INTRODUCTION
The human organism requires small quantities of vitamins and minerals for growth, brain development, immune function and several other important functions (1). Since the body cannot synthesize these nutrients, they must be obtained through the diet or other sources. An inadequate intake and/or absorption of micronutrients will entail serious consequences, including learning difficulties, impaired work capacity, illness and even death (2).
Food fortification (FF) has long been employed as a strategy to address micronutrient deficiency concerns within populations (3). The concept of fortifying staple foods with micronutrients occurred at the beginning of the 20th century to tackle the deficiencies of vitamins and/or minerals in Europe and North America (4). The first fortified food developed to combat iodine deficiency and thus prevent goitre was iodized salt, which was introduced in Switzerland and Michigan in the 1920s (5). From the early 1940s onwards, the fortification of cereal products with iron (Fe), vitamins B1, B3 and B2 became common practice to reduce the incidence of anaemia, beriberi, pellagra and riboflavin deficiency, respectively. Since 1996, vitamin B9 has also been added to cereal products in the United States to prevent the development of neural tube defects during embryonic development. Canada and some Latin American countries adopted the same strategy (5 - 8).
FF can assume many forms: mass fortification (MF), target fortification (TF) and market-driven fortification (MDF) (2, 6, 8). The latter occurs when, voluntarily, food manufacturers take a business-oriented approach, adding one or more specific nutrients within government-set regulatory quantities to some food products, increasing their commercial added value. While TF and MF programs focus on micronutrients that the general population may need, MDF may also be based on the most recognized micronutrient that appeals to the intended market, usually those with higher education and purchasing power (2, 8).
In the European Union (EU), voluntary FF is regulated by Regulation (EC) No. 1925/2006, which has been regularly amended (9 - 19), to guarantee fair trade for the industry and protection against fraud and health promotion for the consumer. This Regulation recognizes that micronutrients are added to food for a variety of purposes; for example, to improve the nutritional status of the population, to restore losses during manufacturing and processing of foods or to provide a similar nutritional value to foods for which they are intended as alternatives (9-19). The addition of micronutrients must comply with the general conditions of the Regulation. The vitamin/mineral formulations to be added must be authorized, bioavailable and adapted to the conditions on maximum addition and where appropriate minimum amounts (20). To ensure that fortification is beneficial to health, the final food must contain a minimum amount of 15% of the Nutrient Reference Values per 100 g or 100 mL in the case of products other than beverages or 7,5% per 100 mL in the case of beverages (9, 21).
The Regulation of voluntary FF is harmonized in the EU, however, differences in fortification practices between countries remain, with countries harboring separate national policies on voluntary FF (22). E.g., to manage the insufficient vitamin D and iodine status in the Finnish population, the National Nutrition Council recommends fortifying liquid milk products (1 µg/100 mL) and fat spreads (20 µg/100g) with vitamin D3 and the use of iodized salt (25 µg/g) in cooking and food industry (23, 24). In Portugal, FF is voluntary and until today, studies that assess the impact of FF on micronutrient intakes and status are still lacking. To carry out such an evaluation, it encompasses important to include micronutrient-fortified foods (M-FF) supply on the national market as well as the consumption patterns of these food products by different population groups. In our best knowledge, there is no data on M-FF available in Portugal, which reinforces the importance of the present study. In addition, it is essential that the different specialists involved in public health nutrition are aware of the supply of M-FF available in Portugal.
OBJECTIVES
This work aims to assess the supply of M-FF in Portugal through the analysis of the food labels collected within the scope of the National Food, Nutrition and Physical Activity Survey of the Portuguese General Population, IAN-AF 2015-2016 (25). This analysis will enable the identification of which fortified foods are available on the national market, the understanding of which micronutrients are most used in food fortification, which food groups are most frequently fortified and the fortification profile of fortified foods.
