Serviços Personalizados
Journal
Artigo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Acessos
Acessos
Links relacionados
-
 Similares em
SciELO
Similares em
SciELO
Compartilhar
Portugaliae Electrochimica Acta
versão impressa ISSN 0872-1904
Port. Electrochim. Acta v.28 n.3 Coimbra 2010
Testing Natural Fenugreek as an Ecofriendly Inhibitor for Steel Corrosion in 1 M HCl
A. Bouyanzer,1,* B. Hammouti,1 L. Majidi,2 B. Haloui3
1 LCAE-URAC18, Faculté des Sciences, Université Mohammed Premier, 60000 Oujda, Morocco
2 Laboratoire des Substances Naturelles & Synthèse et Dynamique Moléculaire, F S T, Université Moulay Ismail, Errachidia, Morocco
3 Laboratoire de biologie des plantes et des microorganismes, Faculté des Sciences, Université Mohammed Premier, 60000 Oujda, Morocco
DOI: 10.4152/pea.201003165
Abstract
The influence of a natural extract of fenugreek on the corrosion of steel in 1 M HCl has been studied by weight loss, polarisation and EIS measurements. Results obtained show that the natural substance inhibits the corrosion process. It acts on the cathodic domain without modifying the reduction mechanism. The inhibition efficiency increases with fenugreek concentration to attain 94% at extract of 10 g/L of fenugreek at temperatures between 308 and 353K. It is an efficient inhibitor. The effect of temperature on the corrosion behaviour of steel indicated that inhibition efficiency is temperature-independent. The activation energy of adsorption is determined.
Keywords: steel, natural substance, fenugreek, inhibition, corrosion, acid.
Introduction
The use of inhibitors is a practical technique to secure metals and alloys from aggressive environment. Large numbers of organic compounds revealed that N, S and O containing organic compounds may be efficient inhibitors. However, most of these compounds are not only expensive, but also toxic to living beings. It is needless to point out the importance of cheap and safe inhibitors of corrosion. So, considerable efforts are made to find corrosion inhibitors which are environmentally safe, ready available and of relatively low cost. Literature shows a growing trend in the use of natural products known as non-toxic compounds, called also green inhibitors, as corrosion inhibitors. We cited natural honey [1], nypa fructicans wurmb [2], opuntia extract [3], saccharides [4], natural tannins extracted from acacia and from pine bark [5], tryptamine [6]. A part of the program of our laboratory is devoted to research new non-toxic inhibitors. Natural plants are added as extract, oil or pure compounds and are the subject of various contributions: limonene [7], ginger [8], henna [9], jojoba oil [10], rosemary oil [11], artemisia oil [12], bgugaine [13-15], thym [16], eugenol and acetyleugenol [17], pulegone [18], menthols [19], cedar [20], rosmarinus [21], pulegone [22], have been found to be very efficient corrosion inhibitors for metal in aggressive media.
Our choice is made on fenugreek seeds, which contain the unique major free amino acid 4-hydroxyisoleucine (4-OH-ILeu) [23], to test as a non-toxic inhibitor for the protection of steel against corrosion in acid media.
The aim of the present paper is to study by gravimetric, polarisation and EIS measurements, the effect of the extract of the natural substance fenugreek as a non-toxic compound, on the corrosion of steel in 1 M HCl solution in the temperature range 308 - 353 K.
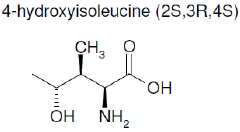
Figure 1. Molecular structure of 4-hydroxyisoleucine (4-OH-ILeu).
Experimental
The investigated steel materials of chemical composition in weight (0.21% C, 0.38% Si, 0.09% P, 0.01% Al, 0.05% Mn, 0.05% S) are used. The aggressive solution (1 M HCl) is prepared by dilution of Analytical Grade 37% HCl. The fenugreek seeds were ground and dried. The inhibitory solutions are prepared from the stirred 1 M HCl solution containing 10 g/L of fenugreek during 3 days. The extract was filtered to serve as initial solution.
Gravimetric measurements were carried out in a double walled glass cell equipped with a thermostat-cooling condenser. The solution volume is 60 mL. The steel specimens used have a square form (2.0 cm x 2.0 cm x 0.05 cm). Prior to immersion, the steel samples are polished with different emery papers up 1200, degreased with acetone, washed thoroughly with doubly distilled water, and finally dried in air. The immersion time for the weight loss is 6 hours at 308 K and 1 hour at other temperatures. To ensure the reproducibility, each experiment is repeated three times.
