INTRODUCTION
Cutaneous lesions of the vulva are common and may be associated with considerable physical and psychological distress. There is a tendency to attribute an infectious or venereal origin to any vulvar lesion, which contributes to significant anxiety.1-3However, the vulva may be site of noninfectious lesions including widespread dermatoses which may have distinct or atypical presentations in this anatomic area (like psoriasis or lichen planus), other clinical entities that occur only in the genital area (such as Zoon vulvitis), as well as benign and malignant neoplasms.4-6Therefore, its identification is challenging, and a cutaneous biopsy may be required for the correct diagnosis.
To date, only a few studies have reviewed the relative frequencies of biopsy-proven vulvar diseases.7,8
The objective of our study was to assess all cases of noninfectious vulvar diseases that required a biopsy over a 10-year period in a tertiary hospital, including its relative frequencies, treatment, and clinical evolution.
MATERIAL AND METHODS
A retrospective study of all noninfectious vulvar diseases with histological confirmation diagnosed in Centro Hospitalar e Universitário Lisboa Norte, in Lisbon, Portugal, during a 10-year period was conducted. The histopathology archive was searched for all the vulvar biopsies received between January 1, 2008 and December 31, 2017 (a total of 403 biopsies). The clinical files were then reviewed and all cases of noninfectious diseases with histological diagnosis and clinical information were included. The exclusion criteria were infectious diseases (22), noninfectious diseases without clinical information (11), nonspecific features on histopathology (30) and repeated biopsies of the same patient addressing the same pathological process (17).
The diseases were classified in two main types (inflammatory and neoplastic), considering the pathological findings. Treatment and cli-nical evolution of the most frequent conditions was examined. Results were analyzed by SPSS Statistics 25® software. Informed consent was obtained from the patients.