METHODOLOGY
Food Labels' Database
The identification and characterization of the supply of M-FF in Portugal were carried out based on a food label's database collected within the scope of the IAN-AF 2015-2016 (25). Among other dimensions, this National Survey investigated the dietary and nutritional intake of a representative sample of the Portuguese population, aged between three months to 84 years (n= 5811: 2793 men and 3018 women). For this purpose, two non-consecutive days of food diaries for children (<10 years old) and two nonconsecutive 24-hours recalls for the older age groups were used, with a time interval between 8 and 15 days, complemented with a Food Propensity Questionnaire. An electronic platform based on client-server architecture was used to manage the fieldwork and to assist the data collection. A detailed description of the survey design and methodology for the IAN-AF 2015-2016 is available elsewhere (25). During the data collection the participants were asked to describe the food consumed according to the standardized food classification and description system FoodEx2, including the characteristic ingredients (26). In addition, information on the commercial brands was also collected. If a food did not have a brand associated (e.g. fresh or homemade foods), or if the participant did not remember, the brand was classified as "Not applicable" or as "missing information", respectively. Then, a dataset was built that included the label information about the ingredients and the nutrient content (per 100 g) of each food-brand-characteristic ingredient combination.
The label information was collected from February to November 2018 in two stages. First, an online search was conducted to obtain the ingredients and nutrient information for each food. Here, the products' official website was the priority source of information. However, if there was incomplete information on the product's official website, the label information was collected through food retail chains' websites. When specific food brands information wasn't available online, several retail stores from the north region of Portugal were visited to physically collect the required information from the product's labels.
All the information regarding the ingredients, including the respective percentages whenever available, and the information on allergens was collected. Each time a product's label presented composed ingredients (e.g. Fortified Wheat Flour (Wheat Flour, Calcium Carbonate, Iron, Thiamin)), all sub-ingredients were included and identified in the database. When the nutrient content was expressed per portion size only, that information was collected and then converted to 100 g.
Finally, the databases were verified, corrections in spelling or typing errors were applied and the standardization of the terminology for some food ingredients was performed.
The food labels' original database comprises 6510 labels relating to foods for which it was possible to obtain all the information in the food and nutrition labeling. However, for the purpose of the present study, some food labels had to be excluded as the foods were not eligible to be fortified with micronutrients. According to regulatory requirements the micronutrients cannot be added to alcoholic beverages and fresh foods and for that reason alcoholic beverages and some food subgroups (e.g., fresh vegetables, meat, fish, olive oil) were excluded from the analysis (9 - 19). Food intended for specialized nutrition, namely foods for infants and young children, foods for calorie-restricted diets, dietary foods for specific medicinal purposes, and food supplements, were also excluded since they are covered by entirely separate legislation (27-31). Therefore, food labels belonging to the IAN-AF 2015-2016 subgroups "infant formulas" and "infant cereals" fitting in the groups "Dairy" and "Cereals, cereal products and starchy tubers", respectively, were excluded from the identification and characterization of fortified foods, as well as the subgroups "meal replacements" and "food and nutritional supplements" (IAN-AF 2015-2016 food group "Supplements").
Only the labels of processed foods, namely food that underwent a series of mechanical or chemical operations to modify or preserve their original structure, were considered qualified to identify M-FF. Based on these exclusion criteria, 806 labels were removed from the database and the analysis of the supply of M-FF in Portugal was performed on 5704 labels belonging to different food types within IAN-AF 2015-2016 food groups: Fruit, vegetables and legumes (FV&L); Dairy; Cereals, cereal products and starchy tubers (CCP&T); Meat, fish and eggs (MF&E); Fats and oils (F&O); Sweets, cakes and biscuits (SC&B); Non-alcoholic beverages (NAB); Salty snacks and pizzas; Meat substitutes; Milk and milk products substitute (MPs) and Table Salt (TS).
Identification of Micronutrient-fortified Foods
M-FF were identified by the presence of any micronutrients (vitamins and/ or minerals) allowed to be added to food in accordance with Regulation (EC) No. 1170/2009 in the ingredient list and simultaneously in the nutritional declaration on the food label, to ensure that food products were effectively fortified, regardless of whether they may contain certain micronutrients naturally (11). The manufacturers are obligated to provide the overall content (naturally occurring and added) of the fortified nutrient in the food label. Since it is mandatory (Regulation (EC) No. 1169/2011) that the amount of salt present in the food shall be included in the nutritional declaration - regardless of whether it is intrinsic to the food or added -, sodium was excluded from the analysis (21).
Statistical analyses were performed using Microsoft 365® - Excel®. For each IAN-AF 2015-2016 food group, the group determined the relative frequency of M-FF and the distribution of fortified foods. The number of foods fortified in each micronutrient individually considered and the prevalence of the different profiles of micronutrient fortification was analyzed. The distribution of micronutrient addition across the different IAN-AF 2015-2016 food groups was also assessed.
