Introduction
Spinal cord ischemia (SCI) is an infrequent but distressing complication following fenestrated/branched endovascular (f/bEVAR) thoraco-abdominal (TAAA) and complex abdominal aortic aneurysm (CAAA) repair leading to an important impact in morbidity, mortality and quality of life of affected patients.1-3 Reported rates of SCI vary widely between studies, ranging from 1% to 36% in current literature.4
Prior studies have evaluated various risk factors for its development, such as extension of aortic coverage, prior aortic surgery and internal iliac artery occlusion, as well as several measures for its prevention, including prophylactic cerebrospinal fluid drainage (CSFD), staged procedures, temporary sac perfusion and staged occlusion of segmental arteries to achieve ischemic preconditioning and permissive intra- and post-operative hypertension.1-3,6-7 However, despite much effort in developing strategies to minimise the incidence of this complication, results are still conflicting, and the best protocol is still lacking.
The aim of this study is to analyse the incidence of SCI after endovascular treatment of TAAA and CAAA in our centre and to provide a descriptive analysis of its risk factors and outcomes.
Methods
A single-centre retrospective study of a prospectively maintained database from a tertiary vascular surgery institution was conducted.
Study population
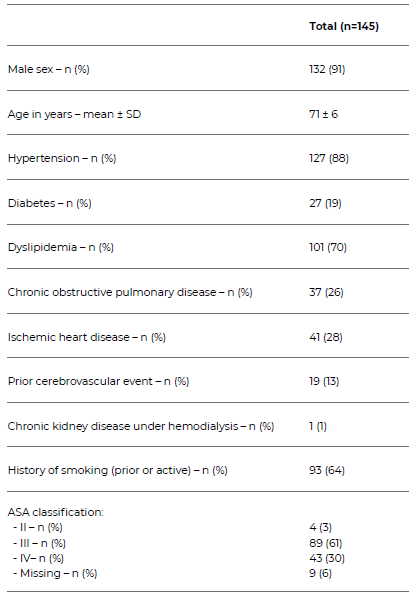
One hundred and forty-five patients with degenerative TAAA or CAAA who underwent endovascular repair with a fenestrated/branched endograft between June 2010 and February 2025 were included. Patients with TAAA of Crawford extent I-V, suprarenal, pararenal, and juxtarenal abdominal aortic aneurysms were included. Indications for repair included asymptomatic patients with aneurysm diameter above the European Society for Vascular Surgery threshold for repair, as per the Guidelines on the Management of Abdominal Aorto-Iliac Artery Aneurysms, and urgent/emergent repair for symptomatic or ruptured aneurysms.8Other indications for repair included post-dissection TAAA, type Ia or Ib endoleaks following EVAR or TEVAR, respectively, and aortic false aneurysms. Custom-made devices with branches and/or fenestrations, and off-the-shelf devices (Cook Medical, Inc., Indiana, USA, and Artivion, Inc., Florida, USA) were considered. Physician-modified endografts and other endovascular techniques, such as parallel grafts, were excluded from this study.
Data collection and definitions
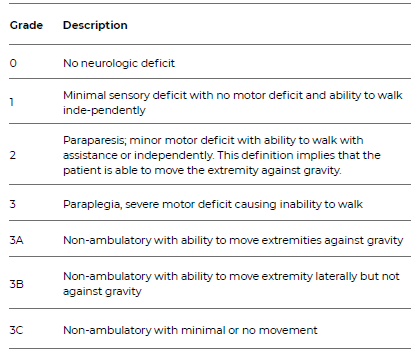
Institutional medical record review was performed, and baseline characteristics, clinical presentation, and peri-procedural and follow-up data were obtained. Spinal cord ischemia (SCI) was defined according to the Society for Vascular Surgery’s (SVS) reporting standards as a new-onset neurological (motor or sensory) deficit after endovascular repair.8 Immediate SCI was defined as any deficit occurring during surgery and identified at the end of the procedure or in the first examination after. Delayed SCI was defined as any deficit occurring after a normal post-operative examination. Classification and grading of symptoms were performed according to Table 1.
