Introduction
Chronic mesenteric ischemia (CMI) is an uncommon disease. Its insidious natural history and the variable nature of symptoms often lead to delayed diagnosis and treatment, with associated morbidity.1 The most common cause is progressive occlusive atherosclerotic disease of the visceral arteries, with the involvement of at least two mesenteric vessels in 90% of the symptomatic patients.2,3 Revascularization is generally recommended for symptomatic CMI patients.4,5Endovascular revascularization (ER) is considered the first-line therapy, however it has been associated with high rates of symptom recurrence, restenosis and reintervention.1Open revascularization (OR) is frequently reserved for failed endovascular therapy or anatomy unfavorable for endovascular treatment, although at the expense of higher perioperative morbidity.1 This study aimed to compare outcomes in patients treated for CMI with an OR or ER approach.
Methods
Study population
A prospectively maintained single-center database from a tertiary academic institution was retrospectively consulted for a study period of 10 years, from January 2013 to December 2023.
Thirty-three consecutive CMI patients were treated by an OR or ER approach during the study period. Were included all CMI patients with a diagnosis made by the presence of classic symptoms (postprandial pain, weight loss) and radiologic evidence of high-grade stenosis (>70%) or occlusion of at least one mesenteric artery. Non-atherosclerotic mesenteric disorders and acute mesenteric ischemia presentations were excluded.
Data acquisition
Baseline clinical data included sex, age, clinical presentation (postprandial abdominal pain, weight loss, diarrhea, nausea, vomiting, and the duration of symptoms), and comorbidities. Specific data included the morphological characteristics of the affected mesenteric arteries, surgical technical details, and follow-up. The institutional medical records were reviewed for data collection.
Diagnostic and operative details
All patients underwent computed tomography angiography (CTA) to assess individual anatomy and preoperative planning. ER was performed by primary stenting or percutaneous transluminal angioplasty (PTA) and/or stent of one or two mesenteric vessels. When possible, procedures under local anesthesia and percutaneous access were performed in an angiography suite. ER procedures were performed under general anesthesia in the operating room when brachial access via surgical exposure was planned, and when an expected longer procedural time was required for more complex procedures. OR was performed by bypass grafting, including aorto-mesenteric or iliac-mesenteric bypass.
Outcomes and definitions
The primary endpoint was the 30-day and 3-year reintervention. The secondary endpoints were 30-day and 3-year overall survival and reintervention-free survival. The follow-up period was the period from hospital discharge until the last available clinical examination. Reintervention was defined as revascularization due to restenosis/re-occlusion on a primarily successfully treated target artery associated with symptom recurrence. The surveillance strategy included clinical examination and duplex ultrasound of the visceral arteries. Clinical examination was performed at 1 month, 6 months, and annually after revascularization. Duplex ultrasound was performed before discharge, 6 months after revascularization, and then annually. If restenosis/re-occlusion on duplex ultrasound was associated with symptom recurrence, diagnostic angiography was conducted for additional evaluation.
Statistical analyses
Patients were analyzed as treated in two groups: ER and OR. Dichotomous variables were recorded as absolute frequencies (number of cases) and relative frequencies (percentages). Continuous data are presented as means, and non-symmetrical data are presented as medians. Normally distributed continuous variables were compared using the Student’s t-test, while non-normally distributed variables were compared using the Mann-Whitney U test. Reintervention and survival rates were estimated using the Kaplan-Meier model and compared with a Log-Rank test. A two-sided p-value ≤ .05 was considered statistically significant, and a 95% confidence interval was employed for the analysis. Statistical analyses were performed using IBM SPSS for Mac, Version 28.0 (IBM Corp., Armonk, NY).
Results
Patients
The study included 33 patients, 60% males (n=20), with a mean age of 67 (range 45-88) years. The treatment modality of CMI was ER in 27 (81%) and OR in 6 patients (22%).
