INTRODUCTION
Chronic urticaria (CU) is a skin disorder defined by the presence of recurrent erythematous wheals and pruritus, with or without angioedema, for at least 6 weeks. Wheals can occur in any part of the body, last individually for less than 24 hours and blanch with pressure due to vasodilation and superficial dermal edema in their pathogenesis.1,2
This disease has a complex pathogenesis, mostly dependent on the discharge of pro-inflammatory molecules from mast cells present in the superficial dermis. When the symptoms of urticaria are induced by a stimulus, such as exercise, pressure, water or cold, it is classified as chronic inducible urticaria (CIU), but in most cases no trigger is found and it is defined as chronic spontaneous urticaria (CSU).1
Costa et al 2016 reported a Portuguese incidence of 1% of CU and the same study highlighted the economic burden of this disease, with an average cost of over 6000€ per 5 years to treat a single patient with severe CU.3
Treatment of CU is based on an integrated approach of several factors, including eviction of triggers, dermatological care and pharmacological therapy, it is however most times insufficient to control the symptoms and patients suffer a considerably negative impact on their quality of life (QoL). Therefore, assessment of their “silent suffering” is essential to understand the patients’ view of their disease and its consequences. Guidelines recommend the regular use of patient-reported outcomes (PRO) for this purpose.4,5Currently, there are three disease-specific instruments validated for CU: Urticaria Activity Score Over 7 Days (UAS7), Urticaria Control Test (UCT) and Chronic Urticaria Quality of Life Questionnaire (CU-Q2oL). In addition to these scales, others are also used in daily practice.5-8
The impact of disease on quality of life may not be directly related to its clinical severity, but to the stigma and discomfort caused, as urticaria has been associated with a negative emotional impact, leading to somatization, depression, anxiety, insomnia, stressful life events, and others.9-12A medical databases search revealed a high prevalence of psychosocial factors in patients with chronic spontaneous urticaria.13-15
According to the latest guidelines for urticaria, further research is needed in order to assess CU disease activity and its impact in patient’s QoL and both psychiatric and psychosomatic conditions. It has been pointed out the main need for future application of CU-Q2oL in different populations to obtain patients’ views of their disease, aid treatment decision-making and to help allocating resources.6,8
The aim of the present study is to estimate the impact of chro-nic urticaria on the quality of life of Portuguese patients and clarify in which domains they feel more affected. It is also our goal to identify how variables, such as age, gender, association with angioedema, CU severity and control, influence patients’ QoL and emotional state of depression and anxiety. Quality of life is the main outcome measure of this study, while emotional state of depression and anxiety are secon-dary outcome measures.
MATERIAL AND METHODS
We present an observational study conducted at the Urticaria consultation of the Dermatology Department of the Coimbra Univer-sity Hospital Centre, between December 2016 and December 2018. This work, included within a more extended study conducted at the Urticaria consultation, received the approval of the Ethics Committee of the Coimbra University Hospital Centre, and the National data protection authority.
The study sample comprised a total of 112 patients, both male and female, aged above 16 years, with a clinical diagnosis of CU who after detailed explanation of the objectives of the study signed the informed consent, and voluntarily participated after their regular appointment.
An anonymous questionnaire was made including: individuals’ characteristics (gender, age, duration of disease, education level, professional status and familiar situation), a score of clinical activity of urticaria over the last week (UAS7), a test to evaluate the control of urticaria (UCT), general and urticaria specific aspects on quality of life (respectively DLQI and CU-Q2oL), and questions related to the anxiety and depression state (Hospitalar Anxiety and Depression Scale - HADS), which will be furtherly detailed. Only patients able to understand the terms and to sign the term of free and informed consent were admitted to the study.
Patients read and answered the questionnaires alone, but the re-searcher remained available to clarify any possible questions. All patients answered the CU-Q2oL, but not all filled the other questions. A total of 110 patients filled the HADS and 108 the UAS7 questionnaire. A subsample of 40 patients additionally filled the UCT and DLQI scale. Not all patients filled all the sociodemographic data. The analysis was preformed respecting each subsample of patients.
