Serviços Personalizados
Journal
Artigo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Acessos
Acessos
Links relacionados
-
 Similares em
SciELO
Similares em
SciELO
Compartilhar
Portugaliae Electrochimica Acta
versão impressa ISSN 0872-1904
Port. Electrochim. Acta vol.34 no.2 Coimbra mar. 2016
https://doi.org/10.4152/pea.201602131
Cefuroxime axetil: A commercially available drug as corrosion inhibitor for aluminum in hydrochloric acid solution
Paul O. Ameh* and Umar M. Sanib
Department of Chemistry, Nigeria Police Academy Wudil, Kano State Nigeria
Abstract
Cefuroxime axetil (CA) a prodrug was tested as corrosion inhibitor for aluminum in hydrochloric acid solution using thermometric, gasometric weight loss and scanning electron microscope (SEM) techniques. Results obtained showed that this compound has a good inhibiting properties for aluminum corrosion in acidic medium, with inhibition efficiencies values reaching 89.87 % at 0.5 g / L . It was also found out that the results from weight loss method are highly consistent with those obtained by hydrogen evolution method and gasometric method; and all indicate that inhibitor efficiency increases with increasing inhibitor concentration. Cefuroxime axetil inhibited the corrosion of aluminum in solutions of HCl through the mechanism of physiosorption as confirmed by values of activation energy and free energy of adsorption. The adsorption of the inhibitor was also found to be spontaneous, exothermic and best fitted the Langmuir adsorption model. SEM analysis confirmed the existence of an absorbed protective film on the aluminum surface.
Keywords: Adsorption, Aluminium, Corrosion Inhibition, Cefuroxime axetil, SEM.
Introduction
In developed and developing countries, billions of dollars every year are spent on capital replacement and control methods for corrosion infrastructure [1 -2]. In recent years, owing to the growing interest and attention of the world towards the protection of the environment and the hazardous effects of using chemicals on the ecological balance, the use of eco-friendly inhibitors to replace the older, which is more toxic and harmful to the environment are been intensified [3-5]. Research has shown that for an inhibitor to be an effective protector against metal corrosion, it should be readily adsorbed on the metal surface through either physisorption or chemisorption processes [6-11]. Either of these adsorption processes depends primarily on the physicochemical properties of the inhibitor group such as functional groups, electronic density at the donor atom, molecular structure, etc. For instance, organic molecules, which have had a wide applicability and that have been extensively studied and used as corrosion inhibitors, often contain nitrogen, oxygen, and sulfur atoms, as well as multiple bonds in their molecules. Several researches have been carried out on the use of drugs as corrosion inhibitors for several metals in various media. For example, Fouda et al. [12], studied the corrosion inhibition characteristics of floxacillin, cloxacillin, dicloxacillin, cefadroxil and cephalexin on aluminum in 0.5 M H3PO4 using weight loss and galvanostatic polarization techniques. Results obtained revealed that the inhibition occurs through adsorption of the inhibitor molecules on the metal surface.
Other drugs that have been found to be good corrosion inhibitors include norfloxacin [13], Streptomycin [14], Cefatrexyl [11], Cefazolin [15]. The choice of some of the drugs used as corrosion inhibitors may be due to the fact that they have a large number of functional adsorption centres, are biodegradable, can be easily produced and purified.
In the present investigation, the corrosion inhibiting behavior of Cefuroxime axetil was investigated on aluminum in hydrochloric acid at 303 and 333 K using weight loss, thermometric gasometric and scanning electron microscope techniques.
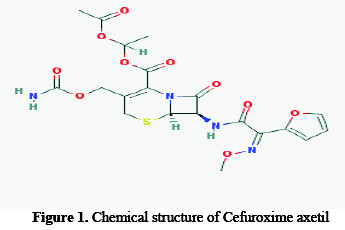
Cefuroxime axetil is a second generation oral cephalosporin antibiotic. It is an acetoxyethyl ester prodrug of cefuroxime which is effective orally [16]. The selection of this inhibitor is based on the following facts:
-They contain three kinds of heteroatom (four nitrogen, ten oxygen and one sulphur atom) as reactive center through which they can adsorb readily on the metal surface.