Table 1 Spinal cord ischemia classification of symptoms, according to the Society for Vascular Surgery’s reporting standards8
Reintervention was defined as any subsequent surgical intervention due to a post-operative complication. Intentional staged interventions were not considered reinterventions.
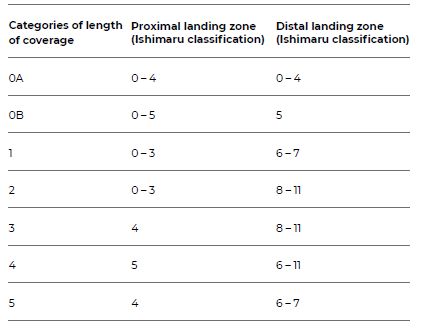
The extent of aortic coverage was defined according to the classification by Aucoin et al., based on the landing zone system previously defined by Ishimaru.1 This classification method, detailed in Table 2, correlates with the Crawford anatomical classification of TAAA with the addition of two categories (0A and 0B).
Table 2 Categories of length of aortic coverage created by Aucoin et al., based on Ishimaru’s landing zone system1
Endpoints
The primary endpoint was the occurrence of post-operative SCI. Secondary endpoints were 30-day reintervention and 30-day mortality rates. An additional analysis was performed comparing patients who developed SCI with those who did not.
SCI prevention protocol
An institution-based SCI prevention protocol was implemented in all cases. All procedures were performed under weight-adjusted unfractionated heparin (100 UI/kg), controlled with an activated clotting time (ACT) goal of 250-300 seconds. A prophylactic cerebrospinal fluid drainage (CSFD) was selectively placed in patients considered high-risk for SCI. Intra- and post-operative strict monitoring was performed considering a target cerebrospinal fluid pressure < 10-15 mmHg with a maximum drainage rate of 10-15 ml/h for a medullary perfusion pressure goal of > 70 mmHg.
Other optimisation measures to prevent SCI included maintaining a mean arterial pressure > 90 mmHg using vasopressors if needed, a haemoglobin > 10 g/dl and a platelet count > 100x109/L threshold for transfusion and a target peripheral oxygen saturation > 98%. Procedural staging was performed based on an estimated SCI risk following multidisciplinary assessment by the vascular surgeons and the anesthesiologists. In the immediate post-operative period, patients were transferred to an intensive care unit for strict surveillance. In cases of new-onset neurological deficits, a rescue CSFD was placed if the patient did not already have one in place.
Prophylactic CSFD were clamped at 48 hours (h) and removed at 72h post-surgery if no neurological symptoms occurred.
Statistical analysis
Descriptive statistics were used to report data. Normal continuous data were presented as mean ± standard deviation. Kolmogorov-Smirnov and Shapiro-Wilk tests were used to assess the normal distribution of data, and t-test or Mann-Whitney U statistics were used, respectively, to compare continuous Gaussian and non-Gaussian data. Dichotomous variables are expressed as counts (percentages) and compared between groups using Pearson’s chi-square test.
All statistical tests were two-sided and considered statistically significant when the p-value was <0.05. All analyses were performed using IBM SPSS Statistics version 29.0 software.
Results
Patient demographics and characteristics
One hundred and forty-five patients were included in this study. Most patients were male (91%) with a mean age of 71 ± 6 years. The most common cardiovascular risk factors were hypertension (88%), dyslipidemia (70%) and ischemic heart disease (28%). Table 3 describes the patients’ demographics and characteristics.
Anatomical characteristics and periprocedural details
The majority of patients (51%) presented with a TAAA, 10% being post-dissection. 74% were Crawford type I-III/V. Of the 39% of patients presenting with a CAAA, most were juxtarenal aneurysms (63%).
Mean aneurysm diameter was 66 ± 14 mm. The main indication for surgery was elective repair (n = 138, 95%), with seven cases (5%) being performed in an urgent/emergent timing, Table 4.
