Serviços Personalizados
Journal
Artigo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Acessos
Acessos
Links relacionados
-
 Similares em
SciELO
Similares em
SciELO
Compartilhar
Acta Obstétrica e Ginecológica Portuguesa
versão impressa ISSN 1646-5830
Acta Obstet Ginecol Port vol.12 no.2 Coimbra jun. 2018
CASE REPORT/CASO CLÍNICO
Bilateral massive vulvar edema and thyroid dysfunction in ovarian hyperstimulation syndrome
Edema vulvar maciço bilateral e disfunção tiroideia na síndrome de hiperestimulação ovárica
Ana Galvão*, Cláudia Lourenço**, Susana Fraga***, Isabel Sousa Pereira**, Márcia Barreiro****
Centro Materno Infantil do Norte (CMIN), Centro Hospitalar do Porto (CHP)
*Interna de Obstetrícia e Ginecologia, Centro Materno Infantil do Norte, Centro Hospitalar do Porto
**Assistente Hospitalar Graduada de Ginecologia e Obstetrícia, CMIN - CHP
***Assistente Hospitalar de Ginecologia e Obstetrícia, CMIN - CHP
****Assistente Hospitalar de Ginecologia e Obstetrícia, Diretora do Centro de Procriação Medicamente Assistida/Banco Público de Gâmetas, CMIN - CHP
Endereço para correspondência | Dirección para correspondencia | Correspondence
ABSTRACT
Ovarian hyperstimulation syndrome (OHSS) can be a serious condition. There are very few cases described of massive vulvar edema in OHSS. Ovarian hyperstimulation can also have an important impact on thyroid function.
We present a case of severe OHSS, massive vulvar edema and severe thyroid dysfunction in a young woman with a congenital hypothyroidism submitted to a second IVF cycle for anovulation. The particularities of this case are related to the fact that it had severe and rare complications, even being a second IVF cycle, conducted in the same way as the previous one, in which there was no pregnancy.
Keywords: Vulvar edema; Ovarian hyperstimulation syndrome; Thyroid function; Controlled ovarian hyperstimulation.
Introduction
Ovarian hyperstimulation syndrome (OHSS) is a potentially life threatening condition that can occur after ovulation induction or ovarian stimulation for assisted reproduction. It is characterized by a massive enlargement of both ovaries and by increased capillary permeability, which can explain the full range of symptoms that may occur1. In severe cases, women can present with ascites, pleural effusion, increased coagulability and eletrolyte disorders2.
Massive vulvar edema is a rare complication of OHSS, with few cases described in the literature. Several mechanisms have been proposed as being able to produce this kind of edema in a scenario of OHSS2.
On the other hand, the high levels of estradiol characteristic of multifollicular development and aggravated in case of OHSS force the hypotalamic-pituitary--thyroid axis to give a rapid response in thyroid hormone production. In women with compromised thyroid function this high demand for thyroid hormones can precipitate a thyroid disfunction during the first trimester of pregnancy, when the fetus is highly dependent on maternal thyroid hormones for its neurologic development1.
We describe a patient with OHSS who developed bilateral massive vulvar edema and severe thyroid dysfunction at the same time, after embryo transfer.
Case report
A 27-year-old Caucasian woman underwent in vitro fertilization (IVF) in our unit for primary infertility, presumed to be caused by anovulation. She had congenital hypothyroidism controlled with levothyroxine 125 µg (her pre-stimulation TSH value was 1.24 µUI/ /mL). She had undergone unilateral left adnexectomy in adolescence because of a dermoid cyst and now she had polycystic ovarian syndrome. This was her second IVF cycle. She had undergone ovarian stimulation in an antagonist protocol, starting on day 2 of the cycle with recombinant FSH 200 IU/day and triggering was performed with 0.2 mg of triptorelin SC. In the first cycle, similar doses have been used with the same scheme, with a day 5 embryo transfer, without obtaining pregnancy and without OHSS.
Sixteen oocytes were retrieved. Nine embryos were obtained and two of them were transferred on day 5. The luteal phase was supported with vaginal progesterone and oral estrogen and her levothyroxine dose was mantained.
One day after embryo transfer, the patient developed abdominal distension due to ovarian enlargement, with abdominal disconfort but no ascites. Her blood tests were normal, without signs of hemoconcentration. She was advised to be at rest and was given prophylatic enoxaparin (because of the rest and the clinical-laboratory discrepancy), cabergolin and a hiperproteic diet and she was mantained in ambulatory surveillance. Five days after embryo transfer, she developed abdominal pain and resting dyspnea, presenting with moderate ascites and her single ovary was 105 mm larger in diameter, although there were no analytical alterations. Given the disparity between the clinical and analytical results, it was decided to hospitalize the patient in our gynecology unit for surveillance. She was treated with bed rest, appropriate liquid intake, intravenous albumin and mantained in prophylactic enoxaparin.