RESULTS
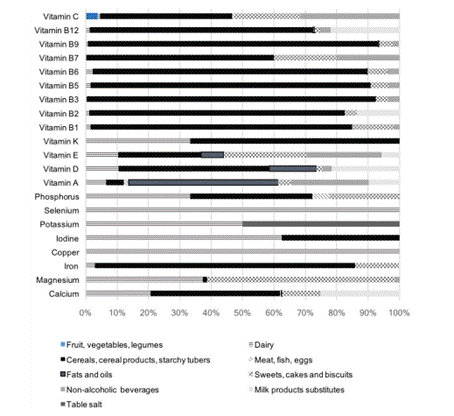
Table 1 shows the level of completeness of the label information (including the number of missing values) collected across food groups and subgroups and the identification of M-FF within each IAN-AF 2015-2016 food group. The missing information varies widely from 0% in the "canned vegetables", "processed potato products" and "baking ingredients, gelatins" subgroups to a maximum of 90.6% in the "water" subgroup. A closer look at the groups traditionally more likely to undergo fortification (e.g., dairy, cereal products; milk substitutes, oils) shows that the missing information varies from 8% in the "margarines" subgroup to 42% in the "milk" subgroup. This demonstrates an adequate coverage of the label information to perform further analysis. Of the 5704 food labels analyzed, 911 (16%) fit the definition of M-FF as described above. The largest proportion of M-FF was found in the CCP&T group (43%), followed by SC&B, (15%), Dairy (14%), NAB (11%) and MPs (10%) and F&O (7%). A smaller proportion (≤ 1%) was found in the FV&L, MF&E and TS. No M-FF were found in "Salty Snacks and Pizzas", "Meat substitutes" and "Other foods" groups. Figure 1 shows the prevalence of the different profiles of micronutrient fortification observed in the 911 fortified foods and the prevalence of use of each micronutrient in fortified foods. All the vitamins that can be added to food matrices in compliance with Regulation (EC) No. 1925/2006 and further amendments (9-19) were found in the identified M-FF. The B-complex vitamins (except vitamin B7) and vitamin D are the most frequently added to food products. Vitamin B2 is the most added vitamin (49%), followed by B6 (45%), B3 (44%), B9 (43%), D (42%), B1 (37%), B12 (35%) and B5 (35%). Vitamin C (29%) and the liposoluble vitamins A and E (both at 14%) are added to a less extent. Vitamins K and B7 were the least added and were found in a small number of food products (≤ 1%). Regarding minerals, Fe (45%), calcium (Ca; 37%) and, to a lesser extent, magnesium (Mg; 8%) are the most common minerals used in FF. A small number of M-FF (≤ 2%) with the addition of phosphorus (P), iodine (I), selenium (Se), copper (Cu) and potassium (K) was observed. Foods fortified with zinc, manganese, chromium, molybdenum, fluoride and boron were not found.
Approximately 37% (n=339) of the food products are fortified with one or two micronutrients. Of these, 70% (n=237) are fortified with one micronutrient. The number of foods fortified with three or four and with nine or ten micronutrients is similar - n=157 (17%). About 21% (n=187) of the food products are fortified with seven or eight micronutrients, while only 6% (n=56) are fortified with five 5 or six. Less than 2% (n=15) are fortified with ≥11 micronutrients, with one food product with a maximum of 17 micronutrients added.
One hundred and thirty-seven micronutrient fortification profiles, single micronutrient addition or combinations of micronutrients, were found (Figure 1). Vitamin C is the most frequently single added micronutrient to different foods (n=108), followed by Ca (n=58), Mg (n=29), vitamin E (n=10), Fe (n=9), vitamins A (n=7), D (n=7), P (n=5), vitamins B1 (n=2) and B6 (n=1) and mineral K (n=1). Vitamins B2, B3, B5, B7, B9, B12, K and minerals I, Se, Cu are always added in multiple combinations. Single addition of vitamin C occurs mainly in fruit juices, nectars and soft drinks subgroups (n=47), sweet deserts subgroup (n=46) and in commercial fruit jars subgroup (n=10). Single addition of Ca is frequently observed in plain milk, cheese, yoghurt/fermented milk subgroups (n=42), cookies and biscuits subgroup (n=8) and MPs (n=5) food group. The single addition of Mg is mainly observed in the yoghurt/ fermented milks subgroup (n=27) (data not shown).
