follow‑up1.
Concerning the topic of OAC, several studies have evaluated antithrombotic efficacy and hemorrhagic risk of VKAs and NOACs. Warfarin reduces stroke by approximately 60% compared with no antithrombotic treatment, it is more efficacious than antiplatelet therapy and the absolute reductions in stroke are higher than the absolute hemorrhagic events16. However, it is associated with multiple drug and food interactions, and it requires regular monitoring and therapeutic adjustments due to the narrow therapeutic window, to achieve and maintain a target INR in the therapeutic range of 2.0‑3.01,14,17,18.
Relative to NOACs, a meta‑analysis from 2014 shows that apixaban, dabigatran, edoxaban and rivaroxaban have a significantly lower risk of stroke and systemic embolic events, intracranial hemorrhage and mortality comparing with VKAs, and a similar efficacy in preventing ischemic stroke, but an increased gastrointestinal bleeding risk19.
These drugs do not require frequent coagulation monitoring, which enhances therapeutic adherence and quality of life1,20, and have fewer interactions with drugs and food18. In fact, the benefits of these drugs in reducing the risk of major bleeding compared with VKAs are higher when INR control was poor(19). However, it is importante to note that all NOACs have some degree of renal clearance, mainly dabigatran (80% excreted via kidneys), followed by edoxaban (50%), and then rivaroxaban and apixaban (35% and 27%, respectively), so renal function should be evaluated periodically, to adapt the dose if any change occurs21.
Nevertheless, the prevention of thromboembolism in patients with concomitant CKD is a hot‑topic on AF management, and the risk/benefit ratio remains less clear for those with severe CKD (creatinine clearance [CrCl] < 30 mL/min) and ESKD (CrCl < 15 mL/min or on dialysis), where randomized clinical trials data on the safety and efficacy of OAC are lacking. CKD is by itself a prothrombotic and a prohemorrhagic condition1,12,22,23, and coexists in some degree in about 40‑50% of AF patients1. The presence of simultaneous AF and CKD promotes higher rates of thromboembolic events, with an increased risk of death9.
Pathophysiological factors behind CKD‑related thrombus formation include endothelial dysfunction, accelerated arteriosclerosis and coagulation abnormalities, and are summarized in Table II 9,23). Paradoxically, CKD also increases the hemorrhagic risk, mainly intracerebral and gastrointestinal bleeding, which is even more significant in dialysis9. Once more, the pathophysiology of these events is multifactorial and associated with uremic platelet dysfunction, hemostasis abnormalities and nitric oxide metabolism9. In fact, in contrast to what happens in individuals without kidney failure, where the benefits of OAC in stroke prevention are well established, in patients with ESKD the bleeding risk can outweigh the thrombotic risk24.
Table II Pathophysiology of thromboembolism in CKD.
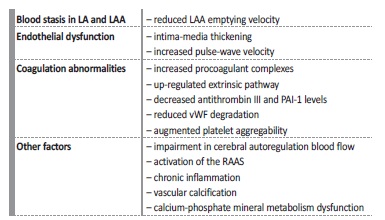
LA - left atrium; LAA - left atrial appendage; PAI‑1 - plasminogen‑activator inhibitor‑1; vWF - von Willebrand factor; RAAS - renin‑angiotensin‑aldosterone system
Taking into account the characteristics of these patients, the management of AF can be challenging. As already mentioned, there is a lack of high‑quality evidence for the effectiveness and safety of OAC in CKD, and principally in ESKD. No randomized clinical trials (RCT) have yet studied OAC in patients with concomitant AF and severe CKD or ESKD, because this group is systematically excluded from the trials1,12,18. Indeed, CKD patients at all stages of disease were excluded from almost 90% of the RCT evaluating anticoagulants25. Thus, the few recommendations on this topic are based on observational and retrospective studies and meta‑analyses, which have provided no consistent results8.