Outcomes and SCI subgroup analysis
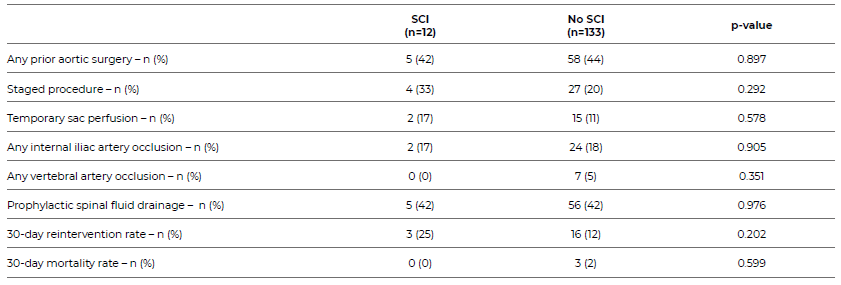
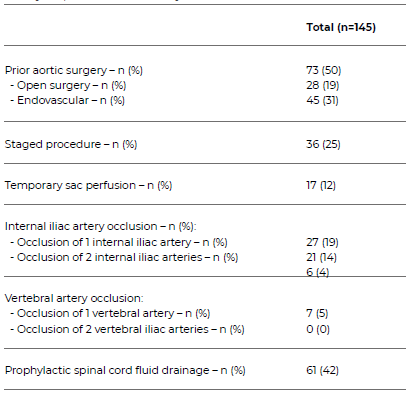
Risk factors for SCI were evaluated in all patients and are described in Table 5. Half had a history of aortic surgery, mostly endovascular (n = 45, 62%).
A staged procedure was performed in 21% of patients, and 12% had temporary sacral perfusion. Internal iliac artery occlusion was present in 27 (19%) and vertebral artery occlusion in 7 (5%) of patients. According to our SCI prevention protocol, a prophylactic CSFD was placed in 61 patients (42%).
Table 4 Anatomical characteristics and periprocedural details of patients undergoing fenestrated or branched endovascular repair of thoraco-abdominal or complex abdominal aortic aneurysms, included in the study.
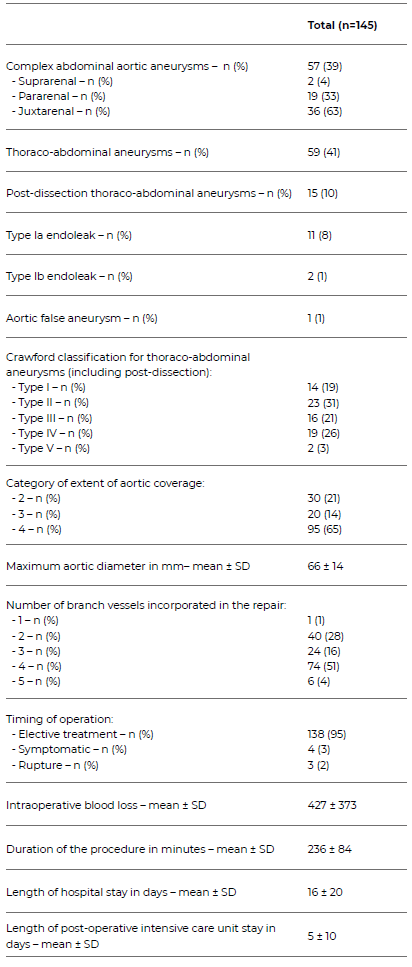
Table 5 Risk factors for spinal cord ischaemia and preventative procedural measures adopted in patients undergoing fenestrated or branched endovascular repair of thoraco-abdominal or complex abdominal aortic aneurysms, included in the study.