-These compounds are readily soluble in medium.
-They do not cause any health hazards, but also find their diverse application in various biological and pharmacological activities [16]; hence the use of Cefuroxime as corrosion inhibitors is safe.
Materials and Methods
Weight loss method
A previously weighed metal (aluminum sheet) was completely immersed in 250 ml of the test solution in an open beaker. The beaker was inserted into a water bath maintained at a temperature of 30 °C. Similar experiments were repeated at 60 °C. In each case, the weight of the sample before immersion was measured using Scaltec high precision balance (Model SPB31). After every 24 hours, each sample was removed from the test solution, washed in a solution of NaOH containing zinc dust and dried in acetone before re-weighing. The difference in weight for a period of 168 hours was taken as total weight loss. The inhibition efficiency (% I) for each inhibitor was calculated using equation 1 [17].
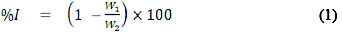
where W1 and W2 are the weight losses (g/dm3) for mild steel in the presence and absence of inhibitor in HCl solution respectively. The degree of surface coverage θ is given by the equation 2 [18]:

The corrosion rates for mild steel corrosion in different concentrations of the acid was determined for 168 h immersion period from weight loss using equation 3 [6]
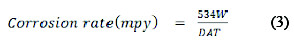
where W = weight loss (mg); D = density of specimen (g/cm3), A = area of specimen (square inches) and T = period of immersion (hour).
Gasometry method
The method used for hydrogen evolution measurement is as described elsewhere [19]. The test solution (different concentrations of acid, inhibitor or their mixtures) was poured into the reaction vessel (gasometer). Upon the introduction of mild steel, the flask was quickly corked and the rise in volume of the paraffin due to hydrogen evolution was noted after every minute until a steady volume was observed. From the results obtained, the corrosion inhibition efficiency was calculated using the following equation,

where Vb is the volume of hydrogen gas evolved by the blank and Vt is the volume of hydrogen gas evolved in the presence of the inhibitor, after time, t.
Thermometric method
This was also carried out as reported elsewhere [20]. The reaction number (RN) of each system was calculated by dividing the difference between the highest and lowest temperature attained by the time interval. From the reaction number, the inhibition efficiency (% I) of the inhibitor was calculated using equation 5
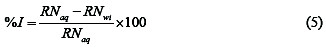
where RNaq is the reaction number in the absence of inhibitors (blank solution) and RNwi is the reaction number of 2 M HCl containing the studied inhibitor.
Scanning electron microscopy
The scanning electron microscope (SEM) Model No JSM-5600 LV was used to study the morphology of corroded in the presence and absence of inhibitor. The photographs were taken from that portion of the specimen where better information was expected.
Results and discussions
Effect of Cefuroxime axetil
The corrosion rate (CR) and reaction number (RN) of aluminum in HCl containing various concentrations of Cefuroxime axetil determined for 168h immersion period from gravimetric, gasometric and thermometric are presented in Table 1.
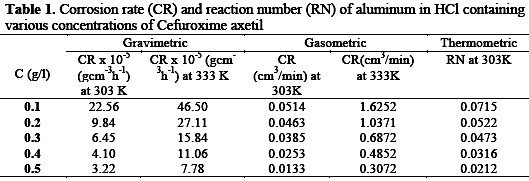
The results obtained indicates that the aluminum corrosion is reduced by the presence of Cefuroxime axetil in 1M HCl at all concentrations used in this study, since there is a general decrease in the rate of corrosion of the aluminum with increase in concentration of the inhibitor. This may be ascribed to the adsorption of this compound on the aluminum, producing a barrier, which isolates the surface from the corrosion environment.
Table 2 presents inhibition efficiencies of various concentrations of Cefuroxime axetil in HCl.