As pointed out, most of the M-FF included in this sample are fortified with multiple micronutrients (between 3 and 17 micronutrients; n=572; 63%) with specific combinations of vitamins and/or minerals added to the products. The most prevalent profile of multiple fortification comprise nine vitamins, B2, B6, B3, B9, D, B1, B12, B5 and C, and one mineral, Fe (10 micronutrients, n=56), followed by the fortification profiles with five vitamins, B2, B6, B3, B9 and B5, and two minerals, Fe and Ca (7 micronutrients, n=50); three vitamins, B2, D and B12, and one mineral, Ca (4 micronutrients, n=49); seven vitamins, B2, B6, B3, B9, D, B1 and B5, and two minerals, Fe and Ca (9 micronutrients; n=46); two vitamins, D and A (2 micronutrients; n=44); and seven vitamins, B2, B6,
B3, B9, B1, B12 and B5, and one mineral, Fe (8 micronutrients; n=40).
Table 1: Missing labels by food subgroup/categories where fortified foods were or can be found and prevalence of micronutrient fortified foods within each IAN-AF 2015-2016 food group
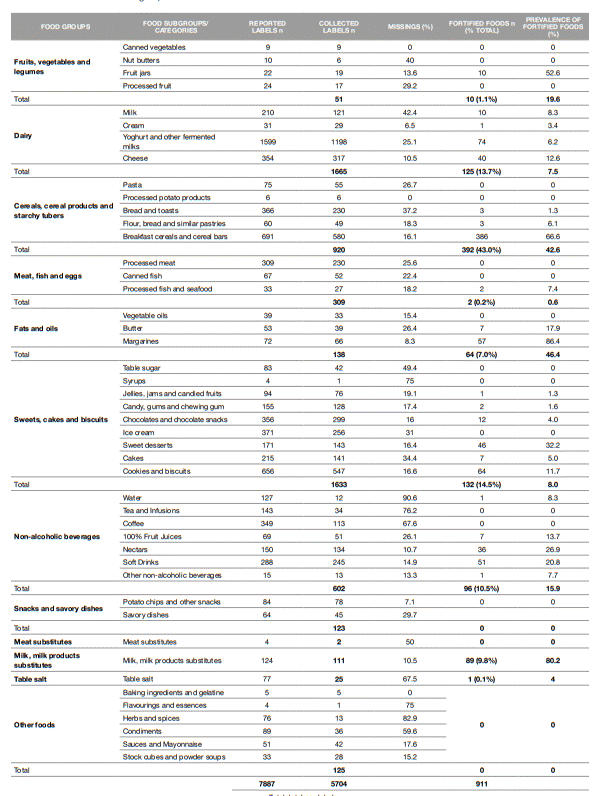
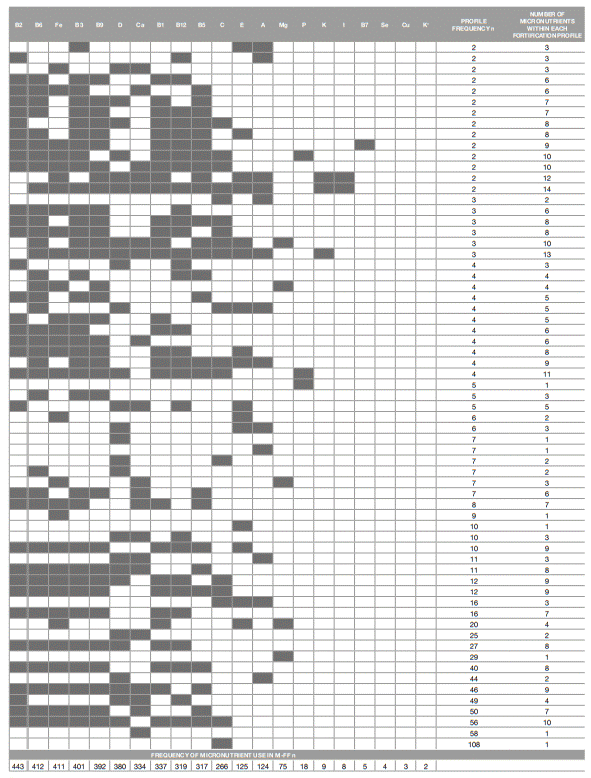
Figure 1 : Frequency of foods fortified with each micronutrient and micronutrient fortification profile found in the analyzed sample of fortified foods

Figure 1 Frequency of foods fortified with each micronutrient and micronutrient fortification profile found in the analyzed sample of fortified foods (continuation)
Graphic 1 refers to the distribution of micronutrient addition across the different IAN-AF 2015-2016 food groups, regardless of whether they are single added or added in combination.