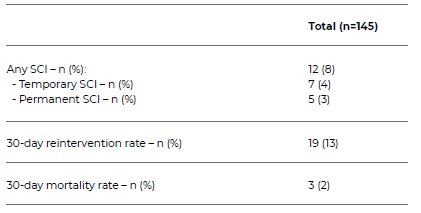
SCI, defined as any motor or sensitive deficit, occurred in 12 patients (8%), Table 6. Of these, 7 (58%) had temporary symptoms, being completely recovered at discharge, and 5 (42%) had permanent SCI with or without any improvement before discharge. Regarding secondary outcomes, the 30-day reintervention rate was 13% and 30-day mortality was 2%.
Table 6 Outcomes of patients undergoing fenestrated or branched endovascular repair of thoraco-abdominal or complex abdominal aortic aneurysms, included in the study.
An individual analysis of all patients who developed SCI was performed and is represented in Table 7. Of the 12 patients, half presented with a TAAA, five with a CAAA and one with a post-dissection TAAA. All TAAA cases were Crawford types I-III. Most patients (83%, n = 10) presented with delayed SCI.
Regarding patients with Grade 3 SCI (n = 7), a complete recovery was observed in three patients, one patient experienced partial recovery and three did not recover. One patient presented with Grade 2 ischemia and did not improve before discharge. The three patients who presented with Grade 1 ischemia had complete recovery.
A prophylactic CSFD was placed preoperatively in five patients (42%) who developed SCI. After symptom onset, 58% (n = 7) of patients required a rescue CSFD.
All SCI patients who had a preprocedural CSFD placed had complete recovery of symptoms. Of all patients with either a prophylactic or rescue CSFD placed, only one complication (an intradural hematoma) occurred. A subgroup analysis was performed comparing SCI to no-SCI patients, Table 8. No differences were found regarding comorbidities, except for chronic kidney disease under hemodialysis which was present in one patient out of the 145 enrolled in this study, p < 0.001. A longer length of both total hospital and ICU stays was found in SCI patients - p < 0.001 and p = 0.025, respectively. No differences concerning SCI risk factors, preoperative CSFD placement and 30-day reintervention and mortality rates were found between SCI and no-SCI patients, Table 9.
Table 7 Individual analysis of patients with spinal cord ischemia after fenestrated or branched endovascular repair of thoraco-abdominal or complex abdominal aortic aneurysms, included in the study
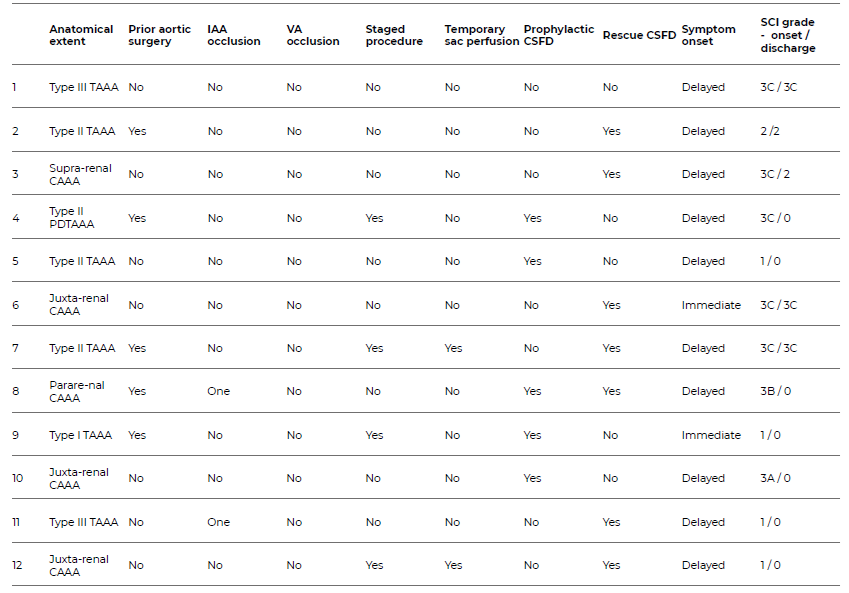
CAAA: complex abdominal aortic aneurysm; CSFD: cerebrospinal fluid drainage; IAA: internal iliac artery; PDTAAA: post-dissection thoraco-abdominal aortic aneurysm; SC: spinal cord ischaemia; TAAA: thoraco-abdominal aortic aneurysm; VA: vertebral artery
Table 8 Baseline and anatomical characteristics and periprocedural details of SCI and non-SCI patients.