RESULTS
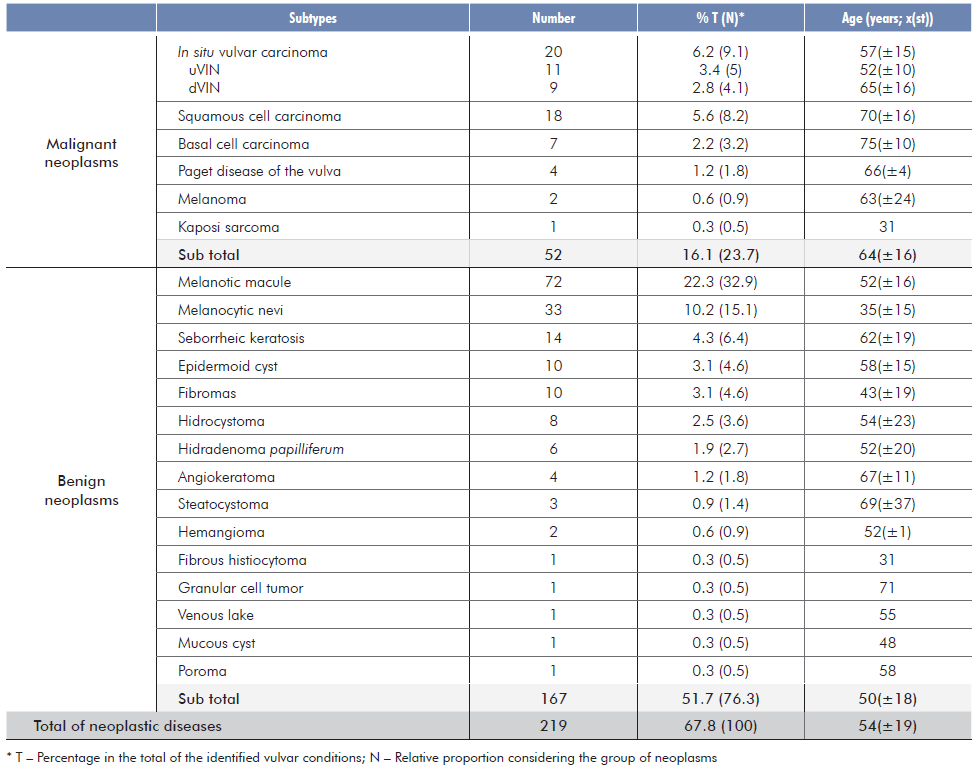
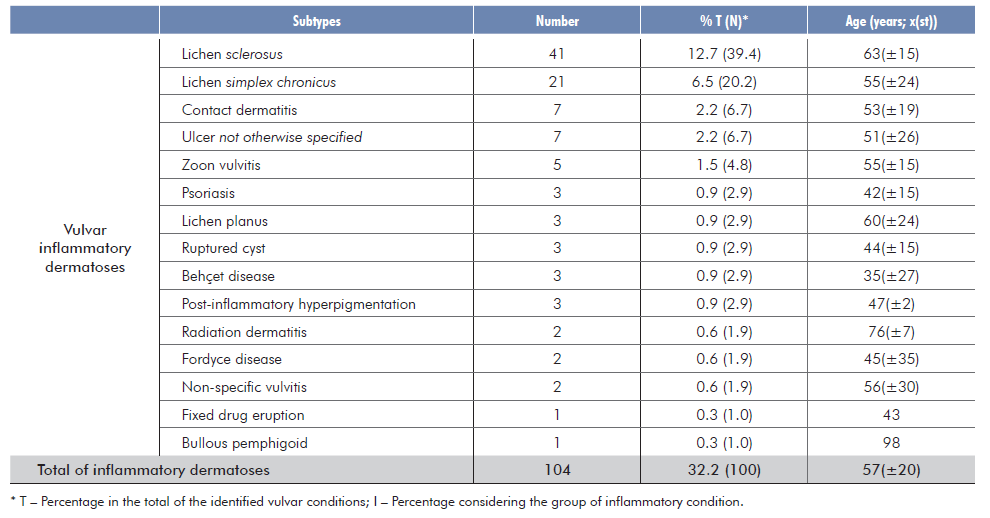
A total of 323 biopsies from 317 patients were included. The age of the patients ranged from 11 to 98 years, with a mean of 54.2 years σ=19). A total of 36 vulvar conditions was identified (21 neoplastic and 15 inflammatory). Neoplasms were present in approximately 68% of the patients (219/323) and inflammatory dermatoses in 32% (104/323) [Tables 1 and 2]. Overall, the most common identified condition was melanotic macule (22.3%, 72/323), followed by lichen sclerosus (LS) (12.7%, 41/323).
Prevalence of neoplasms
Neoplasms were mainly benign tumors (76.3%, 163/219), particularly melanotic macules (44.2%, 72/163) and melanocytic nevi (20.2%, 33/163). Amongst these, the most common were intradermal nevi (42.4%, 14/33), followed by dysplastic nevi (21.2%, 7/33) and compound nevi (8.2%, 6/33). The remaining patients had other benign tumors such as seborrheic keratosis (8.6%, 14/163), epidermoid cysts, fibromas (6.1%, 10/163 each), hidrocystomas (4.9%, 8/163) and other even less frequent lesions (Table 1).
Amongst the malignant tumors (23.7%, 52/219), the most frequent was vulvar intraepithelial neoplasia (VIN), corresponding to 38.5% (38.5%, 20/52). Of these, 11 cases were associated with Human Papillomavirus (HPV) infection and thus classified as usual-type VIN (uVIN), while the other 9 cases corresponded to differentiated vulvar intraepithelial neoplasia (dVIN) and were linked to chronic inflammatory conditions. The second most common malignant neoplasm was invasive squamous cell carcinoma (SCC) (34.6%, 18/52). There were seven cases of basal cell carcinoma (BCC) (13.5%, 7/52), three cases of Paget’s disease of the vulva (PDV) (5.8%, 3/52) and two cases of malignant melanoma (3.8%, 2/52). Kaposi sarcoma was diagnosed in only one patient (1.9%, 1/52).