Electrochemical measurements were carried out in a conventional three electrode electrolytic cylindrical Pyrex glass cell. The working electrode (WE) had the form of a disc cut from the steel sheet. The area exposed to the corrosive solution was 1 cm2. A saturated calomel electrode (SCE) and a disc platinum electrode were used, respectively as reference and auxiliary electrode. The temperature was thermostatically controlled at 308 ± 1 K.
Electrochemical experiments were recorded using an EG&G potentiostat (263 A), coupled to a computer equipped with a 352 Soft Corr III software. Before recording the polarisation curves, the test solution was de-aerated and magnetically stirred for 30 min in the cell with pure nitrogen. Gas pebbling was maintained throughout the experiments. WE was then inserted and prepolarised at -800 mV for 10 min, in order to remove the oxide film from the electrode, and Ecorr was monitored until stationary (30 min). The scan rate was 1 mVsec-1.
Electrochemical impedance spectroscopy (EIS) was carried out with a Tacussel electrochemical system at Ecorr after immersion in the solution without bubbling, the 1 cm2 circular surface of steel exposed to the solution being used as working electrode. After the determination of the steady-state current at a given potential, sine wave voltage (10 mV) peak to peak, at frequencies between 100 kHz and 10 mHz were superimposed on the rest potential. Computer programs automatically controlled the measurements performed at rest potentials after 30 min of exposure. The impedance diagrams are given in the Nyquist representation.
Results and discussion
Gravimetric measurements
The corrosion rate in 1 M HCl (W°corr) and at various concentrations of the tested compound (Wcorr) is determined after 6 hours of immersion at 308 K. Values of corrosion rate and inhibition efficiencies are given in Table 1. The inhibition efficiency E is calculated by the relation :
E % = 100 . ( 1 - Wcorr / W°corr )
where Wcorr and W°corr are the corrosion rates of steel with and without fenugreek.
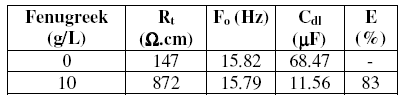
Table 1. Weight loss of steel in 1 M HCl at different concentrations of fenugreek and the corresponding inhibition efficiencies at 308 K after 6 h immersion.
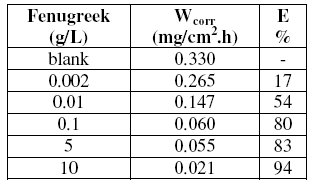
It is clear that the addition of fenugreek reduces the corrosion rate (CR) in 1 M HCl solution. It is clear from Table 1 that CR decreases more and more with the increase of natural extract. This phenomenon is widely observed from the reduced quantity of hydrogen formed on the metal surface. The inhibitory effect increases then with the increase of fenugreek concentration to attain 94% at 10 g/L of fenugreek extract. From these essays at 308 K, we may conclude that fenugreek is an efficient inhibitor of steel corrosion in 1 M HCl solution.
Polarisation measurements
Polarisation essays were performed to know how inhibitory effect acted. Fig. 2 shows the influence of fenugreek extract on the polarisation curves for the steel electrode in 1M HCl. Values of associated electrochemical parameters and corresponding inhibition efficiencies (E%) are given in Table 2. The examination of Fig. 2 and Table 2 shows that the addition of the fenugreek decreases only the cathodic current density. The decrease is more pronounced with the increase of the inhibitor concentration. The Tafel plots indicate that the mechanism of hydrogen reduction is activation control.
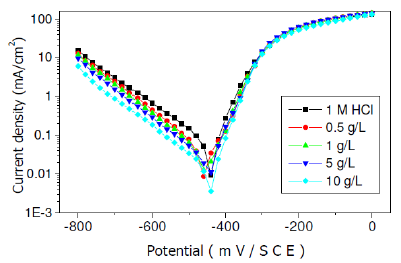
Figure 2. Polarisation curves of steel in HCl at various concentrations of fenugreek.
Table 2. Electrochemical parameters of steel in 1 M HCl + fenugreek extract at various concentrations and the corresponding inhibition efficiency.
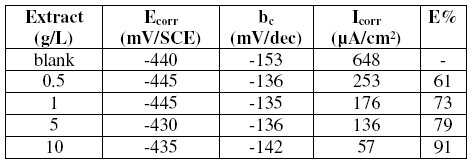
The presence of the tested oil does not affect the cathodic Tafel slope, indicating that the mechanism of H+ reduction is not modified with the oil concentration. Also the corrosion potential is almost the same in the presence of inhibitor. In the other hand in the anodic domain, a slight decrease of anodic current is observed in the presence of natural product. The cathodic effect is then dominant against the anodic one. In other words, the decrease of the corrosion rate may be explained by the inhibitory action of the inhibitor on cathodic branches of the polarisation curves by blocking the reduction of H+ ions on cathodic sites. The observed inhibitory action of fenugreek may be due to the adsorption of its molecules on the metal surface, making a barrier for the access of hydrogen ions, and then their reduction on the cathodic sites of the steel surface.