Owing to this knowledge gap, the European guidelines recommend the use of warfarin or NOACs in patients with AF and CrCl > 30 mL/min, whereas they do not recommend the use of any of the NOACs on CrCl < 15 mL/min or dialysis patients. Relatively to those with CrCl 15‑30mL/min, despite the lack of robust evidence, this document considers a dose reduction of rivaroxaban, edoxaban and apixaban a feasible option in patients with severe CKD(1). On the other hand, KDIGO statements do not support VKAs for stroke prevention in CKD5(D), although they suggest that clinicians may ponder the use of lower doses of apixaban (2,5 mg, orally, twice daily) in this group13.
In contrast, ACC/AHA/HRS guidelines recommend anticoagulation with warfarin and reduced dose of apixaban in patients with ESKD with a CHA2DS2‑VASc score ≥ 2 in men and ≥ 3 in women, despite safety not yet having been demonstrated (recommendation class IIb, level of evidence B‑NR)11. Finally, the European Stroke Organization guidelines state that it might be reasonable to use NOACs over VKAs for stroke prevention in patients with mild to moderate renal impairment, but recognizes that there is a lack of evidence for OAC in ESKD patients26.
VALIDITY OF THROMBOEMBOLIC AND HEMORRHAGIC RISK SCORES IN ESKD
As said, before the institution of anticoagulant treatment in a patient with AF, clinicians should assess individual thrombotic and hemorrhagic risks. In the general population, there are some risk scores, like CHA2DS2‑VASc and HAS‑BLED scores that are broadly validated and applied in clinical practice. However, these risk scores have shown a lower predictive value in CKD and the dialyzed population.
Beginning with stroke risk scores, the first widespread one in the medical community was the CHADS2 score, which included recente congestive heart failure, age ≥ 75, hypertension, diabetes mellitus, and history of stroke or transient ischemic attack. The latter risk factor scored 2 points, and each one of the others 1 point27. The CHA2DS2‑VASc risk score appears as an extension of the CHADS2, with the addition of the following risk factors: age between 65‑74, sex and vascular disease, which includes previous myocardial infarction, peripheral artery disease or aortic plaque28. Concerning the application in CKD patients, these scores have a lower accuracy to discriminate patients who will or will not have thrombotic events, mainly in CKD stages 4, 5 and 5(D)6. Chao et al., in a study with more than 10000
patients, showed that both CHADS2 and CHA2DS2‑VASc can be applied in the ESKD population with AF, as well as CHA2DS2‑VASc value in ischemic stroke prediction and in accurate identification of those at low stroke risk compared with CHADS2, even more refining the risk of those with CHADS2 0‑1, showing who had a higher stroke risk29.
Still, with the analysis of severe bleeding rate without OAC treatment and the annual rate of ischemic stroke in patients with every rating of CHA2DS2‑VASc, the authors believe that only when CHA2DS2‑VASc > 6 the benefits in stroke prevention outweigh the hemorrhagic risk in ESKD patients, so they propose to consider OAC for individuals with CHA2DS2‑VASc > 629.
Since CKD is per se a prothrombotic condition, some investigators proposed the addition of CKD or renal parameters, such as proteinuria and estimated glomerular filtration rate (eGFR) to improve the performance of risk scores in CKD patients. R2CHADS2 and ATRIA scores have incorporated these factors, and have been validated(5). R2CHADS2 is an extension of CHADS2, adding CrCl < 60 mL/min (2 points) to the score, and later, in a modified version, eGFR <60 (1 point) and <30 (2 points)30,31,32.
The prediction ability of the first model was better than CHADS2 and CHA2DS2‑VASc, and the score was validated in some cohorts, including one with ESKD patients(5). However, adding CKD to the CHADS2 and CHA2DS2‑VASc stroke risk scores did not independently add predictive information32. Likewise, a Swedish study did not find added value for stroke prediction by considering CKD33.