Prevalence of inflammatory dermatoses
The most frequent inflammatory condition was LS (39.4%, 41/104), followed by lichen simplex chronicus (20.2%, 21/104), eczematous dermatitis (Fig. 2b) and ulcer not otherwise specified (NOS) (6.7%, 7/104 each) and Zoon vulvitis (4.8%, 5/104). Less frequent derma-toses included psoriasis, lichen planus, ruptured cyst, Behçet disease, post-inflammatory hyperpigmentation (2.9%, 3/104 each), Fordyce disease and radiodermatitis (1.9%, 2/104). There was just one case of fixed drug eruption and one case of bullous pemphigoid.
Treatment and clinical outcome
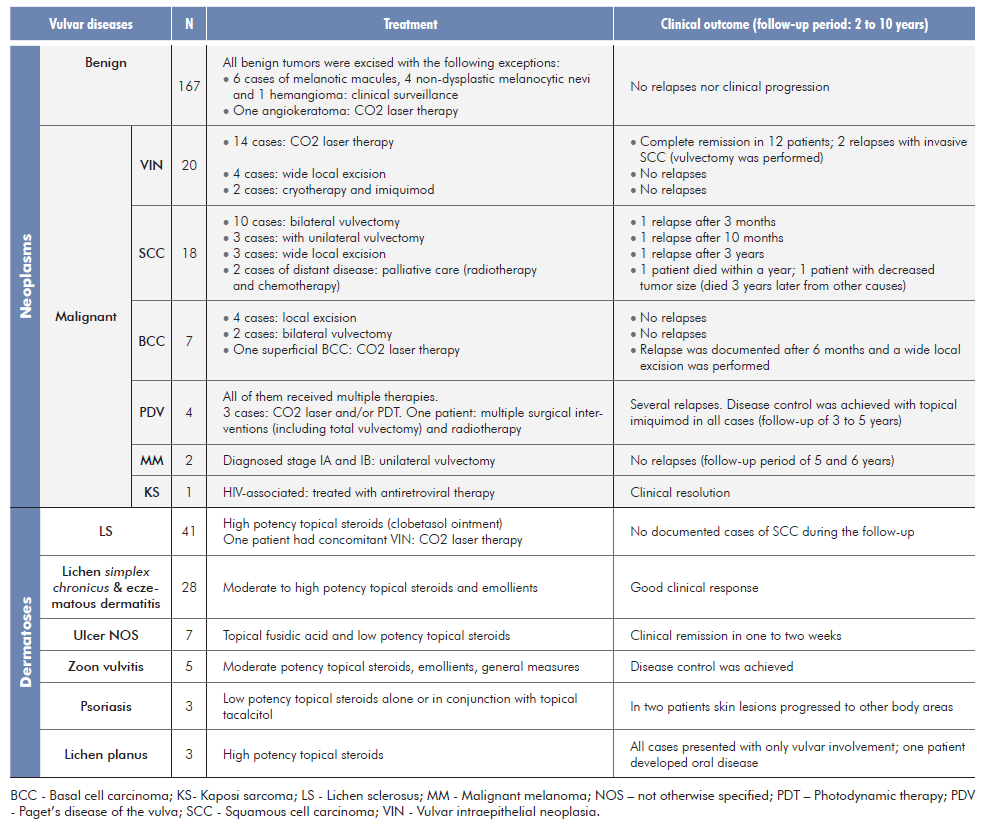
Treatment and clinical outcome for the different conditions is elicited on Table 3.
DISCUSSION
The aim of this retrospective study was to evaluate which vulvar diseases required a biopsy in our Dermatology department. As expected, most of these cases are of neoplastic etiology. Amongst neoplasms, benign tumors (76.3%, 163/219) were by far the most frequent. Vulvar malignant neoplasms are relatively uncommon (0.4% of all cancers)9 with an overall incidence of 2-7 cases per 100 000 women.10 Their incidence increases with age10 with a median age at diagnosis between 60-70 years.9,11In our study median age at diagnosis was 64 years.