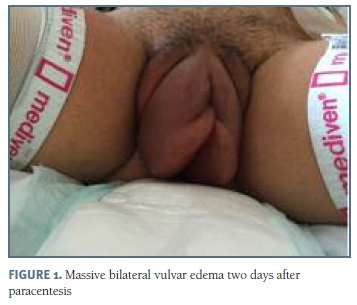
On hospitalization day 3, the patient had respiratory distress, abdominal tension and signs of hemoconcentration (haematocrit of 47.0 %), hypoproteinaemia (total proteins 5.0 g/dL, albumin level 3.3 g/dL) and it was decided to perform a culdocentesis, with draining of 725 mL of serohematic fluid. The procedure was repeated on hospitalization day 5 with drainage of 2000 mL. On day 7, the patient developed worsened dyspnea and since the accumulation of ascitic fluid reached the upper floor of the abdomen, it was decided to change the drainage pathway to paracentesis, which was performed at the classic point, in the opposite site of the ovary stimulated, with drainage of 2000 mL and resolution of this symptom. However, two days latter, she developed massive bilateral vulvar edema, that prevented her from walking (Figure 1). This was a pitting edema, painful, with tense skin, with labia minora reaching almost 5 cm. On the same day, her TSH value was 12.18 µUI/mL and the levothyroxine dose was increased to 150 µg. The vulvar edema subsided slowly in the next four days with local application of ice and with postural drainage and perineal massage and it was solved without long term sequelae. Three days later, the value of TSH continued to increase, reaching the value of 28.50 µUI/mL and the levothyroxine dose was increased again. During all these days and according to the water balance, a daily therapeutic review was carried out, with adjustments on the doses of diuretics and fluid therapy.
Her hospitalization lasted 32 days and two more paracentesis and several adjustments in the dose of levothyroxine were required until normalization of thyroid function, which occurred still during hospitalization. On the day of discharge, her TSH value was 4.92 µUI/mL, the haematocrit value was 28.6 % and the total proteins were 6.2 g/dL. Since 6 weeks of gestation, obstetric ultrasound has always revealed a single-fetus pregnancy. The remaining pregnancy had no complications. At 38 weeks, the patient went into labor and had a vaginal delivery with the birth of a healthy male newborn with 3035 g.
Discussion
The reported incidence of moderate OHSS after gonadotropin stimulation for IVF is 3% to 6% and for severe OHSS it is 0.1% to 2%3. It is a systemic disease thought to result from vasoactive peptides released from the granulosa cells in hyperstimulated ovaries and the most important mediator of this phenomenon is thought to be Vascular Endothelial Growth Factor (VEGF). OHSS may occur either as an early (early onset) or a late pattern (late onset); early pattern is associated with exogenously administered hCG and with a higher risk of preclinical miscarriage, whereas late OHSS may be closely associated with the conception cycles, as was the case of our patient4,5.
Several risk factors have been demonstrated to increase the risk of developing OHSS3. Our patient was younger than 30 years, Caucasian, with a body mass index of 19 Kg/m2, had a polycystic ovary, presented with an antral follicle count between 10 and 15, had a large number of small follicles on ultrasound during ovarian stimulation (between 12 and 15) and a large number of oocytes have been retrieved. However, at the same time, the presence of a single ovary, the fact that she had a estradiol serum value of 2854.0 pg/mL in trigger day and the presence of a normal ovarian response to a previous stimulation cycle have led us to presume a decreased risk in this particular case.
In terms of secundary prevention, coasting, using a GnRH agonist as a trigger for ovulation, cycle cancellation or freezing all embryos with subsequent late embryo transfer can all be effective6. The strategy of using a GnRH agonist as a trigger for ovulation makes the risk of hyperstimulation very low, which made us decide to proceed with the embryo transfer in this case, which was performed on day 5 also to see the patient's progress and to verify that she did not develop an early OHSS. Luteal phase support was made with progesterone but also with estrogen because of the possible deleterious effects of the agonist (used as a trigger). Besides that, elective single embryo transfer is also possible in patients at high risk for OHSS3, but it was not used in this case, considering the results of the previous FIV cycle.
From the moment an OHSS is installed, it is always possible that its aggravation causes the presence of edema in several places of the body, but there are very few cases of massive vulvar edema described in this context. The first of them was described in 1995, with the authors arguing that vulvar edema was the result of decreased oncotic pressure and increased hydrostatic pressure caused by OHSS7. However, of the 10 or so posterior cases found in the literature, it has been hypothesized that vulvar edema is related to paracentesis itself, that is, whenever the lower abdomen is used to make a paracentesis, a needle with a large gauge is used, which may cause a fistula formation between the peritoneum and subcutaneous abdominal cell tissue which, together with the increase in intra-abdominal pressure caused by ascites, may lead to the development of vulvar edema2. A patent Nuch channel or an occult inguinal hernia would be other differential diagnosis. The temporal relationship between the realization of paracentesis and the appearance of vulvar edema in our patient make fistula a very credible hypothesis, but the fact that this has not happened again despite repeated paracentesis may raise some doubts about its etiology. Some authors even propose the use of the upper abdominal region for paracentesis or the preference for culdocentesis, as a way to avoid this complication, which is rare but causes great temporary incapacity and pain2.