Finally, the ATRIA score is composed of many CHA2DS2‑VASc risk factors (age, prior stroke, female sex, diabetes mellitus, heart failure and hypertension), and eGFR < 45 mL/min/1,73 m2 or ESKD requiring dialysis, proteinuria and interaction between age and prior stroke34.
It shows better performance than CHADS2 and CHA2DS2‑VASc, however a study shows that CHA2DS2‑VASc was still better to identify the truly low risk patients35. Not only did McAlister et al. not find improvements with ATRIA, the authors also recognized that this model has worse predictive ability in patients with any degree of CKD36.
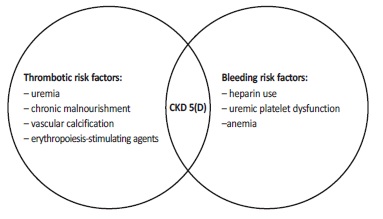
Indeed, it is known that the performance of the traditional tools in predicting such events is worse with the increasing in severity of CKD36. The CKD population has unique stroke risk factors that are not taken into account in risk scores (Figure 1). Moreover, in CKD stage 5(D) patients, the AF attributable stroke risk is not yet clarified, and also seems to be lower than in the general population. Of note, dialysis patients present a higher competing risk of death, that outweighs the stroke risk imputable to AF, and have unique thrombotic risk factos associated to ESKD: uremia; chronic malnourishment; vascular calcification and use of erythropoiesis‑stimulating actos5.
That said, the applicability of the traditional risk scores to dialysis patients is not clear(5). Furthermore, not only is it challenging to extrapolate stroke risk scores from the general population to CKD 5(D) patients, such also holds true for bleeding risk scores. Nowadays, the most widely used hemorrhagic risk score is the HAS‑BLED, which includes hypertension (uncontrolled, systolic pressure > 160 mmHg), renal disease (chronic dialysis, kidney actos s o r creatinine ≥ 2.26 mg/dL), abnormal liver function, previous stroke, bleeding history or predisposition (e.g. medication), labile INR (defined as the time in therapeutic range < 60%), age > 65, medication usage predisposing to bleeding (aspirin, clopidogrel or NSAIDs), and alcohol use (> 8 drinks/week), as risk actos37. Other bleeding scores used are HEMORR2HAGES, which includes kidney or hepatic disease, alcohol abuse, malignancy, age > 75 years, reduced platelet count or dysfunction, prior bleeding, uncontrolled hypertension, anemia, genetic actos (CYP 2C9 single nucleotide polymorphisms), excessive fall risk, and history of stroke, and ATRIA, which scores 3 points for anemia and severe kidney dysfunction (each one), 2 points for age ≥ 75, and 1 point for prior bleeding and diagnosed hypertension (each one)38,39. Table I actos summarizes the characteristics of the 3 bleeding risk scores.
Comparing these scores, although all show a modest performance in predicting any clinically relevant bleeding, the HAS‑BLED was better at predicting major bleedings, and had the advantage of simplicity, in opposition, for actos , to HEMORR2HAGES, which includes genetic actos, na added difficulty in clinical practice40,41,42. In addition, the HAS‑BLED demonstrated predictive performance for intracranial hemorrhage5.
To our knowledge, there are no studies validating these risk scores in ESKD and dialyzed patients. McAlister et al. found that they have poor discrimination abilities in patients with non‑dialyzed CKD36.
Furthermore, a cohort evaluating their performance in patients on dialysis showed poor predictive abilities, mainly in the low‑risk group, where the incidence rates of bleeding were higher than predicted with the scores43.
These findings may be due to:
1. Bleeding causes cannot be the same in dialyzed patients and the general population. Risk factors included in the scores can have less relative importance in dialyzed patients, who have unique hemorrhagic risk factors (Figure 1) such as heparin use in dialysis and uremic platelet dysfunction, which are not included in the existent scores. Even anemia, an important and a very frequente risk factor present in this population, is only present in HEMORR2HAGES and ATRIA, not in the HAS‑BLED score. Additionally, heparina use can be the main reason for bleeding in low‑risk group5,43,44.
