Introduction
Peripheral artery calcification is prevalent among individuals with peripheral artery disease (PAD) and is generally most pronounced in the femoropopliteal territory. Severe calcification is mainly present in patients with diabetes mellitus, chronic kidney disease, and smokers.1-3
Vascular calcification is associated with an elevated risk of cardiovascular mortality and morbidity, which correlates with a poor prognosis.4,5 Furthermore, the extent of calcification may influence immediate technical success and outcomes following endovascular interventions,6-8 However, limited evidence exists regarding the impact of calcification severity on endovascular treatment (EVT) and their outcomes.6,9
This study aimed to assess the influence of the degree of femoropopliteal calcification on the EVT of patients with PAD.
Methods
The authors designed a retrospective, single-center, comparative study. The population includes all patients with PAD who underwent EVT by a single operator (RC) as a first limb revascularization procedure for femoropopliteal lesions only, from January 2023 to February 2024.
Patient data, procedure, and outcomes were collected from medical records. The disease extent was staged according to the GLASS (Global Limb Anatomical Staging) system after digital subtraction angiography (DSA) evaluation. Although some calcium scoring systems have been previously described, we considered it more appropriate to develop a new system that integrates the parameters essential for lesion patency.
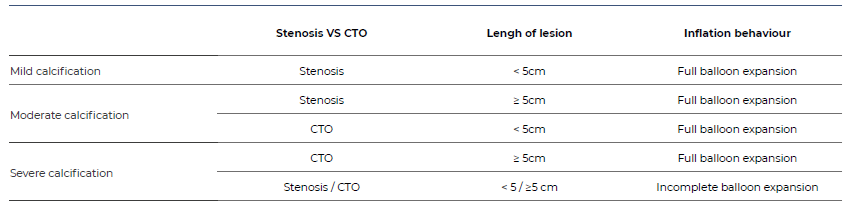
The calcification pattern and severity were assessed by the same single operator using three qualitative parameters from fluoroscopic and DSA evaluation: stenosis vs chronic total occlusion (CTO); length of lesion (<5cm vs >5cm); inflation behavior using a non-compliant standard balloon angioplasty (POBA). We defined the calcification degree as follows (Table 1):
Mild calcification included stenotic lesions with <5cm allowing complete expansion of POBA or absence of visible calcium at the target lesion;
Moderate calcification included stenotic lesions with ≥5cm or CTO lesions with <5cm, allowing complete expansion of POBA;
Severe calcification included CTO lesions ≥5cm allowing complete expansion of POBA and all lesions that prevent complete expansion of POBA;
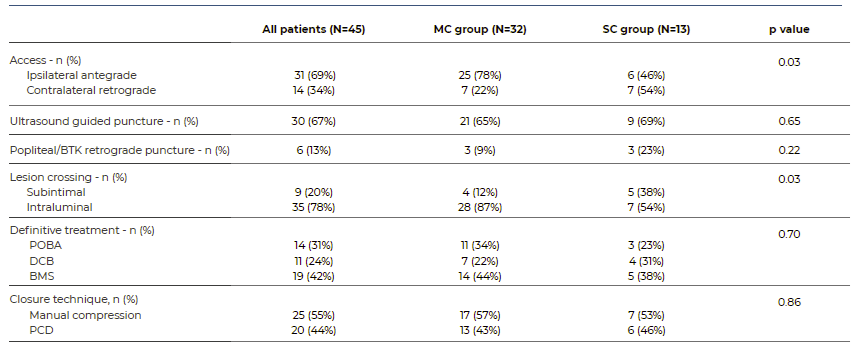
According to the degree of calcification, patients were divided into two groups: those with mild to moderate calcification (MC group) and those with severe calcification (SC group). They were compared regarding the following endpoints: rates of reintervention, rates of major amputation, and overall survival. We also analyzed the technical details of EVT in both groups, primarily focusing on the following aspects: access (ipsilateral antegrade or contralateral retrograde, fluoroscopic or ultrasound-guided puncture), popliteal or below-the-knee (BTK) retrograde puncture, cross-lesion pathway (intraluminal or subintimal), definitive treatment modality (POBA, drug coated balloon [DBC], bare metal stent [BMS]), and closure technique (compression or percutaneous closure device [PCD]).