Weight loss and postprandial pain were present in 100% of the OR group patients and 96% of the ER group patients. Less common symptoms were nausea, vomiting, and diarrhea. Symptoms were present for over 6 months in 63% of ER and 50% of OR patients. Classic cardiovascular risk factors and comorbidities were common in this sample, with hypertension, hyperlipidemia, and smoker status being the most frequently observed. A trend for higher prevalence of cardiovascular risk factors in OR patients was observed, such as hypertension (100% vs. 59%), hyperlipidemia (100% vs. 63%), and coronary heart disease (33% vs. 26%), but these differences were not statistically significant. Cerebrovascular disease was more prevalent in OR patients (50% vs 7%) with statistical significance (p=.031). Demographic and clinical details of patients are presented in Table 1.
Table 1 Patient demographics, comorbidities, and clinical presentation of patients with chronic mesenteric ischemia treated with endovascular and open revascularization
| ER (n=27) | OR (n=6) | Total (n=33) | p value | |
|---|---|---|---|---|
| Age (years) - mean (SD) | 67 (45-88) | 66 (46-81) | 67 (45-88) | .797 |
| Male - N (%) | 18 (67) | 2 (33) | 20 (61) | .182 |
| Hypertension - N (%) | 16 (59) | 6 (100) | 22 (67) | .077 |
| Hyperlipidemia - N (%) | 17 (63) | 6 (100) | 23 (70) | .145 |
| Diabetes mellitus - N (%) | 7 (26) | 0 | 7 (21) | .301 |
| Coronary heart disease - N (%) | 7 (26) | 2 (33) | 9 (27) | - |
| Previous stroke/ TIA - N (%) | 2 (7) | 3 (50) | 5 (15) | .031 |
| Peripheral arterial disease - N (%) | 8 (30) | 1 (17) | 9 (27) | - |
| Chronic kidney disease - N (%) | 3 (11) | 0 | 3 (9) | - |
| COPD - N (%) | 0 | 1 (17) | 1 (3) | .182 |
| Smoker status - N (%) | 25 (93) | 4 (67) | 29 (88) | .142 |
| Weight loss - N (%) | 26 (96) | 6 (100) | 32 (97) | - |
| Post prandial pain - N (%) | 27 (100) | 6 (100) | 33 (100) | - |
| Nausea and vomiting - N (%) | 0 | 1 (17) | 1 (3) | .182 |
| Diarrhea - N (%) | 7 (26) | 1 (17) | 8 (24) | - |
| Symptom onset > 6 months - N (%) | 17 (63) | 3 (50) | 20 (61) | .604 |
ER: endovascular revascularization; OR: open revascularization; TIA: transient ischemic attack; COPD: chronic obstructive pulmonary disease.
Morphological characteristics
Of the total population, four patients (12%) presented one-vessel disease and 29 patients (88%) had multi-vessel disease, defined as involvement of at least two mesenteric vessels. The superior mesenteric artery (SMA) was affected in 32 patients (97%), the celiac trunk (CT) in 21 patients (78%) and the inferior mesenteric artery (IMA) in 22 patients (81%). In one-vessel disease, the SMA was involved in all cases. Longer SMA lesions (>2cm) were more common in the OR group (100% vs 44%, p=0.027). In both groups, CT lesions were short (< 2cm) and located less than 2cm from the origin. Lesions longer than 2 cm were present in 7% of the patients of the ER group, Table 2.
Operative details
In the ER group (n=27), 26 patients underwent SMA single-vessel revascularization with stent angioplasty: 23 patients with covered stents, three patients with bare-metal stents and one patient with a drug-eluting stent. In one patient a two-vessel target revascularization was performed with SMA stenting with a covered stent and balloon angioplasty for ostial high-grade stenosis of IMA. The use of covered stents was the standard of treatment. Uncovered stents were chosen for high-grade stenosis for which a lower profile was unavailable for covered stents or for preservation of critical collaterals. Specifically, in one case, a drug-eluting stent was used and extended proximally with a second drug-eluting stent in a woman presenting with SMA restenosis. In 70% of the patients, the brachial access was used, in 19% the femoral access and in 11% of the patients the SMA stenting was performed by retrograde open mesenteric stenting (ROMS), Table 3.
When brachial access was chosen, it was performed via brachial artery exposure in 6 cases and via brachial percutaneous access in 13 cases. In brachial percutaneous access, closure devices were used in two cases: a Perclose ProGlide® in one case and the other an ANGIO-SEAL® without complications. For the remaining 11 brachial accesses, local hemostatic control was achieved with manual compression, two of which were complicated by local hematoma without surgical revision. No access wound-related complications were registered for cases of brachial exposure. The femoral accesses were all percutaneous and closed with a closure device: Perclose ProGlide® in 3 cases and ANGIO-SEAL® in 3 cases, without complications. No access complications were reported in ROMS cases.