Instruments used: Urticaria Activity Score Over 7 days (UAS7): A simple, validated, scoring system based on the assessment of itch intensity and number of hives. It is documented by the patient, once daily, for seven days, allowing doctors to access disease activity during the previous week. The summed scores, over a week, creates the weekly UAS (UAS7) with a score ranging from 0 to a maximum of 42. UAS7 scores are then categorized into five ranges describing ‘CSU health states’.3,16,17
Urticaria Control Test (UCT): The UCT is a validated instrument designed to assess the level of disease control in CU patients and thereby help guiding treatment decisions. It consists in four simple questions related with the preceding four weeks, including signs and symptoms of the disease, QoL impairment, efficacy of treatment and overall disease control.3,7Each question is graded from 0 to 4 and the total score is calculated by summing the individual scores. The lowest possible score is 0 (no control) and the highest score possible is 16 (complete control). A score ≥12 indicates well-controlled urticaria, while a score of ≤11 represents poor disease control.18
Dermatology Life Quality Index (DLQI): The DLQI is a questionnaire designed for the assessment of patients over the age of 16 suffering from any dermatological disease. Its aim is to estimate the impact of disease and its treatment on their lives, in the last seven days. It is a useful tool since it is simple to use and interpret, requiring an average of one or two minutes to be completed. The score of each question varies according to the intensity of the affected area in the patients’ life from 3 (very much) to 0 (not relevant). The DLQI is calculated by summing the score of each question resulting in a minimum of 0 and a maximum of 30. Higher scores correspond to a lower QoL.17,19
Chronic Urticaria Quality of Life Questionnaire (CU-Q2oL): The CU-Q2oL is a validated instrument for the assessment of the quality of life impairment in patients with CU, over the last two weeks. It was created by Baiardine et al 200520 and recently its Portuguese version has been created.21 It includes the evaluation of the physical, psycho-social and practical aspects of CU with a total of 23 questions that cover six key CU-specific domains: pruritus, swelling, impact on life activities, sleep problems, limits, and looks (Table 1).
Within each dimension, items are scored on a 5-point scale, from 0 (not at all) to 4 (extremely), according to the patients’ perception of the disturbance caused. These items are then summed to obtain an overall score, which is usually converted to a scale from 0 to 100. In this study, we used final scores and did not convert to percentage, as it would not change the results found. Higher values always correspond to higher QoL impairment, which means worse quality of life.21,22
Hospital Anxiety and Depression Scale (HADS): Used to measure symptoms of anxiety and depression over the previous week. It consists of 14 items, of which seven are for the anxiety subscale, and the other seven for the depression subscale. HADS-Anxiety (HADS-A) evaluates symptoms of generalized anxiety disorder, while HADS Depression (HADS-D) is focused on negative emotions. Each item has four alternatives, ranging between 0 and 3, not all in the same order of impairment. After obtaining the final score for the two subscales a cut-off score of 8 or above for both HADS-A and HADS-D, was used to identify the anxious and depressed patients.23
About the statistical methods, the variables were described accor-ding to their level of measurement. For the qualitative variables, absolute and relative frequencies were presented as for the quantitative variables we used mean and standard deviation, as well as median with quartiles, and amplitude of variation (minimum and maximum) for their description.
The comparison of the main outcome measure, QoL, whether assessed specifically for urticaria through the CU-Q2oL scale or generally related to a dermatologic condition, by the DLQI scale, was performed through the Mann-Whitney U Test whenever the factor comprised only two categories. The Kruskal-Wallis Test was used in case three or more categories were involved. Adjusted multiple comparisons were applied a posteriori when justified. Nonparametric tests were applied due to the lack of normality in quality of life distribution, according to factors involved. The adjustment to a Gaussian distribution was evaluated using the Shapiro-Wilk Test. Due to this fact, whenever there was the need to evaluate correlation between variables, Spearman’s Rank-Order correlation was applied.
