Serviços Personalizados
Journal
Artigo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Acessos
Acessos
Links relacionados
-
 Similares em
SciELO
Similares em
SciELO
Compartilhar
GE-Portuguese Journal of Gastroenterology
versão impressa ISSN 2341-4545
GE Port J Gastroenterol vol.22 no.1 Lisboa fev. 2015
https://doi.org/10.1016/j.jpge.2014.07.003
IMAGES IN GASTROENTEROLOGY AND HEPATOLOGY
Gastric Mixed Adenoneuroendocrine Carcinoma
Carcinoma Adenoneuroendócrino Misto Gástrico
Dália Fernandesa,b,∗, João-Bruno Soaresb, Carla Rolandab,c
a Department of Gastroenterology, Hospital Cova da Beira, Covilhã, Portugal
b Department of Gastroenterology, Hospital de Braga, Braga, Portugal
c Life and Health Sciences Research Institute, University of Minho, Braga, Portugal
* Corresponding author.
Gastrointestinal mixed adenoneuroendocrine carcinomas (MANECs) are rare tumors and, as the name implies, are characterized by the presence of both components, exocrine and neuroendocrine, each one representing at least 30% of the lesion.1 It is unclear if they originate from proliferation of different cell lineages or from stem cells capable of differentiating along multiple cell lineages. They may present as polypoid lesions, with variable size. Treatment and prognosis is determined by the more aggressive component.2
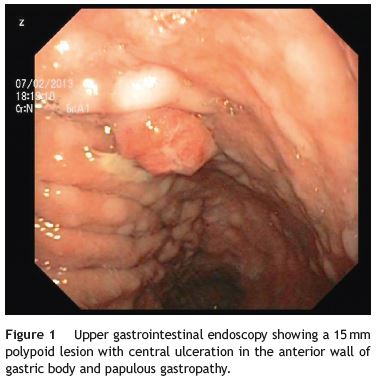
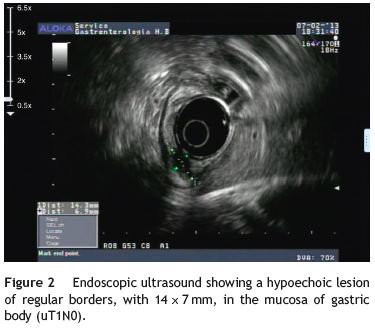
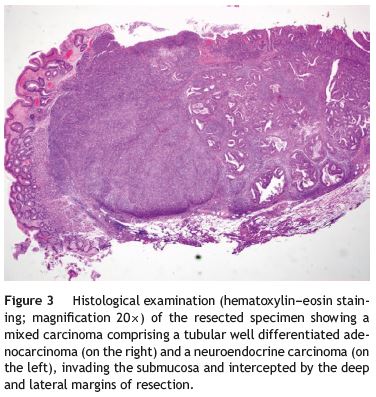
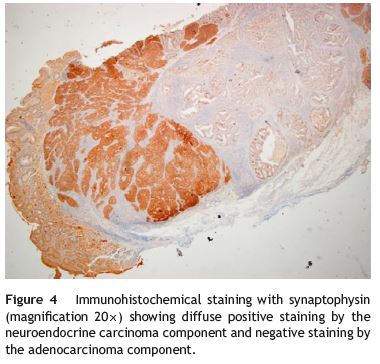
An 84-year-old woman was referred to upper gastrointestinal endoscopy for evaluation of dyspepsia. Endoscopic examination showed a 15 mm polypoid lesion with central ulceration in the anterior wall of gastric body and papulous gastropathy (Fig. 1). Biopsies of the gastric lesion showed a neuroendocrine tumor and random biopsies of the gastric mucosa showed chronic atrophic gastritis and intestinal metaplasia. Serum antibodies to parietal cells and to intrinsic factor were negative. Serum level of chromogranin A level was 129.6 ng/mL (normal level < 80 ng/mL). Thoracic and abdominal CT revealed neither locoregional adenopathies nor distant metastasis. Single-photon emission computed tomography showed no lesion with uptake of 99mTc-tektrotyd. Endoscopic ultrasound (EUS) revealed that the gastric lesion was an uT1N0 tumor with 14×7 mm (Fig. 2). The gastric lesion was resected endoscopically in one fragment using the lift and cut technique. Histological examination of the resected specimen showed a mixed adenoneuroendocrine carcinoma, comprising two closely juxtaposed elements each representing almost 50% of the tumor: (i) tubular well-differentiated adenocarcinoma; (ii) neuroendocrine carcinoma staining positively for chromogranin A and synaptophysin and with a mitotic index higher than 20 mitoses per 10 high-power fields (Figs. 3 and 4). The tumor invaded the submucosa and was intercepted by the deep and lateral margins of resection (Fig. 3) and so the patient was proposed for surgery. We decided for laparoscopic atypical gastrectomy which has significant lower rate of complications than the recommended complete or partial gastrectomy (en bloc resection) with local lymph node resection due to patients age and poor performance status, the low risk (11%) of lymph node metastasis in cases of tumors with <2 cm and no extension into muscle on EUS,3,4 and the absence of adenopathies in EUS and abdominal CT. The surgical specimen showed no residual lesion.
The natural history of the gastric MANECs is unclear, but is mainly determined by the more aggressive component (the neuroendocrine component in our case). Like the pure gastric neuroendocrine tumor, the prognosis of MANEC depends primarily of stage (TNM) and also of histologic grading and mitotic count/Ki67 index. Despite the scarce information about this type of cancer, the survival rate at five years was slightly better than pure gastric adenocarcinoma, 33.3% vs 24% in one study.5
References
1. Rindi G, Arnold R, Bosman FT. Nomenclature and classification of neuroendocrine neoplasms of the digestive system. In: Bosman FT, Carneiro F, Hruban RH, Theise ND, et al., editors. WHO classification of tumors of the digestive system. Lyon: IARC; 2010. p. 13. [ Links ]
2. Öberg K, Knigge U, Kwekkeboom D, Perren A. Neuroendocrine gastro-entero-pancreatic tumors: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol. 2012;23:124-30. [ Links ]
3. Saund MS, Al Natour RH, Sharma AM, Huang Q, Boosalis VA, Gold JS. Tumor size and depth predict rate of lymph node metastasis and utilization of lymph node sampling in surgically managed gastric carcinoids. Ann Surg Oncol. 2011;18:2826-32. [ Links ]
4. NCCN clinical practice guidelines in oncology. Neuroendocrine tumors (version 2.2014) (accessed 12.02.14). Available: http://www.nccn.org/professionals/physician gls/f guidelines.asp [ Links ]
5. Borch K, Ahrén B, Ahlman H, Falkmer S, Granérus G, Grimelius L. Gastric carcinoids: biologic behavior and prognosis after differentiated treatment in relation to type. Ann Surg. 2005;242:64-73. [ Links ]
*Corresponding author
E-mail address: daliafernandes09@gmail.com (D. Fernandes).
Ethical disclosures
Protection of human and animal subjects. The authors declare that no experiments were performed on humans or animals for this study.
Confidentiality of data. The authors declare that no patient data appear in this article.
Right to privacy and informed consent. The authors declare that no patient data appear in this article.
Conflicts of interest
The authors have no conflicts of interest to declare.
Financial disclosures
This work had no grant support or other assistance.
Author contributions
All the authors (Dália Fernandes, João-Bruno Soares and Carla Rolanda) contributed for conception and design, analysis and interpretation of the data, drafting and critical revision of the manuscript for important intellectual content and final approval of this article.
Received 24 February 2014; accepted 29 July 2014