Quantitative variables are expressed as mean ± standard deviations (SD) or as median (interquartile range - IQR), as appropriate. Qualitative variables are expressed as absolute values and percentages. Shapiro-wilk normative tests were used to assess the distribution pattern in quantitative variables. Student’s t test, one-way Anova and the respective non-parametric tests and χ2 test were used on univariate analysis. Kaplan Meier estimates were created to assess the effect of calcification severity on rates of reintervention, amputation and overall survival, and groups compared using the Log Rank test. A p-value of <0.05 was considered statistically significant. Statistical analysis was performed using SPSS software (version 26.0; SPSS, Chicago, IL, USA)
Results
The study included 45 patients, eight (18%) of whom presented with intermittent claudication (IC) and 37 (82%) with chronic limb threatening ischemia (CLTI). The MC group included 32 (71%) of the patients and the SC group included 13 (29%). Baseline characteristics are described in Table 2. The median time of follow-up was 7 months. The median femoropopliteal GLASS classification was 4 in both groups.
Table 2 Baseline characteristics of patients with femoropopliteal occlusive disease treated with endovascular techniques, included in this study
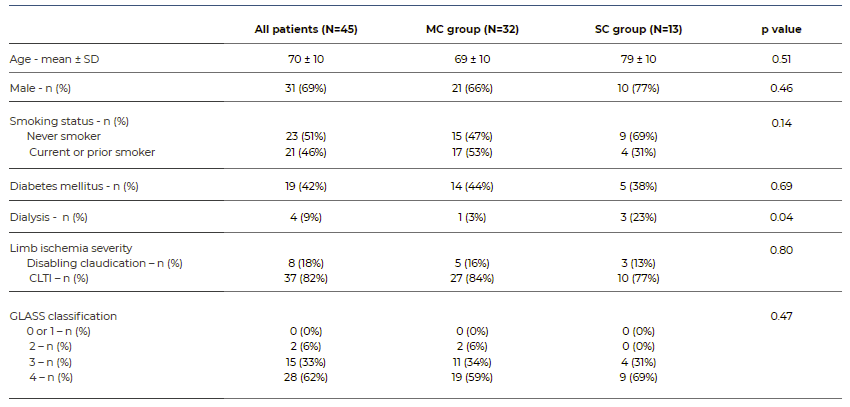
MC - Mild to moderate calcification; SC - Severe calcification; SD - Standard deviation; CLTI - Chronic limb threatning ischemia; GLASS - Global Limb Anatomical Staging System
Regarding the study endpoints, no statistically significant differences were found between groups. There were no statistically significant differences in reintervention rates. During the follow-up period, five patients in the MC group and two patients in the SC group required reintervention. In the MC group, most reinterventions (three out of five) occurred between months one and six of follow-up. In the SC group one reintervention occurred immediately postoperatively, and the other occurred after eight months of follow-up.
Kaplan Meier estimates for reintervention in the MC and SC groups were 3.1±3% and 7.7±7% at 1 month, 16±7% and 7.7±7% at 6 months and 23.6±10% and 26.2±17% at 11 months, respectively (p=0.97), Figure 1.
There were no statistically significant differences in amputation rates between groups. There were two major amputations in the MC group (6%) and one in the SC group (8%) after a median time of eleven months, all occurring within the first month of follow-up.
Amputation rates in the MC group and the SV group were 6.2±4% and 7.7±7% at 11 month (p=0.86), Figure 2.
No statistically significant differences were found in overall survival between groups.