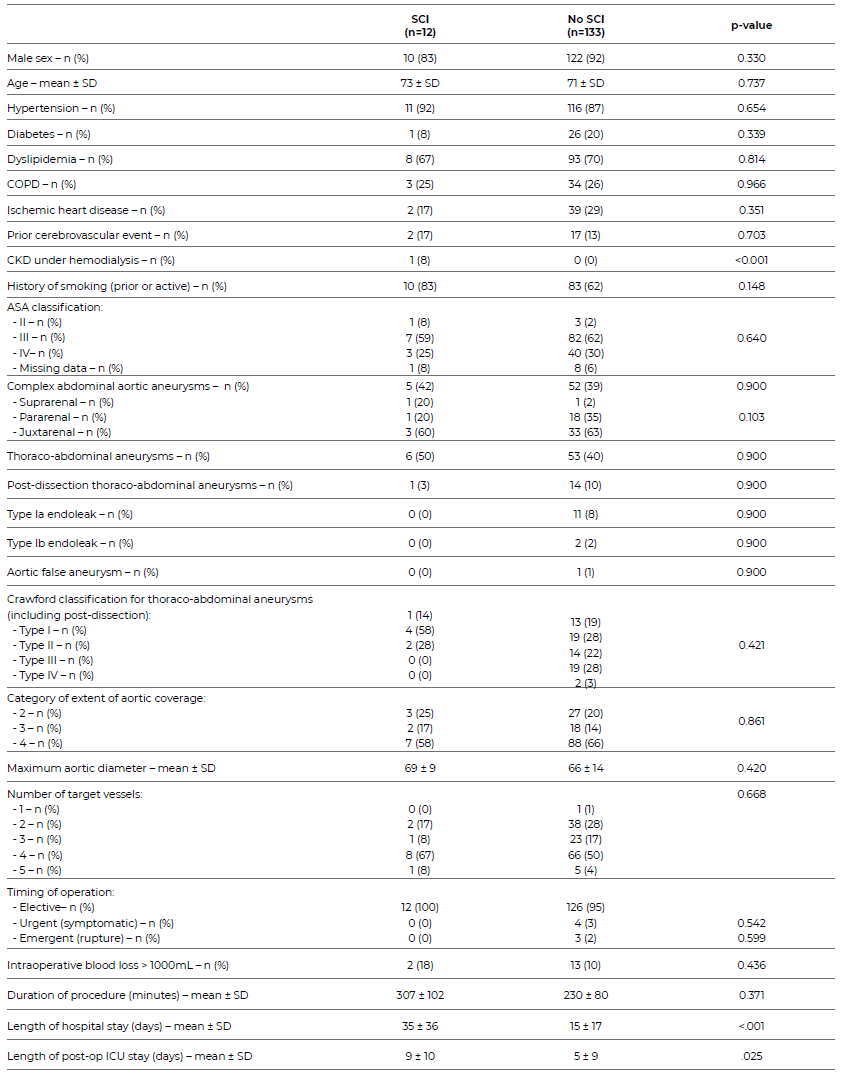
COPD: Chronic obstructive pulmonary disease; CKD: Chronic kidney disease; ICU: Intensive care unit
Discussion
In our study, we report an 8% incidence of all SCI, with permanent paraplegia occurring in 2% of all patients, which is a low rate and on par with previous studies. Complete paraplegia (SCI SVS Grade 3) was the most common symptom presentation with either complete or partial recovery in most patients.