Vitamin C is used in fortifying foods belonging to the CCP&T, NAB, SC&B and FV&L groups. Generally, the B-complex vitamins are used to fortify foods from CCP&T, SC&B, NAB groups and, except for vitamins B3 and B7, Dairy. Additionally, vitamins B2 and B12 are used to fortify foods from the MPs group. Broadly, fat-soluble vitamins A, D, E and K are added to foods belonging to the Dairy group. Vitamin A fortification is more prevalent in foods from the F&O, NAB and MPs groups. Vitamin D is particularly used in fortifying foods belonging to the CCP&T, F&O and MPs groups. The addition of vitamin E is more prevalent in foods from the CCP&T, SC&B and NAB groups. Selenium and Cu are used to fortify only a small number of dairy products. Iodine fortification is present in a few foods from the CCP&T group and Dairy. Iron fortification is very common in foods that fit into CCP&T, SC&B, and dairy groups. Calcium fortification is more prevalent in foods from the CCP&T, MPs, Dairy and SC&B groups. Magnesium fortification is mainly present in foods from the SC&B and Dairy groups. Most of NAB are fortified with vitamins C, A and E. Foods belonging to the F&O group are mostly fortified with fat-soluble vitamins.
The fortification of foods from CCP&T, especially breakfast cereals and cereal bars, SC&B and MPs groups with multiple micronutrients is frequent, with several different combinations that involve the supply of B-complex vitamins, minerals such as Fe and Ca and fat-soluble vitamins. By way of example and given the fact that they occur frequently, the specific combinations of ten, seven, nine, and eight micronutrients that are repeated 56, 50, 46 and 40 times, respectively (Figure 1), are found in different products included in the "breakfast cereals" subgroup. In turn, the pattern of addition of four micronutrients that occurs 49 times is found in foods that are part of the MPs. The patterns of addiction of four (vitamins B1, E and minerals Fe, Mg) and seven micronutrients (vitamins B2, B6, B3, B1, B5 and minerals Ca, Fe) occur 20 and eight times, respectively, are found in commercial cookies and biscuits subgroup.
Regarding NAB, fortification with multiple nutrients is also frequent although most of the combinations involve only vitamins: from the most frequently added antioxidant combination (vitamins C, A and E; 16 times) to the least, yet more complex 9 vitamin combination (Vitamins A, B1, B12, B3, B5, B6, B9, C, E; 9 times). Only two food products simultaneously added vitamins (B-complex) and minerals (Ca and/or Mg).
DISCUSSION OF THE RESULTS
FF is a method of deliberately increasing the vitamin and/or mineral (or other essential nutrient) content of specific foods as a public health strategy and an effective way to ensure the nutritional quality of the food supply, to minimize micronutrient deficiencies (32,33). Nevertheless, it can also be at the basis of marketing strategies in product development (32). Knowledge of the current Portuguese trends in voluntary FF by the food industry is essential to further assess the impact of such practice on micronutrient intakes and the adequacy of intakes by consumers beyond commercial interests.
The analysis of a food labels' database collected under the framework of the IAN-AF 2015-2016 (25) to identify and characterize the supply of M-FF available on the Portuguese market revealed that 16% (911/5704) of the products analyzed were considered M-FF. It is important to highlight that there may be more M-FF on the market that have not been covered herein because either they were not identified as consumed or the food label was not collected. In the latter case, the main reasons for their non-existence were either the fact that reported brands were specific from a particular geographical region and were not available in the visited retail stores, or the time gap between report and label collection affected their availability, particularly special or seasonal editions. Also, between the collection of data and the present analysis there is a gap of five years. Since the fortified-food market is very
dynamic, it is possible that, at the current time, there are other M-FF that are not covered in this work.