The inhibition efficiency reaches 80% at 10 g/L. Consequently, fenugreek is a good inhibitor and acts as a cathodic inhibitor.
EIS measurements
In order to get more information about the corrosion behaviour of steel in unihibited acid and inhibited acid at 10 g/L natural extract, electrochemical impedance spectroscopy (EIS) measurements have been carried out at 308 K. Table 3 collects the EIS parameters and corresponding efficiency derived from EIS diagrams. The charge-transfer resistance (Rt) values are calculated from the difference in impedance at lower and higher frequencies. The double layer capacitance (Cdl) and the frequency at which the imaginary component of the impedance is maximal (-Zmax) are found as represented in equation:
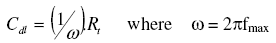
The inhibition efficiency got from the charge transfer resistance is calculated by:
E (%) = 100 . ( 1 – Rt / Rt/inh )
where Rt/inh and Rt are the charge transfer-resistance values with and without inhibitor, respectively.
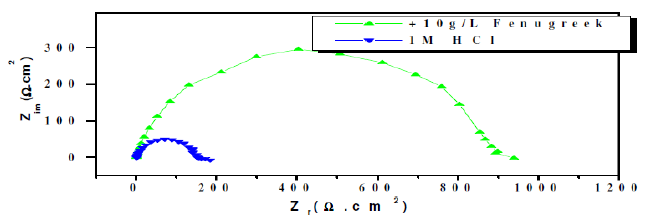
Figure 3. Impedance plots for steel in 1.0 M HCl solution and with fenugreek (10 g/L).
Table 3. Characteristic parameters evaluated from EIS diagrams for steel in 1 M HCl.
The corroding surface of steel in 1 M HCl is inhomogeneous because of its roughness, presence of impurities, dislocations, grain and subgrain morphology, etc. These phenomena explained the depression of the capacitance semi-depressed obtained in Fig. 3 [25]. The increase of Rt and the decrease of the capacitance with the concentration of natural extract can be explained by some decrease of the surface heterogeneity, due to the adsorption of the inhibitor on the most active adsorption sites.
It is important to note that the IE values calculated using different methods are smaller than the values obtained by the other methods. However, three methods were used in the present work to determine the inhibitive action of fenugreek toward corrosion of steel in HCl solution. If the techniques which give the highest and the lowest values are excluded, we still have the proof of the good inhibitive power of the fenugreek.
Effect of temperature
Since temperature has a great effect on the corrosion rate and acid pickling of steel, usually carried out at elevated temperatures up to 333 K in hydrochloric acid, measurements were made in 308-353 K range. Inhibitors are expected to be chemically stable to provide high protective efficiency in hot acid solutions.
Gravimetric measurements are also taken at various temperatures 308-353 K with and without inhibitor, during a period of one hour and at a maximal concentration; the corresponding results are given in Table 4.
It is clear from Table 4 that the corrosion rate increases with the rise of temperature in absence and presence of the inhibitor.
The increase in corrosion rate is more pronounced with the rise of temperature for blank solution. In the presence of fenugreek, the attack of steel is highly reduced. The inhibition efficiency is constant with the temperature; it oscillates around 94%.
Table 4. Effect of temperature on the corrosion rate of steel at 10g/L (t=1h).
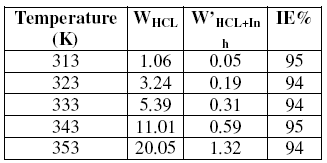
Fig. 4 shows Arrhenius plots of the corrosion rate for both the blank and the inhibitor. The activation energies are determined by the relation:
W = K exp (-Ea / RT) and W’ = K’ exp (-Ea’/ RT)
where Ea = 66.5 kJ/mol and Ea’ = 71.9 kJ/mol are the activation energies in the absence and presence of fenugreek, respectively.
The increase of E% with temperature is explained by a specific interaction between the steel surface and the inhibitor [25-26]. It’s also stated that the corrosion process taking place in the presence of powerful inhibitors is characterised by an activation energy whose value is smaller than that of the uninhibited process [27]. Generally, the lower value of Ea in an inhibitor’s presence when compared to that in its absence is attributed to its chemisorption, while the opposite is the case with physical adsorption. In our case, the values of activation energy are close and then we may introduce that the molecules of the extract act by both physical adsorption and chemisorption [25-27].
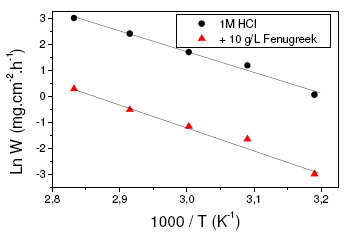
Figure 4. Arrhenius plots for the dissolution of steel/1 M HCl with and without fenugreek.