After this step, it was possible to select all variables which proved to contribute, somehow, to a distinct level of quality of life in patients with urticaria. Therefore, all those who presented p<0.100 in the previous step were selected for a regression analysis. The stepwise method was applied in order to assess variables according to their burden in prediction and include only those that were predictive of quality of life. The assumptions of the regression models were evaluated, particularly the normality of the residuals, assessed by the Shapiro-Wilk Test, as well as residuals’ independency and absence of autocorrelation, through the Durbin-Watson statistic (CU-Q2oL: DW = 1,559; DLQI: DW = 1,577). The robustness of the models was evaluated by the adjusted R2 and the resulting p-value of the ANOVA result for the model.
Statistical analysis was conducted in the SPSS30, version 25, and evaluated at a significance level of 5%.
RESULTS
Patients and urticaria characterization
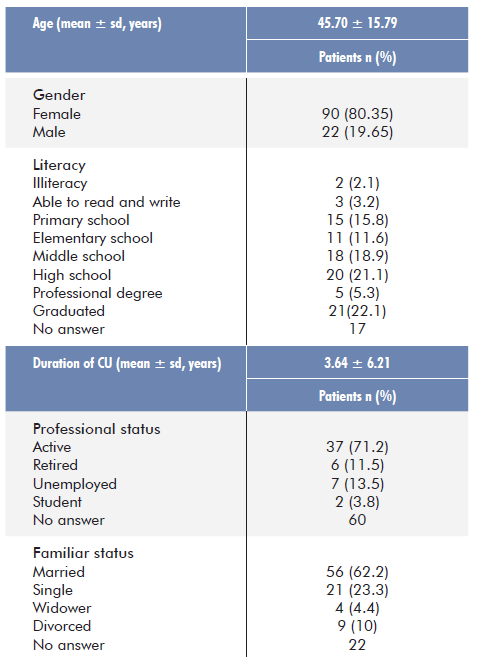
The study enrolled 112 patients with a clinical diagnosis of chronic urticaria followed at the Dermatology Department of the Coimbra University Hospital Centre. Of these, 90 patients were female (80.35%) and 22 male (19.65%), aged between 16 and 82 years (mean 45.70± 15.79) with chronic urticaria for a mean of 3.64±6.21 years. Concerning the sociodemographic characteristics, almost half of the patients had completed high school (48.5%) and more than half were married (62.2%) or professionally active (71.2%) (Table 2).
Regarding the clinical aspects of urticaria, they suffered from chro-nic urticaria, most of them CSU, eventually associated with inducible urticaria. Angioedema was experienced, at any moment of the disea-se evolution, in more than two thirds of patients. The most frequent comorbidity was depression, followed by allergic rhinitis and asthma.
The median value found for UAS7 questionnaires was 9 [3.5; 17] and mean was 11.61 (± 9.98). Both values found allow us to insert our sample in the “Mild Activity” category. Regarding the level of control, measured by UCT, a greater part was “poorly” controlled (85%), and only a minority “well” controlled (15%).
Impact of chronic urticaria on QoL, anxiety and depression
As for the QoL assessment, the mean value found in the sample for CU-Q2oL was 49.13 ± 19.28 in a maximum possible of 115, while DLQI varied from 0 to 25 scoring a mean value of 7.23 ±7.67. Therefore, the sample was included in the category “Moderate effect on patient’s life”. For the CU-Q2oL scale, it was evident that pruritus and wheals are the parameters reflecting the biggest impact in patients’ wellness. The following higher scores found are awakenings during night, difficulties in keeping concentration and decreased quality of sleep. Re-garding the DLQI, analysis revealed skin conditions (itch and pain) and embarrassment caused by these, to be the most affected symptoms. Both QoL assessment scales were shown to have a “high positive correlation” according to the coefficient, (r*=0.883, p<0.001).
The results of the questionnaires evaluating the quality of life was statistically different between genders. Regarding Cu-Q2oL, Mann-Whitney U Test showed significant difference between male and female (p=0.007), with higher values seen in females, representing decreased quality of life.
Concerning the level of education and analyzing CU-Q2oL data, a statistically significant relationship was found between the degree of patients’ instruction and their quality of life. The group with highest level of education (graduated degree) has the lowest mean and median value of CU-Q2oL, which represents a worse QoL. Statistically significant differences were found between the "Graduated" group and the remaining literacy levels, except for two. A similar relation between literacy and QoL can be seen with the DLQI scale.