Table 2 Morphological vessel characteristics of patients with chronic mesenteric ischemia treated with endovascular and open revascularization
| ER (n=27) | OR (n=6) | p value | |
|---|---|---|---|
| Single-vessel disease - N (%) | 3 (11) | 1 (17) | - |
| Multi-vessel disease (≥ 2 vessels) - N (%) | 24 (89) | 5 (83) | - |
| Superior mesenteric artery | |||
| High-grade stenosis/occlusion - N (%) | 26 (96) | 6 (100) | .472 |
| Lesion <2cm from origin - N (%) | 26 (96) | 6 (100) | - |
| Lesion length > 2 cm - N (%) | 12 (44) | 6 (100) | .027 |
| Celiac trunk | |||
| High-grade stenosis/ occlusion - N (%) | 17 (63) | 4 (67) | .238 |
| Lesion <2cm from origin - N (%) | 27 (100) | 6 (100) | - |
| Lesion length > 2 cm - N (%) | 2 (7) | 0 (0) | - |
| Inferior mesenteric artery | |||
| High-grade stenosis/Occlusion - N (%) | 17 (63) | 5 (83) | .637 |
High-grade stenosis as degree of stenosis > 70%. ER: endovascular revascularization; OR: open revascularization
Table 3 Operative technical details for open and endovascular revascularizations of patients with chronic mesenteric ischemia
| ER (n=27) | OR (n=6) | |
|---|---|---|
| Access site | ||
| Femoral - N (%) | 5 (19) | - |
| Brachial - N (%) | 19 (70) | - |
| ROMS - N (%) | 3 (11) | - |
| Treatment details | ||
| PTA only* - N (%) | 1 (4) | - |
| Bare-metal stent - N (%) | 3 (11) | - |
| Covered stent - N (%) | 23 (85) | - |
| Drug-eluting stent - N (%) | 1 (4) | - |
| Aorto-mesenteric bypass - N (%) | - | 1 (17) |
| Iliac-mesenteric bypass - N (%) | - | 4 (67) |
| Iliac-mesenteric bypass+IMA reimplantation - N (%) | - | 1 (17) |
| PTFE/Dacron prosthetic graft - N (%) | - | 5 (83)/1 (17) |
| Vessel revascularization | ||
| Single-vessel (SMA) - N (%) | 26 (96) | 5 (83) |
| Two-vessel (SMA+IMA) - N (%) | 1 (4) | 1 (17) |
ROMS: retrograde open mesenteric stenting; PTA: percutaneous transluminal angioplasty; PTFE: polytetrafluoroethylene ER: endovascular revascularization; OR: open revascularization; SMA: Superior mesenteric artery; IMA: Inferior Mesenteric Artery *Case of two-vessel target revascularization in whom SMA stenting with a covered stent and balloon angioplasty for ostial high-grade stenosis of IMA were performed.
In the OR group (n=6), all patients were submitted to a retrograde mesenteric bypass with proximal anastomosis at the infra-renal aorta or right iliac artery level. In 5 patients, a single-vessel revascularization targeting SMA was performed: one aorto-mesenteric bypass and four iliac-mesenteric bypasses. In one patient, a two-vessel mesenteric revascularization was performed with iliac-mesenteric bypass and IMA reimplantation to the bypass graft. It was used as a prosthetic conduct in all cases, Table 3.
Outcomes
The median hospital stay was two days (1-5) in the ER group and 12 days (4-12) in the OR group (p=.253). The median follow-up was 38 (9-72) months. In Kaplan-Meier analysis, there were no ER or OR reinterventions at 30 days and at 3 years, the reintervention for ER was 18%, with no reinterventions in OR (p=.374), Figure 1.