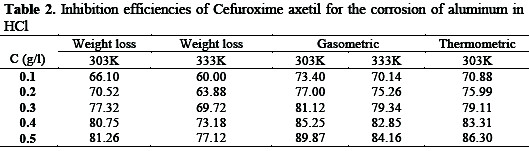
The inhibition efficiency was estimated to be 73.40 % even at extremely low inhibition concentration (0.1 M) and reaches 89.87 % at a concentration of 0.5 M. Such remarkable performances may be due to, the high molecular weight of CA, the presence of C=N, O-H, C=O, etc. which are electron donation groups and the presence of aryl groups. The inhibition efficiency was found to increases with increase in the concentration of the inhibitor but decreases with increase in temperature indicating that the mechanism of physical adsorption favours the adsorption of Cefuroxime axetil on aluminum surface. For a physical adsorption mechanism, the inhibition efficiency is expected to decrease with increase in temperature but for a chemical adsorption mechanism, the reverse is expected [20].
Also, the inhibition efficiency of CA obtained from the two methods were found correlate strongly (R2 = 0.8798 and 0.9644 for gasometric and thermometric respectively) with those obtained from gravimetric method. However, values of inhibition efficiency obtained from the weight loss were higher than the values obtained from thermometric and gasometric methods indicating that the average inhibition efficiency of CA is better than its instantaneous inhibition efficiency. The stability of the inhibitive properties of the studied inhibitor over 168 hours of immersion was studied by plotting values of inhibition efficiencies gotten from weight loss studies against time as shown in Fig. 2.
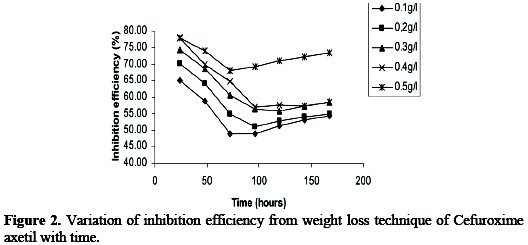
From the plots, it is also evident that the inhibition efficiency first decreased with time until a critical value was attained after which it started increasing. This trend suggests that at first, there was competition between the forces of adsorption and desorption and that after the critical zone, adsorption facilitated the formation of a protective layer and thus protected the metal against further corrosion attack.
Effect of temperature
According to Eddy [21], temperature affects the rate of any chemical reaction such that an increase in temperature leads to a corresponding increase in the rate of the reaction. Hence the effect of temperature on the corrosion of aluminum in HCl (in the absence and presence of CA) was studied using the Arrhenius Equation (equation 6) where the apparent activation energies (Ea) for the corrosion process in absence and presence of inhibitor were evaluated [6, 22].
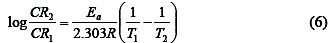
where CR1 and CR2 are the corrosion rates of aluminum in solution of HCl at the temperatures, T1(303 K) and T2 (333 K) respectively, Ea is the activation energy for the adsorption of CA on Al surface and R is the gas constant. Calculated values of the Ea are shown in Table 3.
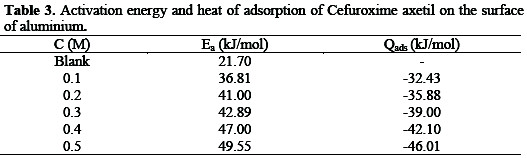
It can be seen from the table that the activation energies are lower than the value of 80 kJmol-1 required for a chemical adsorption mechanism supporting the earlier claims the adsorption of CA on aluminum proceeds by physical adsorption mechanism.
The heat of adsorption (Qads) of Cefuroxime axetil on aluminum surface was calculated using equation 7 [23]
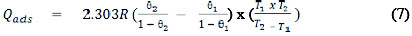
where Qads is the heat of adsorption of CA on Al surface, θ1 and θ2 are the degrees of surface coverage of the inhibitor at the temperatures, T1 and T2 (where T2 and T1 are 303 K and 333 K respectively) and R is the gas constant. Values of Qads calculated from equation 7 are presented in Table 3. These values are negative indicating that the adsorption of CA on aluminum surface is exothermic.
Adsorption considerations
Eddy et al.[24] stated that adsorption isotherms provide importance clues about the nature of metal inhibitor interaction. The general formula for adsorption isotherm is expressed can be as follows

where, f(0,x) is configuration factor, θ is the surface coverage, C is inhibitors concentration, K is adsorption constant and a is the molecular interaction parameter.