Vulvar neoplasms
Most frequently biopsied benign vulvar neoplasms
The most frequently biopsied benign tumors were melanotic macules (44.2%, 72/163) and melanocytic nevi (20.2%, 33/163). Benign tumors were mainly biopsied to rule out malignancy.
Melanotic macules (Fig. 1), the most frequently encountered, are common benign pigmented lesions of small size located on labia minora that follow a benign clinical course.12,13In our study, melanotic macules represented almost 60% of the pigmented vulvar lesions (versus 68% in other studies13). They are more com-monly found among perimenopausal women (median age of 43 years).12,13Our patients were older, with a median age of 52 years.
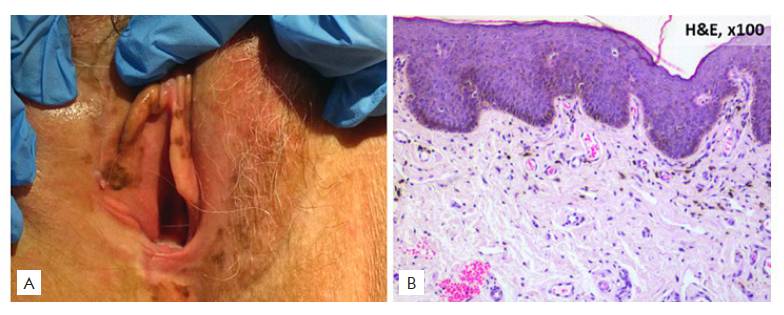
Figure 1 Melanotic macule: (A) Clinical picture: well-defined, brown-colored macule; (B) Histopathological picture: basal lawyer hyperpigmentation, the number of melanocytes is not increased.
Melanotic macules, particularly with a bigger size, can be clinically indistinguishable from melanoma, thus requiring a skin biopsy.
According to the literature, melanocytic nevi account for 23% of all pigmented vulvar lesions,13which is consistent with our results (25% of the identified pigmented lesions). In contrast to melanotic macules, vulvar nevi are generally encountered in premenopausal women, with a median age of 30 to 34 years (in our study median age was 35 years).12,13Common nevi are well demarcated, with regular borders, uniform pigmentation, and typically small size (<1 cm in diameter).12,13The main reason for biopsy is to differentiate from melanoma.
Seborrheic keratoses, the third most common biopsied benign neoplasm, become more prevalent in increasing age (we found a median age at diagnosis of 62 years).11 Their clinical appearance may resemble melanoma and thus require a biopsy.
Most frequently biopsied malignant vulvar neoplasms
Taken together, VIN and invasive SCC (Fig. 2), are by far the most common vulvar malignancy (83% to 90%, in most series).9-11,14,15SCC arises from two major types of VIN, which correspond to two distinct oncogenic pathways: uVIN is HPV-dependent (and is more common in younger women) while dVIN develops independently of HPV infection14,16and accounts for 20% of all cases of SCC occurring in post-menopausal women within a background of a chronic inflammatory dermatosis, most frequently LS.9,10,14,15First-line treatment for SCC is excision with or without sentinel lymph node dissection.9,10,12Treatment for disseminated disease is usually palliative.9 On