Case
A 65-year-old female patient with a personal history of advanced gastric adenocarcinoma and peritoneal carcinomatosis, who was undergoing combination chemotherapy (FOLFOX) with an immunotherapy drug (nivolumab), was admitted to the outpatient clinic with a one-month history of progressive jaundice, choluria and acholia. The patient denied any other symptoms, including abdominal pain, fever or constipation.
Laboratory results were remarkable for elevated total and conjugated bilirubin of 7.19 mg/dL and 5.46 mg/dL, respectively (N < 1.2 mg/dL and 0.3 mg/dL), elevated amylase of 355 U/L (N 13-53 U/L), C-reactive protein of 12.45 mg/dL (N <0.5 mg/dL), CA 19.9 of 56.1 U/mL (N <37U/mL). She was also diagnosed with new-onset uncontrolled diabetes mellitus.
The CT scan revealed diffuse dilatation of intra-hepatic biliary ducts (Figure 1A). Diffuse enlargement of the pancreas and peri-pancreatic fat stranding were also noted, suggesting inflammatory pattern (Figure 1B-C). Alongside dilatation of intra-hepatic bile ducts, the MRI scan showed a long stricture of the common bile duct (Figure 2A, 2C). Increased wall thickness of the common bile duct was also observed (Figure 2A).
Diffuse restricted diffusion of the pancreatic parenchyma (Figure 2E-F) was identified and slight loss of lobulation (Figure 2D), indicating possible autoimmune pancreatitis.
Restricted diffusion of the intrahepatic bile ducts wall (Figure 2B) and increased wall thickness of the gallbladder (Figure 2A) was also noted, suggesting concomitant cholangitis.
These findings were consistent with the diagnosis of immune-induced pancreatitis and cholangiopathy in a patient who completed treatment with nivolumab.
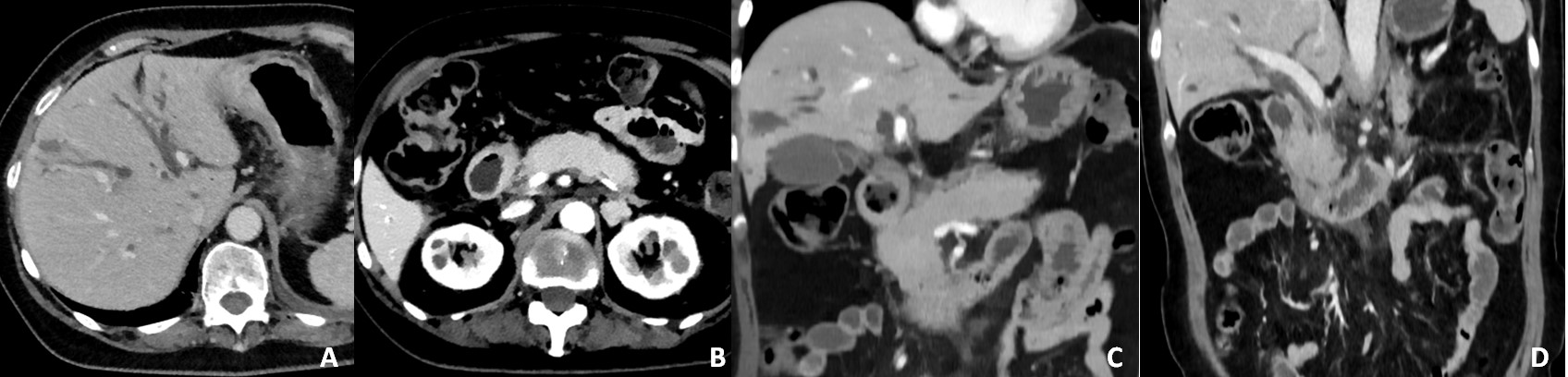
Figure 1: Abdominal CT (A, B) axial portal-phase and (C, D) coronal portal-phase, demonstrating diffuse intra-hepatic bile duct dilatation (A) and diffuse enlargement of the pancreas (B, C) and peri-pancreatic fat stranding (D).