2. Patients on dialysis have higher mortality risk per se, that can compete with hemorrhagic risk43.
3. Few patients on dialysis are categorized as low‑risk, in contrast with general population, since they usually have bleeding risk factors (hypertension, anemia and antiplatelet drugs use) and risk scores include kidney disease, which leads to a worse discrimination of actual risk in this group43.
Finally, it is important to note that these bleeding risk scores were developed and validated in patients treated with VKAs, not contemplating NOACs, which can have a negative impact on the predictive power of current scores. That being said, it is urgent to develop new scores, specific for ESKD, on VKAs and NOACs.
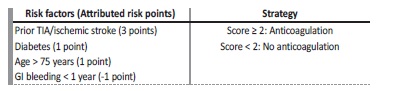
Recently, in an opinion article, De Vriese and Heine suggest na alternative dialysis‑specific score (Table III), to determine the introduction of anticoagulation, which should be analyzed and validated45.
USING VKA OR NOAC IN CKD STAGE 5D PATIENTS
A superior benefit/risk profile of NOACs compared with VKAs, namely warfarin, has been documented in individuals with normal renal function and in patients with eGFR > 25 mL/min45,46. Meta‑analyses of RCTs showed that, despite some heterogeneity across NOACs, these drugs are better to prevent thromboembolic events and are associated with lower rates of intracranial bleeding and death compared with VKAs. Conversely, the results on major bleeding risk are less consistent19,47,48.
In retrospective analyses, the heterogeneity across NOACs was also identified with regard to the risk of major hemorrhages, and it was shown that apixaban and dabigatran have a better benefit/risk profile49.
However, data in OAC in dialyzed patients is sparse: only a few observational studies compared stroke and bleeding rates in dialyzed patients treated with VKAs or NOACs45. This scarcity of high‑quality evidence for the correct prescription of OAC in CKD stage 5D patients has resulted in a great variability in clinical practice and uncertainty among the physicians. In fact, a recente physician‑based survey on the management of AF in patients with CKD showed considerable intra‑and inter‑specialty heterogeneity in the use and dosage of OAC, and across the various stages of CKD.
Moreover, when deciding treatment with OAC, the patient’s individual stroke risk and patient preferences outweigh the estimated bleeding risk12. Warfarin is the most used OAC in patients on dialysis. The main reasons for that include its pharmacokinetics (warfarin is metabolized mainly in the liver and is not eliminated by the kidneys, binds to plasma proteins, and dialysis does not significantly alter its effectiveness) and the fact that it was the only OAC recommended by some clinical guidelines. However, it is important to note that just a small fraction of ESKD patients with AF is treated with warfarin, due to the hesitations and worries of clinicians in its prescription18.
As said, no RCT evaluated warfarin in patients on dialysis, so the clinical practice is guided by the results of observational, retrospective studies. It is consensual among the various studies that warfarin is associated with an increased risk of major bleeding, but the benefit in prevention of ischemic events is less consistent18. Indeed, two recent meta‑analyses showed that warfarin increases the rate of bleeding events, such as hemorrhagic strokes, without improvements in ischemic stroke prevention or mortality, among dialysis patients8,50.
These results call into question whether, contrary to what happens to non‑dialysis patients with AF, the potential benefits of warfarin in preventing thromboembolic events in the dialysis population still prevail over the hemorrhagic risk.
This increased risk of bleeding may be explained by several facts. Firstly, it is common to use heparin during dialysis session51,52. Secondly, warfarin presents multiple drug and food interactions which may increase or decrease its plasma concentration53. Thirdly, it is known that the uremic state interferes with the metabolism of warfarin through hepatic cytochrome P450, making the maintenance of INR in the therapeutic range difficult to achieve51,52. In fact, in a prospective work on the use of warfarin in hemodialysis patients, Genovesi et al. showed that a higher time in therapeutic range (TTR) reduces the bleeding risk, which implies an adequate surveillance of ESKD patients using warfarin54.