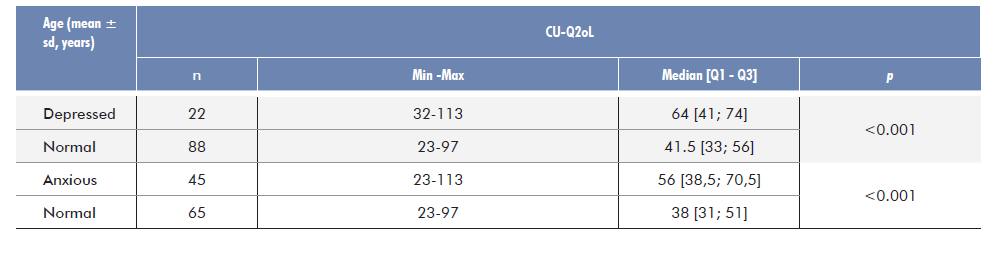
Among the 110 patients who filled the HADS questionnaire, 22 patients were considered “depressed” (18.8%) and 88 mentally healthy (81.2%), whereas in terms of anxiety, 45 were categorized as “anxious” (40.9%).
Correlation between QoL, anxiety and depression and chronic urticaria activity or control
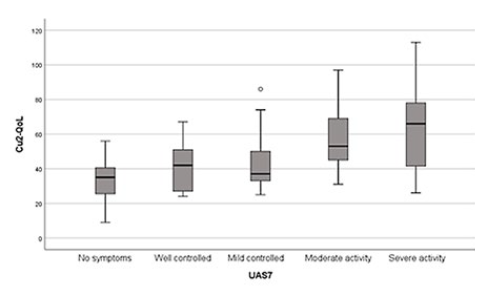
The clinical activity of urticaria is highly correlated with QoL measured both by CU-Q2oL and DLQI, which were found to be statistically associated with UAS7 (p<0.001 for CU-Q2oL and p= 0.002 for DLQI). The range values of quality of life scores are significantly higher as the disease increases its severity (Fig. 1).
A significant difference was observed with any of the two parameters measured by the UAS7, as higher number of papules or a more intense pruritus both contribute to deteriorate one’s psychological and physical state, lowering QoL.
Regarding UCT score, the Mann-Whitney U Test confirmed the existence of a statistically significant difference between QoL in “well” and “poorly” controlled patients for both Cu-Q2oL and DLQI. Patients with good control of the disease reveal a much better quality of life than those with poor control (mean CU-Q2oL value 31 vs 48). In the four parameters evaluated by the UCT, statistically significant differences were found in quality of life among the various categories, particularly between “slight” and “severe”.
The quality of life of this sample revealed to be significantly related to anxiety and depression status, measured by the HADS scale (Table 3).
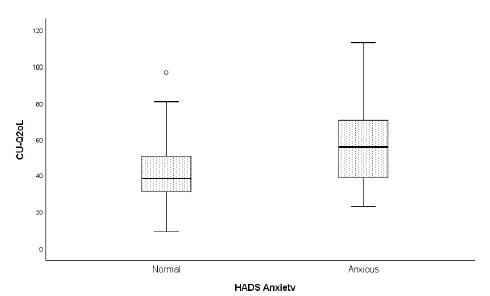
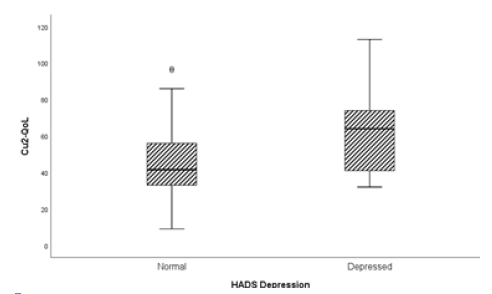
Concerning anxiety and depression (HADS-A/D), data evidenced the contribution of anxiety to lower patients’ quality of life (Fig. 2) and showed a statistically significant difference between CU-Q2oL scores depending if the patient is classified as depressed or not, with depression contributing to a lower quality of life (Fig. 3). These patients evidence worse levels of QoL than anxious patients.