The results showed that vitamins B2, B6, B7, B9, D, B1 and B12 and minerals Fe and Ca were the most common micronutrients used to fortify foods and 63% of the food products delivered an array of vitamins and/or minerals. This qualitative fortification profile is similar to the many observed Europe-wide. Wagner et al. (2005) evaluated the situation of FF in Austria through observation of the number of fortified products by inspections of supermarkets and retail outlets (n=470) and found that vitamins C (73%), B6 (43%) and B3 (37%) and the minerals Ca (23%) and Fe (16%) were the most frequently added micronutrients to foodstuffs (34). Sicinska et al. (2013) identified 588 food products fortified with vitamins in the Warsaw market (Poland) in 2012 (35). The number of vitamins added to food products ranged between one to twelve and vitamins C (58%), B6 (46%) and B12 (45%) were the most frequently added, while vitamin B7, similarly to that encountered in the present work, was present only in a few food products (16%). In a later study, the same research team identified 301 foods fortified with minerals, in which the addition of minerals ranged from one to seven, with Ca (71%), Fe (47%) and Mg (21%) being the most frequently added (36).
Like other studies, the presence of M-FF is more marked in some food subgroups, namely, breakfast cereals and cereal bars, commercial sweets, including cookies and biscuits, dairy products, juice and soft drinks and milk product substitutes (35-39).
Ready-to-eat cereals and dairy products have a long history of fortification given their versatility, availability on the market, affordability, widespread acceptance and consumption around the world (3). They are suitable classical carriers for Fe, B-complex vitamins and Ca and vitamins D and A (3,30,31). This work shows that ready-to-eat cereals are mainly fortified with B-complex vitamins (B1, B2, B3, B5, B9 and B12), Fe and Ca. Fortified dairy products are carriers of several micronutrients, in particular Ca, Mg, Fe, P, vitamins D, A, E and also B6. The SC&B group encompasses various subgroups that include different types of foods. In this study, the subgroups with the greatest representation of M-FF are “cookies and biscuits” and “sweet desserts”. Forty-six gelatin products, belonging to commercial sweet deserts subgroup are mainly fortified with vitamin C. This can be explained by the fact that vitamin C has a multifunctional role, enriching the products from a nutritional point of view and contributing to the extension of products' shelf-life, due to its antioxidant capacity (42). The commercial cookies and biscuits are fortified with several micronutrients, standing out B-complex vitamins, Fe and Ca. One of the main ingredients used in the production of commercial cookies and biscuits is cereal flour, particularly wheat flour. Similarly, to ready-to-eat cereals, it is frequently fortified with B-complex vitamins, Fe and Ca, to restore the levels of these micronutrients lost during cereal processing operations. Operations such as dehulling, milling and refining may alter the nutritional composition of subsequent products (43, 44). Milling grains, for example, results in major losses of vitamins B1, B2, B5, B6, B7, B9 (around 70-80%) and Ca, Fe and Mg (45). In most European countries, the fortification of cereal flour with micronutrients, despite being a current practice, is voluntary (46), except in the United Kingdom where it is mandatory to fortify wheat flour with vitamins B1, B3, and minerals Ca and Fe (47).
NAB, particularly fruit juices and soft drinks also have a long history of micronutrient addition. Beverages can be fortified with micronutrients for several reasons namely, to restore the vitamins lost during processing or to replace or enhance the vitamin and mineral content to improve their nutritional contribution to the diet. While the restoration of vitamins in fruit juices has been carried out for many years, the fortification of carbonated and still soft drinks is more recent (42). Vitamin C fortification has been the most common practice but there are products also containing B-complex vitamins, Ca and vitamin D (32). Our results show that vitamin C is added to almost every product from the NAB group, single or combined with vitamins with antioxidant capacity (vitamins A, E) or/and from the B-complex. The preference for vitamins rather than minerals may be justified by the fact that the addition of minerals to beverage formulations can lead to chemical reactions that may affect the sensory acceptability (color, flavor, texture) and shelf-life stability of the final products (42).
Due to ethical, health and sustainability concerns consumers are increasingly interested in reducing their consumption of animal-based food products, so the food industry is creating new alternative products from plant-based ingredients that simulate many physicochemical and sensory attributes associated with analogous animal-derived foods. In this sense, plant-based dairy substitutes have become increasingly popular over the past few years and numerous commercial products are already available (48,49). Given that they are meant to replace the consumption of dairy products, they must provide similar essential nutrients to be nutritionally equivalent, namely minerals (such as Ca), and vitamins (such as vitamins A, D, and B12) (50). Our results are in line with this.