Elevation of estradiol levels typical of ovarian hyperstimulation imposes additional effort to the hypothalamic-pituitary-thyroid axis and this may lead to important thyroid dysfunction in women with underlying disease. It should always be kept in mind that the impact on thyroid function is always greater in women who do ovarian hyperstimulation than in those who have a spontaneous pregnancy1.
Several studies have compared the impact on the thyroid function of ovarian hyperstimulation in women without previous thyroid pathology between those who developed and those who did not develop OHSS and have come to the conclusion that the presence of OHSS does not confer a greater risk of thyroid dysfunction in women without pathology1,8. However, in women with previous thyroid pathology, cases of severe thyroid dysfunction associated with severe OHSS have been reported9. It is now believed that in cases where thyroid disease has an autoimmune etiology, the levothyroxine dose should be adjusted prior to the initiation of ovarian stimulation as a way to reduce complications and poor pregnancy outcomes related to hypothyroidism. Furthermore, it is believed that TSH levels should be monitored after ovarian stimulation and during pregnancy in these women1. Hypothyroidism is also a differential diagnosis of edema, since deficient production of thyroid hormones leads to changes in metabolism with deposition of mucopolysaccharides in the dermis, causing hard and elastic non-pitting edema, especially of the face and limbs.
In conclusion, the particularities of this case are related to the fact that it had severe and rare complications, such as massive vulvar edema and severe thyroid dysfunction occurring at the same time in the same patient with OHSS, even being a second IVF cycle, conducted in the same way as the previous one, in which the major difference was that the first one did not result in pregnancy.
Our patient presented three important causes of edema at the same time (OHSS, paracentesis and hypothyroidism), however hypothyroidism edema usually has different manifestations, which makes the hypothesis of its contribution unlikely. Dyspnea and orthopnea presented by the patient led her to assume the position of sitting for long periods, which justified that the edema was more pronounced in this dependent area.
The fact that there was no recurrence of edema in later paracentesis raises doubts about the true etiology of this vulvar edema, which resolved without sequelae.
REFERENCES
1. Poppe K, Unuane D, D'Haeseleer M, Tournaye H, Schiettecatte J, Haentjens P, Velkeniers B. Thyroid function after controlled ovarian hyperstimulation in women with and without the hyperstimulation syndrome. Fertil Steril 2011;96(1):241-245. [ Links ]
2. Vavilis D, Tzitzimikas S, Agorastos T, Loufopoulos A, Tsalikis T, Bontis JN. Postparacentesis bilateral massive vulvar edema in a patient with severe ovarian hyperstimulation syndrome. Fertil Steril 2002;77(4):841-843. [ Links ]
3. Corbett S, Shmorgun D, Claman P; Reproductive endocrinology infertility committee. The prevention of ovarian hyperstimulation syndrome. J Obstet Gynaecol Can 2014;36(11):1024--1033. [ Links ]
4. Delvigne A, Rozenberg S. Epidemiology and prevention of ovarian hyperstimulation syndrome (OHSS): a review. Hum Reprod Update 2002;8;559-577. [ Links ]
5. Geva E, Jaffe RE. Role of vascular endothelial growth factor in ovarian physiology and pathology. Fertil Steril 2000;74;429-438. [ Links ]
6. AbdelHafez FF, Desai N, Abou-Setta AM, Falcone T, Goldfarb J. Slow freezing, vitrification and ultra-rapid freezing of human embryos: a systematic review and meta-analysis. Reprod Biomed Online 2010;20:209-222. [ Links ]
7. Coccia ME, Bracco GL, Cattaneo A, Scarselli G. Massive vulvar edema in ovarian hyperstimulation syndrome. A case report. J Reprod Med. 1995;40(9):659-660. [ Links ]
8. Benaglia L, Busnelli A, Somigliana E, Leonardi M, Vannucchi G, De Leo S, Fugazzola L, Ragni G, Fedele L. Incidence of elevation of serum thyroid-stimulating hormone during controlled ovarian hyperstimulation for in vitro fertilization. Eur J Obstet Gynecol Reprod Biol 2014;173:53-57.
9. Skweres T, Wójcik D, Ciepłuch R, Sliwiński W, Czech R, Gruszczyński W, Rogacki M. Thyroid dysfunction during severe ovarian hyperstimulation syndrome. A case report. Ginekol Pol 2014;85(6):472-475.
Endereço para correspondência | Dirección para correspondencia | Correspondence
Ana Galvão
Centro Materno Infantil do Norte
Centro Hospitalar do Porto
Porto, Portugal
E-Mail: ana.m.galvao@gmail.com
Recebido em: 12/07/2017
Aceite para publicação: 03/10/2017