Data obtained from weight loss, thermometric and gasometric measurements were used to fit curves for different adsorption isotherms, including Temkin, Langmuir, Freundlich, Bockris-Swindles, Flory Huggins and Frumkin isotherm.
The test indicated that Langmuir adsorption isotherm best described the adsorption characteristics of CA on the surface of aluminum.
According to Ghasemi and Tizpar [25], if the assumptions of Langmuir adsorption isotherm are valid for the corrosion inhibition reaction, then, it can be assumed that the degree of surface coverage and the concentration of the inhibitor in the bulk electrolyte are related according to the following equation,
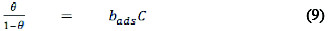
Rearrangement of equation 9, simplifies it to equation 10,
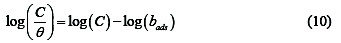
From equation 10, plots of log(C/θ) versus log(C) are expected to yield straight lines with slopes and intercepts equal to unity and log bads. Fig. 3 shows the Langmuir isotherm for the adsorption of CA on Al surface.
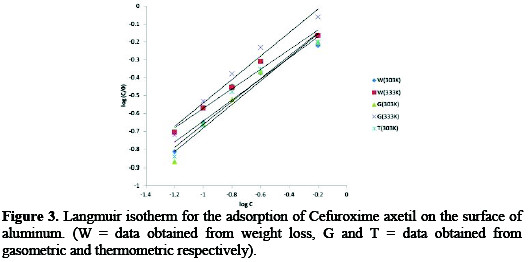
Adsorption parameters deduced from the plots are presented in Table 4.
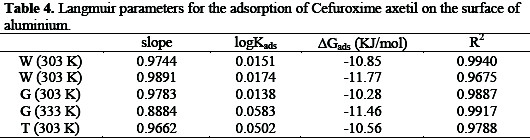
The results obtained reveal that the slopes and R2 values are very close to unity indicating a strong adherence of the adsorption data to the assumptions establishing the Langmuir model.
The equilibrium constant of adsorption (obtained from the Langmuir adsorption isotherm) is related to the equilibrium constant of adsorption according to equation 11 [26],
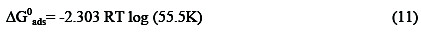
where K is the equilibrium constant of adsorption, 55.5 is the molar concentration of water, ΔGads is the free energy of adsorption of the inhibitor, R is the gas constant and T is the temperature. The free energies calculated from equation 11 are also presented in Table 4. Results presented indicate that the values of ΔG0ads are negative and lies between -10.28 and -11.77 kJ mol-1 . According to Bilgic and Sahin [27], values of standard Gibbs free energy of adsorption in aqueous solution around -20 kJ mol-1 or lower (more positive) indicate adsorption with electrostatic interaction between the adsorbent and adsorbate (physisorption), while those around or higher (more negative) than -40 kJ mol-1 involve charge sharing between the molecules and the metal (chemisorption). Therefore, the adsorption of CA on aluminum surface is spontaneous and supports the mechanism of physical adsorption.
Scanning electron microscope (SEM)
Fig. 4 and 5 show the surface morphology of aluminum exposed to 0.1 M hydrochloric acid in the presence and absence of 0.5 g / L CA respectively.


A uniform severe corrosion can be observed in the absence of inhibitor (Fig. 4). However, the SEM obtained for aluminum in presence of inhibitor (Fig. 5) showed that the metal surface is partially covered with inhibitor giving it a reasonable degree of protection which further proves that CA acts as good inhibitor at 0.1 M acid concentration [28].
Conclusions
From the study, the following conclusions can be drawn. CA efficiently inhibits the corrosion of mild steel in 0.1M HCl medium. Adsorption of CA on the surface of aluminum from 1M HCl obeys Langumur adsorption isotherm. The inhibition efficiency of CA increases with increasing the inhibitor concentration and on increasing the temperature, the corrosion rate increases. The calculated values of Ea, Qads, ΔGads indicates that the adsorption of inhibitor on the metal surface is physisorption, exothermic and followed by spontaneous process. Protective film formation against the acid attack is confirmed by SEM.