Although MDF can contribute to a wide variety of food sources of micronutrients thus helping consumers meet their daily requirements, this practice raises concerns not only regarding the promotion of healthy eating but also about possible risks associated with excessive micronutrients intake (51). There is a risk that food manufacturers could market fortified, but otherwise relatively unhealthy foods (52). Regulation (EC) No. 1925/2006 and its amendments (9 - 19), refers that vitamins and minerals cannot be added to unprocessed food such as fruits, vegetables, meat and fish and to alcoholic beverages however it does not mention which type of processed foods may be fortified, allowing food manufacturers the opportunity to decide which foods are best suitable for fortification with micronutrients regardless of their healthy/ unhealthy nutritional profiles. Indeed, this study shows that two of the IAN-AF 2015-2016 food subgroups not represented in the Portuguese Food Wheel Guide (53) - SC&B and NAB (except water) - are among those with the highest proportion of added micronutrients. According to IAN-AF 2015-2016, the prevalence of daily consumption of soft drinks and nectars in Portugal is 18%. After water, soft drinks are the second most consumed beverages (88 g/day), and the consumption of nectars corresponds to 20 g/day. The consumption of SC&B food group is nearly 70 g/day. The contribution of the SC&B and NAB food groups to the daily intake of added sugars is 59.2% and 15.4%, with the subgroup's sweets, soft drinks, cookies and biscuits and nectars contributing with 16.8%, 11.9%, 8.6% and 4.0%, respectively (54). In this context, fortification of this type of food products, usually accompanied by nutritional claims related to micronutrients, make the products more attractive to consumers, leading them to believe that they are beneficial to health (55, 56). However, this strategy may be misleading since these products tends to be rich in added sugar and/or fat and/or salt. Thus, it is imperative to invest in increasing the nutritional literacy of consumers, educating them to carefully read the food labels, emphasizing the list of ingredients and the nutritional declaration, providing them the knowledge that will empower them to make informed food choices. Furthermore, it is also important that Official Authorities exercise some control over voluntary FF, for example by imposing limits on the addition of essential nutrients to foods with an unbalanced nutritional profile. This issue is particularly important in the case of fortified foods targeted at children and adolescents and may be comparable with Portuguese Law No. 30/2019 of April 23, which restricts advertising aimed at children <16 years old of foodstuffs that contain high energy value, high salt, sugar, saturated fatty acids and processed fatty acids content (57). Currently, the European Commission is planning the introduction of nutrient profiles in product development (58). This strategy will most certainly have an impact on the use of nutrition claims (59) that would only be allowed for foods which fulfil predetermined content requirements regarding total fat, saturated fat, sugars, and salt contents. When this happens, it is plausible to assume that there may also be changes in voluntary FF practices. It should also be noted that currently the EU legislation does not specify any upper limits or maximum amounts for vitamins and minerals in fortified foods, which means that food business operators are responsible for ensuring that the amounts of micronutrients in their food products are safe. Inadequate intakes due to too low micronutrient intakes can have serious health consequences, however excessive intakes may also result in adverse health effects (60). In response to concerns about excessive intakes from the widespread use of dietary supplements and extensive voluntary FF, new EU regulation regarding maximum amounts of micronutrients in food supplements and fortified foods is currently being prepared to enter into force in 2024 (61).
CONCLUSIONS
Through the analysis of the food labels collected under the IAN-AF 2015-2016, it was possible to identify and characterize the supply and consumption of fortified foods in Portugal for the first time. A total of 911 fortified foods were identified from a sample of 5704. B-complex vitamins (B1, B2, B3, B6, B9 and B12), vitamin D, Fe and Ca are the most common micronutrients used to fortify foods. Fortified foods fit mostly within specific product categories such as breakfast cereals and cereal bars, commercial sweets, cookies and biscuits, dairy products, juice and soft drinks and dairy substitutes. Oddly, such groups are not always coupled to balanced nutritional profiles, and fortification should not be used as a tool to mislead the consumer to increased consumption of less healthy food products.
This study is the first step to further investigating the contribution of fortified foods to usual micronutrient intakes by the Portuguese population or specific subgroups, its adequacy, and the potential risks of excessive intake.
AUTHORS’ CONTRIBUTIONS
The authors confirm contribution to the paper as follows: APM: Conceptualization; Methodology; Analysis and interpretation of results; Writing - draft manuscript; DC: Conceptualization; Software; Formal analysis; Data curation; CC: Conceptualization; Data collection; Formal analysis; CL: Resources; Funding acquisition; AMG: Conceptualization; Methodology; Supervision; Writing - review & editing; DT: Conceptualization; Methodology; Supervision; Resources; Writing - review & editing.