During the follow-up period, one death occurred in each group. In the MC group, the death occurred after five months of follow up, whereas in the SV group it occurred after nine months. Overall survival rate was 100% at one month in both groups, 95±5% and 100% at six months, and 95±5% and 75±21% at 11 months, respectively (p=0.50), Figure 3.
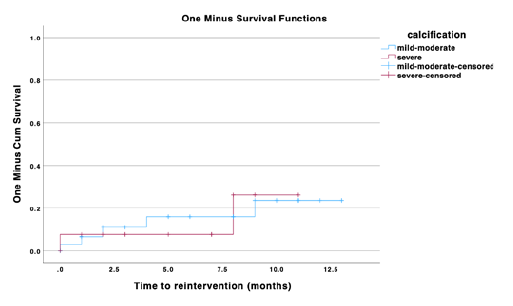
Figure 1 Kaplan Meier estimates for reintervention in patients treated for femoropopliteal occlusive disease using endovascular techniques, stratified by degree of calcification.
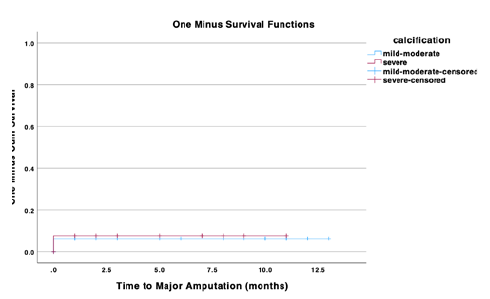
Figure 2 Kaplan Meier estimates for major amputation in patients treated for femoropopliteal occlusive disease using endovascular techniques, stratified by degree of calcification.
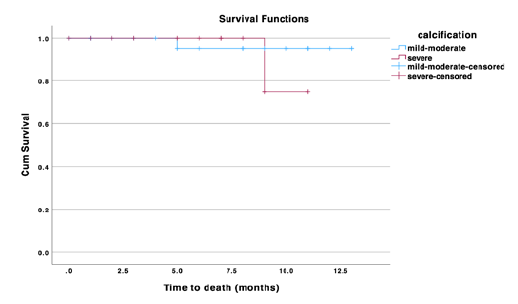
Figure 3 Kaplan Meier estimates for overall mortality in patients treated for femoropopliteal occlusive disease using endovascular techniques, stratified by degree of calcification.
The technical details of EVT are in Table 3. No statistically significant differences were found between groups regarding ultrasound guide puncture (p=0.65) and popliteal or below the knee (BTK) retrograde puncture (p=0.22). In the MC group, ultrasound guided puncture was employed in 66% compared to 69% in the SC group (p=0.65). Popliteal or BTK retrograde puncture was more frequently used in the SC group, accounting for 23% of cases, compared to 9% in MC group. However, 54% of patients in the SC group required contralateral retrograde access, which was significantly higher than the MC group, where only 22% needed contralateral retrograde puncture (p=0.03).
We found statistically significant differences in cross-lesion pathways. In the SC group, 38% (5 of 13 patients) required subintimal recanalization, whereas only 12% (4 of 32) in the MC group required this type of lesion crossing (p=0.048).
The definitive treatment modality did not differ significantly between both groups (p=0.70). POBA was used in 34% of patients in the MC group and in 25% of patients in the SC group, whereas DCB were employed in 33% of patients in the SC group and 22% of patients in the MC group. The deployment rates of BMS were similar between groups, with BMS used in 44% of cases in the MC group and 42% in the SC group.
Closure technique did not differ significantly between both groups (p=0.86), with similar rates of manual compression (57% vs. 53%) and PCD use (43% vs. 46%) for patients in the MC and SC groups, respectively.
Discussion
Advances in endovascular therapies over the past decades have expanded the range of percutaneous options available for managing peripheral vascular disease. However, infra-inguinal vascular calcification remains a significant challenge and a major obstacle in endovascular therapy of peripheral arterial disease, contributing to increased technical complexity.10
In this study, we included only patients with PAD who underwent EVT performed by a single operator. This approach was chosen to standardize the EVT procedure and minimize variability in results caused by operator differences in surgical techniques. Additionally, we restricted the study to patients treated within the last year to reduce potential confounding factors arising from changes in procedural approaches over time, given the continuous advancements in surgical techniques.