A delayed presentation of SCI was the most common timing of symptom onset in our study. Previous reports are in line with this regarding endovascular TAAA repair, contrasting with a higher immediate SCI rate in open surgical repair.4 Various pathophysiological mechanisms have been proposed for delayed SCI, such as post-operative hypotension, hypoxia or diminished oxygen delivery due to anaemia, cardiac dysrhythmias, cardiac failure or sepsis.5Procedure-associated causes may include atheroembolism to segmental vessels from aortic plaques dislodged during the procedure or late-occlusion of type II/III post-operative endoleaks.9 For this reason, it is recommended to maintain a prolonged strict clinical surveillance in patients undergoing endovascular repair of complex aortic aneurysms.
In our study, we did not find significant differences between previously reported SCI risk factors in patients with and without SCI. A history of prior aortic surgery was present in half of all patients, including five (42%) SCI patients and 58 (44%) non-SCI patients. Likewise, despite the extent of aortic coverage being identified as one of the most important risk factors for SCI development, it did not reach statistical significance in our study, likely due to the small number of events. Prior studies show a lower incidence of SCI in patients undergoing endovascular repair of CAAA and type IV TAAAs compared with types I-III/V.1,5-7 However, in our study, 42% of SCI patients (5/12) presented with a CAAA, suggesting the importance of not disregarding rigorous surveillance in these patients. Internal iliac and vertebral artery coverage is also associated with a higher risk of immediate-onset SCI and lack of neurological recovery, given their role in the spinal cord perfusion network.5,10 In our study, 27 patients (19%) had one or both internal iliac arteries occluded, and two of these patients developed delayed SCI with complete recovery of symptoms. None of the patients with an occluded vertebral artery developed SCI.
Other factors, such as procedure duration, intra-operative blood loss, patient comorbidities, and surgery in an urgent/emergency setting, have also been described as associated with a higher risk of SCI.1,5-6,11 However, we did not find significant differences in these parameters between SCI and no-SCI patients. Chronic kidney disease under haemodialysis was higher in SCI patients (p < 0.001). This was present in only 1 patient among the 145 included in the study; therefore, no conclusions may be drawn from this association.
Regarding preventive measures, a prophylactic CSFD was placed in 42% of patients. A gradual decline in its use has been reported in the literature, in favour of a policy of urgent rescue drainage at the onset of post-operative symptoms.12 Nonetheless, it remains part of our preventive protocol for SCI, with satisfactory results, which explains its high rate of use. Additionally, all patients who developed SCI and had a prophylactic CSFD experienced complete recovery of symptoms. In contrast, 4 of the 7 patients who required a rescue CSFD did not recover and were paraplegic at the time of discharge. However, no significant difference in prophylactic CSFD placement was found between SCI and no-SCI patients (p = 0.976), so no definitive conclusions can be drawn regarding its role in our population. It is also important to note that CSFD is not without complications, as an intradural haematoma occurred in our series.11-12
Procedural staging has also been noted as a protective measure against SCI development, due to optimisation of the spinal collateral network, despite a low but present risk of complications, namely rupture.5,11-12 Four of the 12 patients who developed SCI in our study underwent staged endovascular repair, suggesting that, despite its preventive role in SCI, it does not completely avoid the risk.
Literature reports a higher rate of long-term mortality in patients who develop permanent SCI, with no significant differences in the perioperative period, which corroborates our findings of similar 30-day mortality in SCI and no-SCI patients1-2,6,9 Likewise, similar to other studies, SCI patients in our study had both longer hospital and intensive care unit length of stay.2,10
This study has certain limitations. Firstly, it is a single-centre, retrospective study, which is associated with inherent biases and does not allow for firm conclusions to be drawn. Secondly, the small sample size and limited number of events limit outcome analysis and the ability to identify risk factors and predictors of SCI.
Conclusion
In this study, SCI occurred in both TAAAs and CAAAs and manifested primarily as a delayed event, without differences in 30-day reintervention or mortality rates. Prophylactic CSFD placement may prevent permanent injury, despite the lack of statistically significant differences in findings. Prolonged, strict clinical monitoring should be performed in all these patients to allow timely rescue. Despite several risk factors and preventive measures previously described, the optimal protective approach remains unclear. Further studies are required to obtain more robust data to prevent this devastating complication.