Factors that predict influence on QoLCU-Q2oL=37.616+7.315×QoLP+6.113×InsufTt-21.120×UCT +7.845×PSI-13.909×Gender DLQI=-5.925+3.843×PSI+2.417×UAS7icth
To estimate the Predictors of Quality of Life, the variables that proved to have a significant (p<0.05) or approximate (p<0.10) influence on patients’ QoL were selected. The variables assumed as potential predictors of quality of life in urticaria were: gender, higher grade of education, UAS7, UCT, HADS-A, HADS-D, intensity of pruritus in the last week, number of papules in the last week, treatment made for urticaria, self-perception of quality of life, intensity of the physical symptoms, and insufficiency of treatment in the control of symptoms.
As for CU-Q2oL, the regression analysis identified as predictors of better quality of life the good control of CU symptoms, measured by UCT, and being male. The insufficiency of treatment to control the symptoms, the intensity of physical symptoms and the self-perception of a negative impact in QoL contributed to decrease quality of life, increasing CU-Q2oL final score. These factors explain 73% of the variability found in the quality of life, (R2adj=0.730; p=0.029) thus, the quality of life of the patient with urticaria, measured through the Cu-Q2oL scale, can be reliably predicted by the following equation:
Each parameter, QoLp (quality of life self-perception), InsufTt (in-sufficiency of treatment) and PSI (physical symptoms intensity), correspond to the questions included in UCT. This way, each one varies between 0 (nothing) and 4 (very much). UCT represents the result of the UCT scale, taking the value 0 for a good control and 1 for poor control. Gender admits the value 1 for female and 2 for male.
For the DLQI scale, the regression analysis identified only two fac-tors (intensity of physical symptoms and pruritus) able to explain 52.8% of the variability found in quality of life, (R2aj = 0.528; p<0.001) which can be reliably predicted by the following equation:
DISCUSSION AND CONCLUSION
The aim of this study was to estimate the impact of CU in Portuguese patients’ quality of life, using scales that access the patients’ perception of their quality of life rather than an external measurement that could underestimate the real impact in the life of the patient. As recommended by both literature and guidelines, PROs (Patient-reported outcomes) were used to assess patients’ condition, facilitate disease monitoring and management and also to improve patient-physician communication.
With this study, it was enhanced not only the applicability of these tools in Portuguese patients with chronic urticaria, but also its convenience, simplicity and easiness to fulfill.
Regarding results found, in our sample, there was an average of 80% females, which is according to previous studies in chronic urticaria. In international studies, CSU has constantly showed higher prevalence than other types of chronic urticaria and the present study corroborated this data, with an incidence of 86.6% reported for this type.22,24-27
The mean age of our patients was 45.7 years, similar to values reported, and its long-standing duration (average of 3.64 years) is agreeing with most studies.22,24-27The percentage of patients with angioedema (2/3) is higher than values found in other studies (a mean of 1/3 of UC has been reported), reflecting the clinical severity of patients studied in our consultation and included in the study.28,29
There is lack of information from previous studies regarding the association of urticaria with sociodemographic factors, such as level of instruction and professional status. In our study, there was a considerably high percentage of patients professionally active and those who have completed high school.30 More than half of the patients were married.
Comorbidities are present in the majority of patients and depression, allergic rhinitis and asthma are the most prevalent. The strong association between dermatological diseases, highlighting urticaria, and psychological/psychiatric problems had already been proven.11,12,15,31
The parameters that showed the greatest impact on quality of life were skin condition (pruritus and wheals); sleep problems (night awakening, difficulties in keeping concentration, impact in sleep), and embarrassment due to urticaria symptoms. The symptoms of CU have, consequently, been shown to be the most important factor in patients’ perception of QoL, emphasizing the importance of clinical control of urticaria as the first approach to improve patients' quality of life. These results are consistent with studies performed in Spanish patients, in which “sleep” was markedly impaired.26 Like our results, studies in German, Polish and Brazilian population present “itch” and “embarrassment” as the most affected areas, with the Brazilians reporting, in addition items such as “tiredness” and “concentration”.22,24,27A Turkish study also showed “pruritus” and “sleep” to be the most affected scales, whereas “swelling” was the least affected.25
“Eating behavior/ Impact on life activities” and “Limits in choosing food” scored a low count in the present study, contributing slightly to quality of life impairment, which is consistent with the results found.24,27Our information with no need for diets and food eviction may also explain this result.