References
1. Thompson N G, Yunovich M, Dunmire D. Corros Rev. 2007;25:247. [ Links ]
2. Cherry B W, Skerry B S, Clayton V. Corrosion in Australia: the report of the Australian National Centre for Corrosion Prevention and Control feasibility Study. Department of Materials Engineering, Monash University: Australia; 1983. [ Links ]
3. Patni N, Agarwal S, Pallav S. Chin J Eng. 2013:1. [ Links ]
4. Dalo-Abu A M, Othman A A, Rawashdeh Al Fan. Int J Electrochem Sci. 2012;7:9303.
5. Sharma S K, Mudhoo A, Khamis E. Adsorption studies, modeling and use of green inhibitors in corrosion inhibition: an overview of recent research, green corrosion inhibitors: status in developing countries. In: Sharma S K, editor. Green corrosion chemistry and engineering. Wiley-VCH Publications: Weinheim; 2011. p 319. [ Links ]
6. Ameh P O, Oyeniyi S Q, Sani U M. Int J Chem, Mater Environm Research. 2015;2:1. [ Links ]
7. Hameed R S A. J Physical Chemistry. 2013;8:146. [ Links ]
8. Rani P D, Selvaraj S. Rasayan J Chem. 2010;3:473. [ Links ]
9. Saratha R, Kasthuri N, Thilagavathy P. Der Pharma Chemica. 2009;1:249. [ Links ]
10. Satapathy A K, Gunasekaran G, Sahoo SC, et al. Corros Sci. 2009;51:2848. [ Links ]
11. Morad M S. Corros Sci. 2008;50:436. [ Links ]
12. Fouda A S, Al-Sarawy A A, Ahmed F S, et al. Corros Sci. 2009;51:485. [ Links ]
13. Eddy N O, Odoemelam S A. Int J Pure Appl Chem. 2008;3:1. [ Links ]
14. Shukla S K, Singh A K, Ahamad I, et al. Mater Letters. 2009;63:819. [ Links ]
15. Singh A K, Quraishi M A. Corros Sci. 2010;52:152. [ Links ]
16. Sneader W. Drug discovery: a history. Chichester: John Wiley; 2002. [ Links ]
17. Oguzie E E. Corros Sci. 2008;50:2993. [ Links ]
18. Nnanna L A, Nwadiuko O C, Ekekwe N D, et al. Am J Mater Sci. 2011;1:143. [ Links ]
19. Umoren S A, Ebenso E E, Okafor P C, et al. Pigment Resin Technol. 2006;35:346. [ Links ]
20. Umoren S A, Ogbobe O, Ebenso E E, et al. J Appl Polym Sci. 2007;105:3363. [ Links ]
21. Eddy N O. Inhibition of the corrosion of mild steel in H2SO4 by some antibiotics. Ph. D Thesis. University of Calabar; 2008. [ Links ]
22. Momoh-Yahaya H, Eddy N O, Iyun J F, et al. Int J Modern Chem. 2012;2:127. [ Links ]
23. Bhajiwala H M, Vashi R T. Bull Electrochem Soc. 2001;17:441. [ Links ]
24. Eddy N O, Ibok U J, Ebenso E E. J Appl Electrochem. 2010;40:445. [ Links ]
25. Ghasemi Z, Tizpar A. Appl Sci. 2006;252:3667. [ Links ]
26. Flis J, Zakroczymski T. J Electrochem Soc. 1996;143:2458. [ Links ]
27. Bilgic S, Sahin M. Mater Chem Phys. 2001;70:290. [ Links ]
28. Rafiquee M Z A, Khan S, Saxena N, et al. Port Electrochim Acta. 2007;25:419. [ Links ]
Acknowledgement
The authors are grateful to TEFUND for sponsoring the research and Mrs Janet Onoja Ameh for typesetting the manuscript.
*Corresponding author. E-mail address: nocaseoche@yahoo.com
Received 28 July 2015; accepted 08 March 2016