Warfarin use is also complicated by the acceleration of vascular calcification: this drug interferes with the vitamin K‑dependent γ‑glutamyl carboxylase enzyme, necessary for the activation of matrix G1a protein (which is responsible for inhibiting vascular calcification).
The decrease of the activity of matrix G1a protein leads to accelerated vascular or valvular calcification, partially responsible for increasing risk of ischemic stroke6,18,51,53. In addition, this drug is a risk factor for calciphylaxis, a rare complication characterized by calcium deposits in the arteries and tissues, with patient’s predisposition to severe infections and high mortality rates (45‑80% at 1 year)6,18,45.
The warfarin‑related nephropathy is another complication of the use of warfarin, defined by an acute increase in INR to values greater than 3,0 and the evidence of acute kidney injury55. The exposure to this drug can result in glomerular hemorrhage with tubular obstruction by red blood cells, causing the acute kidney injury45,53,55,56.
Furthermore, due to the microbleeds and the vascular calcification, VKAs can accelerate the deterioration of kidney function52,56. Regarding NOACs, their use in patients with CrCl < 15 mL/min or on dialysis is still not approved in Europe(1). Based on pharmacokinetic studies, the US Food and Drug Administration approves the use of rivaroxaban and apixaban in dialysis patients, despite emphasizing that data on clinical efficacy and safety are still lacking57,58,59. NOACs seem to be superior to VKAs in terms of hemorrhagic safety.
However, there is still no evidence that they provide greater protection against thromboembolic events, based on observational studies60‑62.
The decrease of kidney function has a direct impact on NOAC pharmacokinetics, as they are eliminated at least in part by the kidneys. When glomerular filtration is impaired, their clearance reduces and drug exposure increases, which can lead to hemorrhagic events52,53. We know that during hemodialysis, the drug clearance is higher for molecules smaller than 1500 Daltons and intravascular unbound drugs, which leads to two conclusions: firstly, dabigatran is the only dialyzable NOAC due to its lower protein binding rate (35%), and secondly, only rivaroxaban and apixaban are considered valid options for dialysis patients, since they are practically not eliminated by dialysis (plasma protein binding of 92‑95% and 87%, respectively) and they presente the lowest renal clearance45,53,63.
Based on its pharmacokinetic and pharmacodynamic profile, it is understandable that dabigatran is the least used NOAC in dialysis patients. It is a direct thrombin inhibitor and it is mainly excreted by the kidneys and due to its protein binding rate, it can be eliminated up to 60% during one hemodialysis session, competing for a higher bleeding risk owing to the increase in its plasma concentration between dialysis sessions and for a lower efficacy to prevent thrombotic events after its clearance18. Also, some evidence seems to suggest that dabigatran may cause kidney injury similar to warfarin‑related nephropathy64.
Chan et al. compared the bleeding rates in dialysis patients treated with rivaroxaban, dabigatran or warfarin and showed that, despite its higher risk compared with warfarin (68.4 events per 100 patient‑years versus 35.9 events per 100 patient‑years), rivaroxaban is safer than dabigatran (83.1 events per 100 patient‑years)65. However, recente evidence, namely the Valkyrie Study, suggests that a lower dose of rivaroxaban (10 mg daily) can reduce the hemorrhagic complications compared with warfarin in CKD 5D patients, maintaining its effectiveness66. Rivaroxaban is a direct factor Xa inhibitor, non‑dialyzable due to its extensive protein binding (its area under curve showed no significant differences when administrated before or after hemodialysis)53.
Concerning apixaban, also a direct factor Xa inhibitor, only 25% excreted by the kidneys and barely affected by dialysis, it is the main alternative to warfarin in dialysis patients with AF18. Its superiority over warfarin in preventing ischemic and hemorrhagic stroke and its lower bleeding risk was proven in the ARISTOTLE trial. However, it excluded patients with serum creatinine concentration greater than 2.5 mg/dL or CrCl < 25 mL/min67. A post hoc analysis of this trial showed that even in patients with CKD, the rate of thromboembolic and hemorrhagic events was lower in the apixaban‑treatedgroup68.