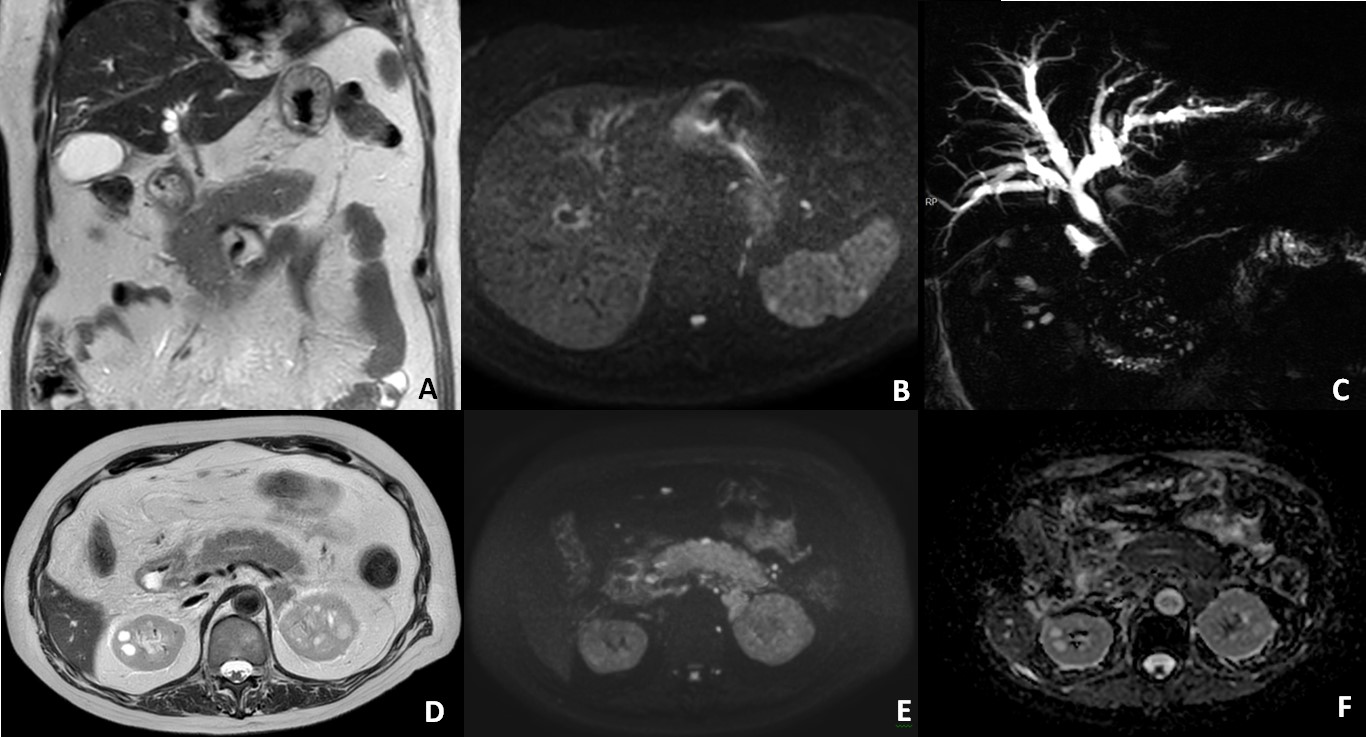
Figure 2: Cholangiopancreatography MRI (A) coronal T2W (C) radial CPRM; (D) axial T2W (B, E) DWI (F) ADC map; showing increased thickness of the pancreatic parenchyma (A, B) and diffuse restricted diffusion of the parenchyma (E, F). Diffuse intra-hepatic bile duct dilatation and dilatation of the distal part of the extra-hepatic bile duct, with increased thickness of the bile duct wall (A, C). A long regular stenosis of the distal part of the extra-hepatic bile duct is also present (C). Restricted diffusion of the intra-hepatic bile ducts is shown (B).
Discussion
Nivolumab is an anti-programmed death receptor (PD-1) monoclonal antibody that belongs to the group of immune checkpoint inhibitors (ICIs).1 This drug is an effective treatment for a variety of tumors, including gastric cancer.
ICIs have been shown to block the programmed cell death protein (PD-1) receptor, thereby releasing the inhibitory effect on T-cell effector mechanisms. This, in turn, results in the inhibition of cancer cells from suppressing the immune system and the augmentation of antitumor immune responses.1 However, it should be noted that these treatments have the potential to induce immune-related adverse events, such as ICI-induced pancreatitis and cholangitis, although these are uncommon.2
ICI-induced pancreatitis and cholangitis can be either asymptomatic or symptomatic. The symptoms that are most commonly observed in such cases include epigastric or diffuse abdominal pain, fever and nausea. Jaundice is typically observed in cases of cholangitis.2
Increased levels of amylase and lipase are significant indicators of suspected ICI-induced pancreatitis.3 However, elevated pancreatic enzymes in isolation, without additional diagnostic criteria, are insufficient for an accurate diagnosis.4 For ICI-induced cholangitis, elevated alkaline phosphatase and gamma-glutamyl transpeptidase levels are hallmark indicators.2
Imaging findings of ICI-induced pancreatitis typically exhibit two patterns: one resembling acute interstitial pancreatitis and another resembling autoimmune pancreatitis.4 On CT, acute interstitial pancreatitis-like features include marked pancreatic swelling, reduced contrast uptake and peri-pancreatic fat stranding. Autoimmune pancreatitis-like features on CT include focal or diffuse enlargement of the pancreas with loss of lobulation.3 On MRI, besides the morphological features similar to the ones described for CT, the most commonly reported findings for auto-immune pancreatitis include restricted diffusion on diffusion-weighted imaging, capsule like rim, and late-phase contrast enhancement with gadolinium. A long and irregular narrowing of the main pancreatic duct may also be observed. MRI is useful when differentiating ductal stenoses from pancreatic ductal adenocarcinoma and inflammatory conditions, with the “penetrating duct sign” seen in MRCP strongly suggesting benignity.5,6Additionally, MRI can show multi-organ involvement in cases of autoimmune pancreatitis, which are common in the kidneys in the context of IgG4-related disease.7,8Increased 18F-fluorodeoxyglucose uptake on PET/CT (18F-FDG-PET/CT) is also a common finding in ICI-induced pancreatitis.4
In the present case, diffuse pancreatic enlargement was observed with peri-pancreatic fat stranding (Figure 1B-D; Figure 2D), along with restricted diffusion, suggesting inflammatory pattern. Additionally, diffuse intrahepatic and focal extrahepatic bile duct dilatation were observed (Figure 2C), with diffuse thickening of the extrahepatic bile duct wall and of the gallbladder wall (Figure 2A). These findings suggest concomitant ICI-induced cholangitis.
ICI-induced cholangitis can be classified as large-duct, small-duct, or mixed type, based on the segments primarily affected.4 Imaging findings of ICI-induced cholangitis may include multiple intrahepatic strictures, resembling primary sclerosing cholangitis, or hypertrophy of the extrahepatic bile duct wall, resembling IgG4-related sclerosing cholangitis.3
Steroids and combined immunosuppressive therapies are generally recommended for the treatment of ICI adverse events.4 In the present case, treatment with nivolumab was already completed at the time of symptom onset and a trial of steroids was initiated with symptom relief.