the other hand, VIN may be treated conservatively with topical imiquimod, photodynamic therapy and laser therapy.9,10,12,14VIN usually manifests as an asymptomatic and solitary pink or red scaly plaque with irregular edges.4,10,14Recurrence rates for vulvar invasive SCC are high (12% to 39%) and regular surveillance is recommended.9 In our study, VIN and SCC corresponded to 73.1% (38/52) of all malignant neoplasms which is slightly inferior to previously reported data.17 For SCC, excluding he two patients with distant disease who received palliative care, there was a recurrence rate of 18.8% (3/16), consistent with the literature.17 The preferred treatment for VIN was CO2 laser therapy with a success rate of 78.6%, identical to other studies.17
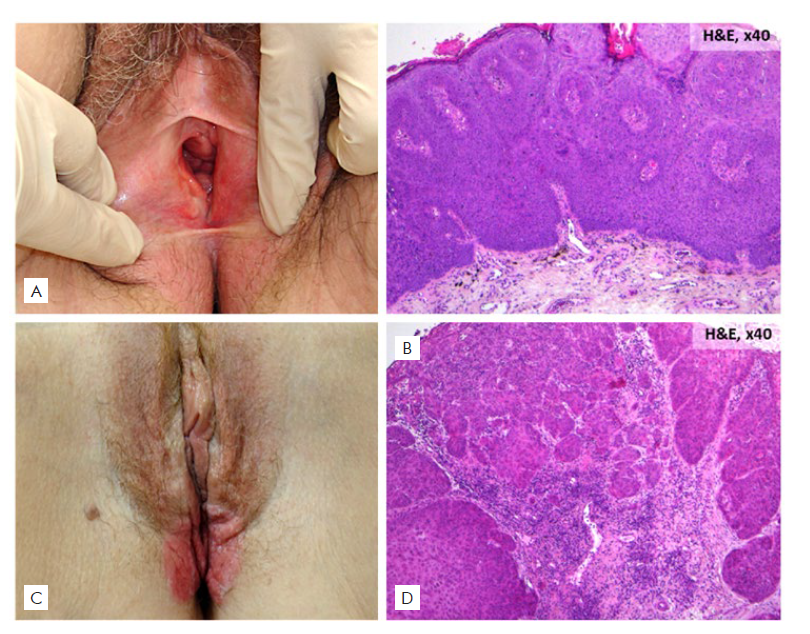
Figure 2 (A, B) Vulvar intraepithelial neoplasia: (A) Clinical picture: pink plaque with irregular edges; (B) Histopathological picture: atypia within the epidermis (H&E 40x). (C, D) Squamous cell carcinoma: (C) Clinical picture: erythematous verrucous indurated growth with ulceration; (D) Histopathological picture: nests of squamous epithelial cells that arise from the epidermis and extend into the dermis.
In our series, BCC was the third most commonly biopsied ma-lignant neoplasm, corresponding to 13.5% (7/52) which differs from previously reported prevalence rates of only 1%-4%.10 Average age at diagnosis was 75 years, consistent with other studies (average age of 68-73 years).10,12Vulvar BCCs usually present as single lesions in labia minora and majora. Metastatic potential is low but local recurrence rates are high (up to 20%).10,12
Paget’s disease of the vulva (Fig. 3) accounted for 1.8% of the vulvar malignancies, with an average age of 66 years, consistent with the literature.9,11,12Regardless of the adopted therapy, all four cases of PDV had a chronic course. Partial disease remission was possible under local therapy with imiquimod. High recurrence rates are typical, and overtreatment should be avoided.18
In our series melanoma accounted for only 0.9% (2/52) of malignant neoplasms which is inferior to the 3%-10% reported rates.9,11It is more common is postmenopausal women.9-12
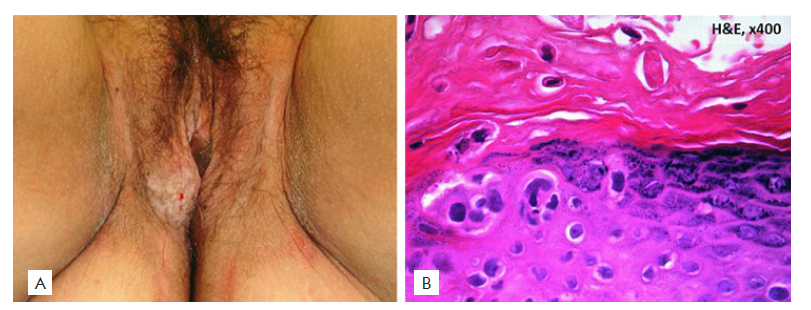
Figure 3 Paget’s disease of the vulva: (A) Clinical picture: red plaque with typical ‘cake-icing' scaling and small superficial erosions; (B) Histopathological picture: pagetoid infiltration of the epidermis by pleomorphic large cells with clear cytoplasm and a prominent nucleolus (H&E 100x).