Regarding the following parameters, sleep problems and low concentration can be deeply related with each other, since urticaria can lead to a difficulty in falling asleep due to the severity of symptoms and awakening several times during night decreases the quality of sleep therefore resulting in sleep deprivation during the day and difficulties maintaining concentration in routine activities and work. As for the embarrassment caused by urticaria symptoms, it reflects the importance of esthetical aspects in a patient’s daily life.
Females feel a greater impact of the disease in comparison to males, which is in agreement with the literature.22,24-27,29,32This can be explained by their high susceptibility in having certain domains affected (embarrassment due to urticaria symptoms; problems in using cosmetics and sleep problems). In addition, female present greater emotional fragility, being the gender with higher prevalen-ce of depression, anxiety and nervousness.9,11,33Also, female have proved to be more affected than male in specific parameters, such as “sleep problems”.25
Higher scores of UAS7, indicating stronger disease activity, were associated with lower levels of QoL as demonstrated in other studies.10,12CU-Q2oL allowed us to find statistically different levels of quality of life between four groups of urticaria severity, whereas the DLQI only detected differences between two groups (mild/severe activity), showing the higher sensibility of CU-Q2oL to detect minor differences in QoL.
UCT results highlighted the importance of an effective (pharmacological or non-pharmacological) control of the clinical symptoms for an increased quality of life.34Pruritus revealed to be the most debilitating symptom.27 Also, one’s perception of his physical symptoms as well as of the insufficiency of treatment in controlling the disease are areas to be taken into consideration for an integrated approach. Patients’ own view of their condition is being increasingly valued as there are no reliable biomarkers to identify and measure disease activity and control.5,35,36
Additionally, we achieved a reliable equation able to explain 73% of the variability found in the quality of life measured by CU-Q2oL, and another explaining 52.8% of the variability found in DLQI.
With respect to psychiatric pathology, CU-Q2oL demonstrated a significantly lower quality of life in pathological groups, i.e., anxious and depressed patients. Depression appears to have a major impact on our study, both because it is present as the most frequent comorbidity and deteriorates patients' quality of life more than anxiety. An anxious or depressed patient will tend to overestimate the physical symptoms of urticaria, leading to a more difficult control. A therapeutic approach should, therefore, encompass patients’ psychopathological domains. An attempt to understand the underlying cause of psychological imbalance, a detailed approach to patient's personal life, or even the physician's request of an assessment by the specialist of this area could be indicated to solve these situations.
Although it remains unclear if psychological imbalances are the cause or the result of dermatological problems, the cross-sectional design of this study does not allow conclusion regarding causal links between the high scores on these parameters and CU. We attempted to establish a temporal relation between the diagnosis of urticaria and depression, but no reliable data was collected. Several studies suggest that multifactorial origin of CU includes psychological and social factors which can lead to depression and decrease patients’ QoL. Other hypothesis suggest that it is the mental debility that increases the probability of the onset of urticaria. Some investigators link vasodilation-induced by emotional status to urticaria lesions in the skin and a previous study presented swelling as the cause of anxiety and mood disturbances in patients.9,11,15,27
Patients with higher education presented worse values of quality of life, which goes against data from the literature.22,32This fact was verified when comparing the category of graduated patients with the others. This may mean that patients with higher literacy are more knowledgeable and tend to work in jobs requiring greater intellectual effort. They have also more integrated perception of their condition, being more critical with their illness and treatment.
As for the limitations of our study, we highlight the difference between periods of time measured by the scales, since for example, CU-Q2oL is used for the last two weeks, whereas DLQI relates only to the last week. Another constraint in this investigation was the different number of patients that filled each of the scales. These limitations were not an impediment, since a strong and positive correlation was found between both scales, which indicates compatibility within the measures used. Another limitation is that our sample was comprised by patients followed in the Dermatological Department, at Coimbra University Hospital Centre. Therefore, a selection effect in every variable cannot be excluded as the sample was not constituted by patients from different hospitals or clinics in the country.