Two recent meta‑analyses came to show the benefit/risk ratio of apixaban compared with warfarin in ESKD patients: in 2018 Chokesuwattanaskul et al. included data on more than 40000 patients with CKD stages 4, 5 and 5D, and evidenced that apixaban was associated with fewer major bleeding events and no differences in thromboembolic risk; two years later Kuno et al. showed that warfarin, dabigatran and rivaroxaban are associated with an increased risk of major bleeding than twice daily apixaban 5 mg or 2.5 mg51,62. This last meta‑analysis also proposed that the use of OAC is not associated with a reduction of stroke or systemic thromboembolic events in patients with AF on long‑term dialysis compared with no anticoagulant51. Regarding the appropriate dosage, an observational study compared 2351 dialyzed apixaban users with 7053 dialyzed warfarin users and showed a reduction in embolic events on the 5 mg twice daily apixaban group, with similar hemorrhagic risk compared to the 2.5 mg twice daily apixaban group61. In contrast, Mavrakanas et al. in a pharmacokinetic study showed that apixaban 2.5 twice daily resulted in similar area under curve in dialysis patients than the standard dosage in non‑CKD patients, with the dose of 5 mg twice daily being associated with supra‑therapeutic levels59.
It is also remarkable to note that when the majority of studies were conducted, there was no reversal agent available for NOACs, which can explain the higher rate of fatal bleeding compared to warfarin. Of note, only in 2018 was the use of an andexanet alfa, a reversal agent for factor Xa inhibitors, approved by the US Food and Drug Administration (and one year later by the European Medicines Agency)18. This is a determining factor in the choice of an anticoagulante agent in the population eligible for transplantation. Until andexanet alfa is promptly available, patients on a waiting list for transplantation may be denied kidney transplantation if medicated with a factor Xa inhibitor, as they are not dialysable and the bleeding risk may be considered unacceptable.
Several RCTs assessing the safety of NOACs and VKAs in hemodialysis patients with AF are currently ongoing or have recently been completed. The RENAL‑AF trial was halted prematurely due to failure to enroll a sufficient number of patients. However, its preliminary data showed no significant differences with respect to stroke, bleeding or mortality between the treatment arms69. As said, the Valkyrie study also found no significant differences in mortality or stroke risk, but showed a higher number of major bleedings on VKA‑treated arm against rivaroxaban‑treated arm70. The ADAXIA trial and the SAFE‑D trial are still ongoing, with the addition in the latter of a control arm without any OAC treatment, to assess whether or not OACs are really effective in preventing stroke in dialysis patients71,72.
Another promising therapeutic target is the coagulation factor XI (FXI), which seems to have a great importance in thrombus growth and stabilization, with a small impact on homeostasis. Thus, some clinical trials (EMERALD, RE‑THINC and CONVERT) are under way to assess the efficacy and safety of FXI inhibitors, which could be a potentially useful approach in patients with CKD with or without AF, due to their great thrombotic and hemorrhagic tendency73.
CONCLUSIONS
In conclusion, AF is a common condition in patients with ESKD, namely in dialysis patients that competes with other risk factors for the increase in ischemic and hemorrhagic events. Although OAC with VKAs or NOACs has been shown to be effective and safe in patients without CKD, data on this topic in CKD stages 4, 5 and 5D patients is sparse and the guidelines are also inconsistent in which is the best approach for these patients.
The recent results of meta‑analyses and Valkyrie and RENAL‑AF trials seem to point that apixaban 2.5 mg twice daily and rivaroxaban 10 mg daily are proper options for hemodialysis patients, and should be preferable to using VKA, mainly in patients with calciphylaxis.
However, these results should be confirmed in large‑scale studies, and new risk scores, targeted to this specific group, should also be developed.