Serviços Personalizados
Journal
Artigo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Acessos
Acessos
Links relacionados
-
 Similares em
SciELO
Similares em
SciELO
Compartilhar
GE-Portuguese Journal of Gastroenterology
versão impressa ISSN 2341-4545
GE Port J Gastroenterol vol.22 no.3 Lisboa jun. 2015
https://doi.org/10.1016/j.jpge.2015.03.009
REVIEW ARTICLE
New Trends in Inflammatory Bowel Disease
Novas Estratégias Terapêuticas na Doença Inflamatória do Intestino
Carolina Palmela, Joana Torres, Marilia Cravo∗
Gastroenterology Department, Surgical Department, Hospital Beatriz Ângelo, Loures, Portugal
* Corresponding author.
ABSTRACT
Inflammatory bowel disease (IBD) is a chronic idiopathic inflammatory disease of the gastrointestinal (GI) tract. In the past decade a shift in the treatment paradigm of IBD has ensued. The availability of drugs capable of inducing mucosal healing, combined with the recognition that IBD is not an intermittent disease, but rather a progressive one causing bowel damage and disability, led us to a more stringent strategy. Tailored therapy with more aggressive treatment in high-risk patients, treating beyond symptoms, intervening early before damage occurs, optimizing therapeutic regimens, and actively pursuing sustained remission and sustained control of inflammation are strategies that are slowly being incorporated in our clinical practice. Furthermore, new drugs targeting different immunological pathways, such a s vedolizumab, have recently been approved and therefore more therapeutic resources for patients failing anti-tumour necrosis factor alpha (anti-TNFa) agents will be available.
The future years look promising for IBD. Hopefully the new trends in IBD management, combined with new drugs, will make possible to change the course of disease and provide better therapy and quality of life for patients suffering from this disabling disease.
Keywords: Antibodies, Monoclonal; Colitis, Ulcerative; Crohn Disease; Inflammatory Bowel Diseases; Molecular Targeted Therapy; Drug Monitoring
RESUMO
A doença inflamatória intestinal (DII) é uma doença idiopática crónica e incapacitante do trato gastrointestinal. Na última década tem-se assistido a uma modificação nas estratégias de abordagem e tratamento do doente com DII. O desenvolvimento de fármacos potentes com capacidade de induzir a cicatrização da mucosa, aliado ao reconhecimento do carácter progressivo da doença, com dano intestinal irreversível e compromisso da qualidade de vida, conduziu à adoção de estratégias mais rigorosas. Tem-se assistido à incorporação de novas estratégias na prática clínica atual, tais como terapêutica individualizada mais agressiva em doentes com mau prognóstico mais precocemente, uso de outros objetivos terapêuticos para além da remissão sintomática, intervenção precoce para evitar o desenvolvimento de dano intestinal irreversível, otimização da terapêutica com objetivo de alcançar a remissão sustentada e o controlo da inflamação. Adicionalmente, a aprovação de novos fármacos com ação em vias imunológicas alternativas, como o vedolizumab, permitirá alargar o espetro terapêutico no doente não respondedor a agentes anti-factor de necrose tumoral alfa.
O futuro parece promissor na área da DII. É expectável que as novas estratégias de abordagem do doente com DII, aliadas ao desenvolvimento de novos fármacos, permitam alterar o curso natural da doença, evitando o dano intestinal irreversível e possibilitando a melhoria da qualidade de vida destes doentes.
Palavras-Chave: Colite Ulcerosa; Doença de Crohn; Doença Inflamatória Intestinal; Terapia Molecular Dirigida; Monitorização de Medicamentos
1. Introduction
In the past years, inflammatory bowel disease (IBD) has witnessed major advances both at the fundamental and clinical levels. Key discoveries in diseases pathogenesis led to the development and refinement of therapeutic strategies paving the way for a new era in targeted drug development.1
Therapeutic management in IBD has also suffered a major shift in the past years. The traditional paradigm of gradually introducing the least toxic but less powerful drugs and repeated courses of steroids has progressively moved to favour the early introduction of immunomodulation and most recently biologic therapy, in order to modify the natural history of disease.2
The discovery and introduction of anti-TNFa agents have constituted a major breakthrough for patients and clinicians. These novel therapies offer the ability to induce and maintain remission, heal the mucosa, and reduce surgeries and hospitalizations.3 However, many patients will still be refractory to treatment or loose response over time. Therefore, therapeutic drug monitoring, as a way of optimizing anti-TNFa use, has been an area of intensive research. Furthermore, new drugs are expected to hit the clinic. Vedolizumab, an anti-integrin, is eagerly awaited for patients who have failed anti-TNFa and it has shown to be able to induce and maintain remission in patients with Crohns disease (CD) and ulcerative colitis (UC).