Inflammatory dermatoses
Vulvar dermatoses were mainly biopsied to rule out malignancy, but some lesions of long duration with a doubtful clinical diagnosis were also biopsied for diagnostic purposes.
As reported in the literature, vulvar LS was the most commonly biopsied dermatosis (39.4%, 41/104) and the second most common of all the identified conditions (12.7%, 41/323).1,3,7LS is a chronic inflammatory skin disease with a predilection for the external genitalia. It is more prevalent in women, especially after menopause, although it can develop at any age. Typical presentation consists of a well-defined white sclerotic plaque with an atrophic wrinkled surface (Fig. 4). Associated symptoms include pruritus, dyspareunia, burning, bleeding, and dysuria. If left untreated, it may lead to scarring and permanent distortion of the vulvar structure.6,19-21Furthermore, untreated lesions carry a 2%-6% risk of vulvar SCC.6,20,21Ultra-potent topical steroids are the mainstay of treatment and even asymptomatic women should be treated. Long-term maintenance treatment arrests disease progression and reduces the likelihood to develop SCC.6,19-21In our institution LS cases were biopsied mainly to rule out associated malignancy.
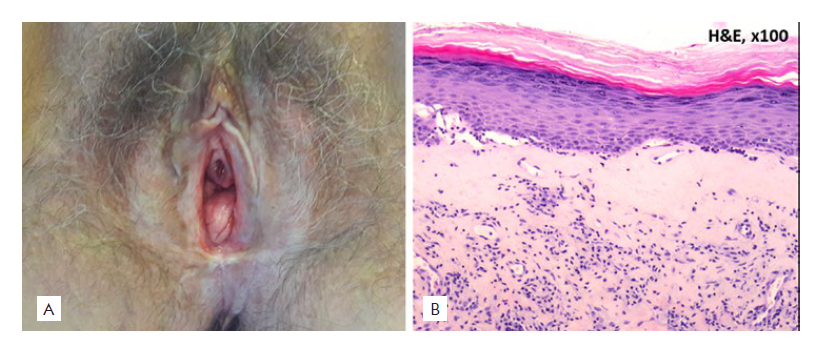
Figure 4 Lichen sclerosus: (A) Clinical picture: white sclerotic plaque with an atrophic surface around the vulvar area; (B) Histopathological picture: hyperkeratosis and epidermal atrophy, a lymphocytic dermal infiltrate and interface changes are also observed.
Lichen simplex chronicus is one of the most common causes of vulvar itching.5,22,23When considering vulvar dermatoses, previous studies reported a similar prevalence of 20%.7 It is caused by persistent itching and scratching which leads to epidermal thickening and lichenifica-tion.6,22,23Vulvar contact dermatitis, on the other hand, is generally caused by repeated contact with an irritant (irritant contact dermatitis) and the lesions are typically limited to the site of contact (Fig. 5A).22 None of these conditions requires a biopsy except when the diagnosis is not straightforward. A bland skin care regimen and moderate-potent topical steroids are advised.6,22
The three patients with ulcers NOS had only genital lesions and associated infection was excluded. These lesions present as well-circu-mscribed round to oval ulcers usually measuring less than 3 cm (Fig. 5B), affecting mainly the vulvar mucous membrane.5,11,15The reason to biopsy was to exclude malignancy.