Conclusion
From the obtained results the following conclusions can be deduced:
Ø Fenugreek extract acts as an excellent inhibitor; its inhibition efficiency attains 94%.
Ø The inhibition efficiency is constant with the rise of temperature.
Ø It may be suggested to be applied at elevated temperature.
Ø The natural oil acts on steel surface as cathodic inhibitor
References
1. A.Y. El-Etre, M. Abdallah, Corros. Sci. 42 (2000) 731. [10.1016/S0010-938X(99)00106-7] [ Links ]
2. K.O. Orubite, N.C. Oforka, Materials Letters 58 (2004) 1768. [10.1016/j.matlet.2003.11.030]
3. A.Y. El-Etre, Corros. Sci. 45 (2003) 2485. [10.1016/S0010-938X(03)00066-0]
4. B. Müller, Corros. Sci. 44 (2002) 1583. [10.1016/S0010-938X(01)00170-6]
5. G. Matamala, W. Smeltzer, G. Droguett, Corros. Sci. 42 (2000) 1351. [10.1016/S0010-938X(99)00137-7]
6. G. Moretti, F. Guidi, G. Grion, Corros. Sci. 46 (2004) 387. [10.1016/S0010-938X(03)00150-1]
7. A. Chaieb, B. Hammouti, M. Benkaddour, M. Berrabeh, Acta Physico - Chimica Sinica 25 (2009) 1254.
8. A. Bouyanzer, B. Hammouti, Bull. Electrochem. 20 (2004) 63.
9. A. Chetouani, B. Hammouti, Bull. Electrochem. 19 (2003) 23.
10. A. Chetouani, B. Hammouti, M. Benkaddour, Pigm. & Resin Techn. 33 (2004) 26. [10.1108/03699420410512077]
11. A. Chaieb, B. Hammouti, M. Benkaddour, M. Berrabeh, Trans. of the SAEST 39 (2004) 58.
12. A. Bouyanzer, B. Hammouti, Pigm. & Resin Techn. 33 (2004) 287. [10.1108/03699420410560489]
13. B. Hammouti, S. Kertit, A. Melhaoui, Bull. Electrochem. 11 (1995) 553.
14. B. Hammouti, S. Kertit, A. Melhaoui, Bull. Electrochem. 13 (1997) 97.
15. S. Kertit, B. Hammouti, A. Melhaoui, Moroccan Patent 23910 (1995).
16. A. Chetouani, B. Hammouti, Bull. Electrochem. 20 (2003) 343.
17. E. Chaieb, A. Bouyanzer, B. Hammouti, M. Benkaddour, Appl. Surf. Sci. 246 (2005) 199. [10.1016/j.apsusc.2004.11.011]
18. Z. Faska, A. Bellioua, M. Bouklah, L. Majidi, R. Fihi, A. Bouyanzer, B. Hammouti, Monatshefte fur Chemie 139 (2008) 1417-1422 . [10.1007/s00706-008-0959-4]
19. Z. Faska, L. Majidi, R. Fihi, A. Bouyanzer, B. Hammouti, Pigm. Resin Techn. 36 (2007) 293. [10.1108/03699420710820405]
20. A. Bouyanzer, L. Majidi, B. Hammouti, Phys. Chem. News 37 (2007) 70.
21. M. Benabdellah, B. Hammouti, M. Benkaddour, M. Bendahhou, A. Aouniti, Appl. Surf. Sci. 252 (2006) 6212. [10.1016/j.apsusc.2005.08.030]
22. Z. Faska, A. Bellioua, M. Bouklah, L. Majidi, R. Fihi A. Bouyanzer, B. Hammouti, Surf. Rev. Let. 45 (2009).
23. A.M. Flammang, M.A. Cifone, G.L. Erexson, L.F. Stankowski, Jr., Food & Chem. Toxic. 42 (2004) 1769. [10.1016/j.fct.2004.07.003]
24. I.A. Ammar, F.M. Elkhorafi, Werkst. und Korros. 24 (1973) 702
25. A. Popova, E. Sokolova, S. Raicheva, M. Christov, Corros. Sci. 45 (2003) 33. [10.1016/S0010-938X(02)00072-0]
26. J.D. Talati, D.K. Gandhi, Corros. Sci. 23 (1983) 1315. [10.1016/0010-938X(83)90081-1]
27. W. Machu, Korros. und Metal. 14 (1988) 324.
Received 20 January 2009; accepted 16 April 2010
* Corresponding author: bouyanzer@yahoo.fr