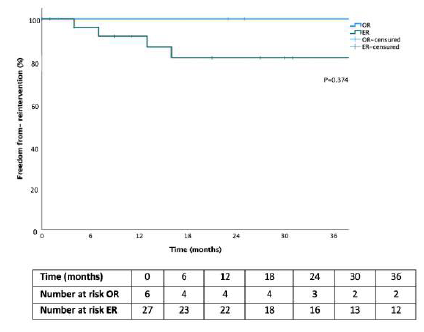
Figure 1 Cumulative Kaplan-Meier estimates for freedom from reintervention for chronic mesenteric ischemia patients treated with endovascular vs open revascularization OR - Open revascularization; ER - Endovascular revascularization
Reinterventions were indicated by symptom recurrence and target artery high-grade restenosis in three patients and occlusion in one patient, after SMA stenting. In all cases, the SMA prior morphological characteristics were lesions less than 2cm from the origin and longer than 2cm. Three patients were submitted to ER reintervention, and one patient to OR treatment, Table 4.
In two patients, proximal SMA stenosis was treated with proximal extension with a covered stent, Figure 2. One patient with SMA stenting stenosis was submitted to SMA angioplasty with a DCB and CT stenting with a covered stent, Figure 3. In one patient with SMA stent reocclusion, a two-vessel revascularization with aorto-mesenteric bypass and IMA re-implantation was performed due to IMA ostial high-grade stenosis and previous evidence of an incomplete right aorta’s arch. Thirty-day and 3-year overall survival in the OR and ER groups were 67% vs. 93% and 44% vs. 87%, respectively (p=.015, Figure 4).
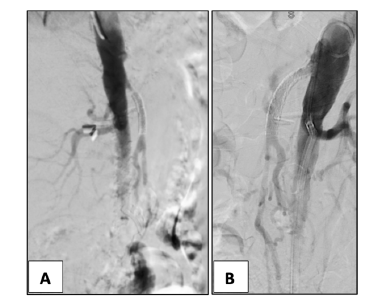
(A) restenosis of the SMA is noted; (B) treatment with proximally extension with a covered-stent Advanta 5x38mm to SMA ostium.
Table 4 Details of patients submitted to reintervention for symptomatic restenosis or occlusion of chronic mesenteric ischemia revascularization
| Patient | Primary intervention | Time to restenosis/occlusion (months) | Secondary intervention |
|---|---|---|---|
| 1 | SMA stenting (covered stent) | 7 months to restenosis | CT stenting (covered stent) PTA SMA (DCB) |
| 2 | SMA stenting (DES) | 16 months to restenosis | SMA stenting (proximal extension covered stent) |
| 3 | SMA stenting (covered stent) | 13 months to occlusion | Aorto-mesenteric bypass IMA reimplantation |
| 4 | SMA stenting (covered stent) | 4 months to restenosis | SMA stenting (proximal extension covered stent) |
SMA - superior mesenteric artery; CT - celiac trunk; IMA - inferior mesenteric artery; DES - drug eluting stent; DCB - drug-coated balloon
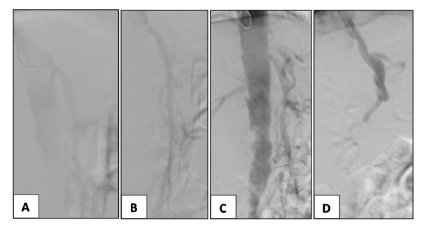
Figure 3 Intra-operative angiography of a restenosis case after SMA stenting (case 2). Proximal stent stenosis (A) dilated with a drug-eluting balloon 6x40 mm (B). CT high-grade stenosis (C) treated with a covered stent Advanta 7x22mm (D).
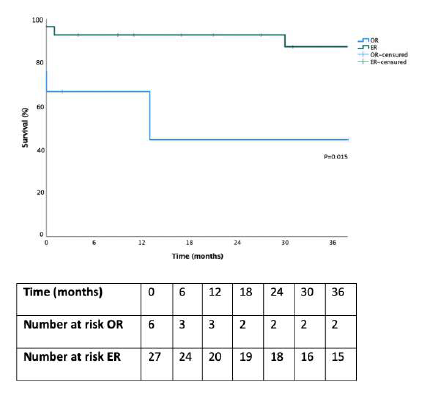
Figure 4 Kaplan-Meier estimate of overall survival for patients treated with endovascular and open revascularization in chronic mesenteric ischemia. ER - Endovascular Repair; OR - Open Repair
In the ER group, 30-day mortality recorded two deaths attributable to respiratory failure in one case and due to sepsis in another. In the OR group, 30-day mortality recorded two deaths, one due to respiratory failure and the other due to gangrenous cholecystitis.