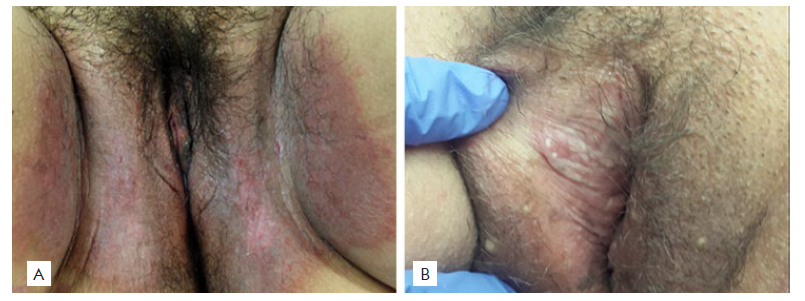
Figure 5 (A) Irritant contact dermatitis, clinical picture: well-demarcated red patch confined to the site of contact with the irritant; (B) Ulcer NOS, clinical picture: well-circumscribed and small round ulcer.
For Zoon vulvitis we found an overall prevalence of 1.5%. Zoon vulvitis is a relatively rare pathology.2,15,23It is a benign and chronic condition with unknown etiology that tends to affect postmenopausal women (mean age at diagnosis was 55 years). It may be asymptomatic, but it is frequently associated with itching, burning, pain and bleeding. Lesions present as well-defined erythematous or brownish-orange plaques with bilateral and symmetrical distribution (Fig. 6) and can be confused with VIN.24 Biopsy is essential for diagnosis.23
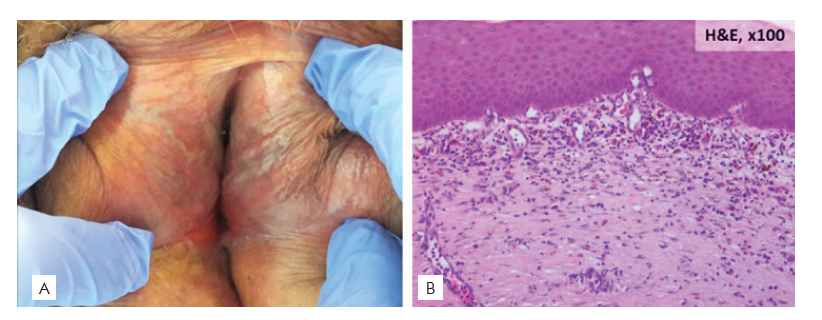
Figure 6 Zoon vulvitis: (A) Clinical picture: bilateral and symmetrical distributed well-defined rusty red plaques; (B): Histopathological picture: dense band-like infiltrate of plasma cells in the upper dermis.
Study limitations
Adding to the fact that this was a retrospective study, the other main limitation of our work relies on the fact that only conditions with a histological confirmation were enrolled. As such, other clinically diag-nosed noninfectious vulvar diseases were excluded.
CONCLUSION
In this study, neoplastic conditions were the most frequently biopsied lesions affecting the vulvar area, with VIN and SCC as the most documented malignant tumors. Concerning the inflammatory dermatoses, LS was by far the most prevalent. These results highlight the main reason to biopsy, which was to exclude malignant processes. Diagnosis of vulvar noninfectious inflammatory lesions is usually made on clinical criteria.
From a practical approach, this study shows that when facing a vulvar pigmented lesion that warrants a biopsy, it will most likely be a benign lesion. On the other hand, if we are facing a red and eroded plaque, there is a considerable likelihood of VIN. Vulvar ulcerations that develop in a background of inflammation or are refractory to treatment and thus require a biopsy carry a high probability of SCC. This study also illustrates the myriad of conditions that may affect the vulva. These conditions cause a considerable burden on the patients and some of them have serious health consequences if left untreated. Correct diagnosis and management are thus essential.
Presentations /Apresentações
Part of the content for this article was presented as an oral communication in the XIX Congresso Nacional de Dermatologia e Venereologia, Sheraton Porto Hotel, Porto, December 6th 8th 2019