In the study population, 18% of included patients were claudicants, while the remaining patients had limb-threatening ischemia. The claudicant group had better general health and were non or former smokers, with limiting claudication.The degree of vascular calcification may also have a prognostic factor for outcomes after endovascular treatment of peripheral artery disease. Severe calcification can restrict sufficient luminal gain required for definitive treatment and optimal intervention, potentially impacting stent patency even after vessel preparation using modern techniques. In this study, no statistically significant differences were observed in the rates of reintervention, amputation, and death between the groups. We believe these results were achieved due to adequate vessel preparation for definitive treatment. However, considering the higher propensity for these outcomes in the SC group, we might obtain different results with a more extensive study population, thereby highlighting the poorer prognosis associated with complex lesions.
Although the influence of vascular calcification in endovascular treatment is well recognized, there is still no consensus on a calcium scoring system for peripheral arterial disease, and only a few studies have assessed the potential benefits of using a calcium scoring system. Among these, the Peripheral Arterial Calcium Scoring System (PACS) is the most commonly cited calcium classification. PACS assessed the degree of calcification at the target lesion by high intensity fluoroscopy and DSA using an antero-posterior projection. In our severity classification, we adapted the criteria from PACS (provided by fluoroscopy and DSA). Additionally, we incorporated the post-dilatation response to non-compliant POBA into our assessment. We believe these are the femoropopliteal calcium correlated aspects that may influence the target lesion treatment patency.
The response to balloon inflation was not separately assessed for the primary endpoints, although it was used as a criterion in the classification applied. Moreover, any stenosis in which full balloon expansion was not achieved was classified as a lesion of severe complexity and no significant differences in primary endpoints were observed based on the degree of severity. Therefore, we cannot conclude that full expansion of the POBA balloon predicts better outcomes. The main findings of this study suggest that the degree of the calcification significantly impacts the approach to endovascular treatment, particularly requiring more significant use of subintimal cross-lesion pathways and contralateral retrograde puncture for severely calcified lesions.
The association between severity of calcification and the choice of lesion crossing pathway can be explained by the challenge of intraluminal crossing, even with CTO guide wires, since the guidewire tends to follow a path of least resistance, which in calcified occlusions is the subintimal space between the intimal plaque and the adventitia.
Conversely, the association between contralateral retrograde puncture and the severity of calcified lesions may seem counterintuitive because in calcified lesions it is generally advantageous to have better device pushability, which is typically achieved when the puncture site is closer to the lesion. However, this finding can be explained by the correlation between calcification severity and more proximal lesions, such as CFA or ostial AFS lesions, which can make anterograde ipsilateral puncture and PCD unsafe. In these cases, contralateral access becomes advantageous and has a lower risk.
Regarding stent deployment, the criterion applied was bail-out stenting, performed in cases of flow-limiting dissection or significant residual stenosis (>30%). No statistically significant differences in the frequency of stent deployment were observed concerning lesion complexity, as rates of residual stenosis and flow-limiting dissection was similar between groups after balloon angioplasty.
This study has some limitations to be noted. It is a single-center retrospective study, which inherently has unavoidable bias. Also, the small sample size may contribute to type 2 statistical errors. Also, the source data depends on appropriate medical records and clinical follow-up. Finally, the vascular procedure selection depends on only one surgeon’s experience, potentially limiting generalizability.
Conclusions
Overall, this study suggests that calcification severity is independently associated with the type of access used and the cross-lesion pathway, with highly calcified lesions being more frequently associated with contralateral retrograde access and subintimal cross-lesion pathway. No difference in clinically relevant outcomes such as amputation rates, reinterventions or overall survival were identified.