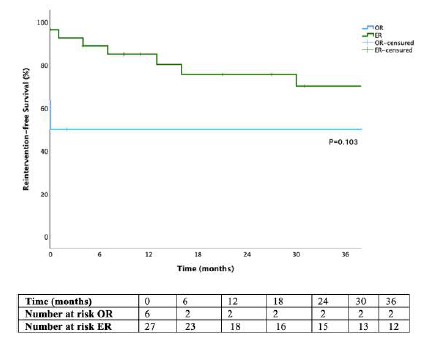
The causes of death registered beyond 30 days, one for each group, occurred outside of the center of the study, not allowing for the collection of data regarding the cause of death. Considering 3-year reintervention-free survival in the OR and ER groups were 50 % vs. 70% (p=.103, Figure 5).
Discussion
In the presented consecutive cohort of patients suffering from CMI, there was no significant difference for the primary endpoint regarding reintervention rate during a median follow-up of 38 months. As in other studies, most likely due to the small sample size, we only observed trends for a higher reintervention rate in the ER group.3 One of the identified problems of endovascular treatment is vessel patency.6 Acceptable morphological characteristics for endovascular treatment would be focal, short lesions with minimal to moderate calcium. As such, the type of endovascular treatment is of utmost importance for vessel patency, with covered stents being associated with higher patency.5 Previous studies have compared the standard mesenteric stenting with bare-metal stents versus covered stents to establish an optimal endovascular approach. Oderich et al reported that after stenting with bare-metal stents, in-stent restenosis occurred in 47% of patients at three years, and 44% required a reintervention within three years. The same study presented for the use of covered stents an 8% 3-year in-stent restenosis rate and only 9% reintervention within three years.7 More recently, the CoBaGi trial reported at 24 months, a primary patency of 81% for covered stents and 49% for the bare-metal stents.8
The use of covered stents was the standard of treatment in our population. The use of uncovered stents was chosen for high-grade stenosis for which a lower profile was not available for covered stents. Although three of four of our reintervention cases corresponded to SMA stenting using covered stents, accounting for a reintervention rate of 18% at 3 years, in all cases, SMA lesions presented characteristics that could be unfavorable to an endovascular approach, compromising patency. Ostial lesions and with longer lengths are associated to endovascular failure.9 Therefore, reintervention rates and worse primary patency rates must be accounted when considering an endovascular-first strategy.10,11
In our population we observed a trend to lower reintervention rate in the OR group. Although a small sample, this trend is expected according to other studies in the field, being mesenteric bypass associated with lower rates of reintervention and freedom from symptom recurrence.12-14
In our study, despite the high prevalence of multi-vessel disease, a single-vessel revascularization was performed more frequently. The role of two-vessel revascularization vs single-vessel revascularization is not well established, with studies showing no superiority regarding patency and freedom from symptom recurrence.15-16 In our population, a two-vessel revascularization was performed in two cases for which visceral collaterality was deemed insufficient.
Thirty-day and 3-year overall survival was lower in the OR group with statistical significance. Patient profiles had typical arteriosclerotic risk factors and were marked by multiple comorbidities. Overall, patients in the OR group had higher perioperative hypertension, hyperlipidemia, coronary disease, and cerebrovascular disease, which may explain the lower survival in this group. However, the sample size probably turns the extrapolation of these results biased. According to the literature, the type of revascularization has not been shown to affect survival, with studies showing no difference between the two modalities.4,17
This study as several limitations. It is a retrospective single-center study, generating bias linked to a retrospective data collection. Additionally, the sample size and the disparity in the number of patients between the two groups limit the ability to make significant comparisons.
Conclusion
According to this study, we should expect lower survival in CMI patients who undergo OR, probably related to the higher burden of atherosclerotic disease observed in this group, reflected in a higher prevalence of cardiovascular and cerebrovascular disease. However, robust studies must be conducted so that mortality relations can be made accurately. The higher survival rate after ER should be put into perspective, considering the trend toward higher reintervention rates.