We are increasingly using prognostic and predictive factors at diagnosis and during follow-up to guide our therapeutic decisions. We now recognize that in order to block disease progression, it is crucial to intervene early before damage occurs.
In this review we will briefly discuss new therapies in IBD, and we will focus on new trends that are emerging in the treatment of IBD patients, with the goal of optimizing current therapeutic strategies and blocking disease progression.
2. Prognostication in IBD
IBD is a heterogeneous condition with a highly variable clinical course, with some patients following a mild course while others experience early and aggressive disease progression.4-6 Therapeutic strategies in IBD result from a delicate and difficult balance between benefits and risks of more aggressive therapies. Early treatment with immunomodulators and/or anti-TNFa is expected to change the natural history of disease, especially for CD, but with the risk of overtreating some patients with mildmoderate disease who would not need such intensive therapy. Therefore, the possibility of patient profiling using prognostic factors at diagnosis (including clinical, laboratory and imaging criteria) for the selection of the best suited candidates for early aggressive therapies is of main importance (Tables 1 and 2).7,8

Several definitions of poor prognosis in CD have been suggested. Disabling CD was arbitrarily defined by Beaugerie et al9 in 2006 by the presence of at least one of the following criteria in the 5-year period after diagnosis: More than two steroid courses and/or steroid dependency; further hospitalization after diagnosis for flare up or complications of the disease; presence of disabling chronic symptoms; need for immunosuppressive therapy; and intestinal resection or surgery for perianal disease. In a recent meta-analysis8 including 1961 patients,9-11 the demographic and clinical characteristics associated with significantly higher risk of developing disabling disease at five years after initial diagnosis were: young age (<40 years) at diagnosis, initial requirement of steroids for treating the first flare and perianal disease.8
In 2012, Zallot and Peyrin-Biroulet12 suggested a different definition for complicated IBD. Complicated CD was defined as the presence of bowel damage (stricture, abscess and/or fistula) and/or the requirement for surgery and/or the presence of extra-intestinal manifestations. Clinical factors associated with complicated CD include young age at diagnosis, small bowel disease (ileal and/or ileocolonic), upper gastrointestinal extent, stricturing or penetrating behaviour, perianal disease, severe endoscopic lesions (deep ulcerations) at index colonoscopy and smoking.12-16
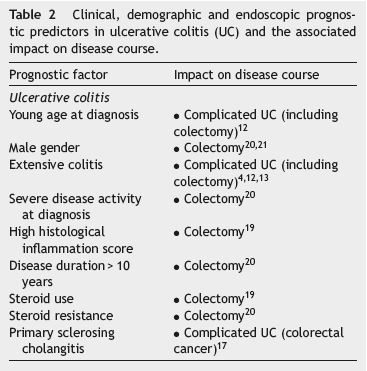
Complicated UC was defined as the development of colon cancer and/or the need for surgery (colectomy) and/or the presence of extra-intestinal manifestations.12 Clinical predictors for complicated UC include young age at diagnosis, male gender, extensive colitis, severe disease activity at diagnosis, high histological inflammation score, the presence of primary sclerosing cholangitis, steroid use and steroid resistance.4,12,13,17-21
Besides clinical characteristics, various immunologic and genetic markers have been associated with a worse disease course. The presence of antibodies to Saccharomyces cerevisiae (ASCA), to Escherichia coli outer-membrane porin C (OmpC), to CD-related bacterial sequence I2, and to the CBir1 flagellin has been linked to early CD onset, stricturing and penetrating behaviour, and need for early small bowel surgery.22,23 In the genetic field, the IBD CHIP project24 concluded that carriage of some NOD2 variants was the most important genetic factor, being an independent predictive factor for ileal CD disease, stenosing and penetrating behaviours and need for surgery. Other genetic markers have been studied, including PRDM1 variants, IL23R, JAK2 and TNFS15 which also appear to be associated with disabling CD.7 However, so far, there is insufficient data to support the recommendation to use antibody or genetic testing in clinical practice to predict IBD course.25,26
3. New endpoints in IBD
In CD, subclinical inflammation often persists and there is a progressive evolution to structural and irreversible bowel damage, which frequently requires surgical intervention.27 Current clinical and endoscopic scores only measure disease activity at a specific time. The recently published Lémann Index (Crohns disease digestive tract damage score) is the first tool that aims to measure the cumulative structural bowel damage, including strictures, penetrating lesions (abscesses and fistulae), and surgical resection. This evaluation includes damage location, extent, severity, progression and reversibility (measured by imaging techniques and the need of resection).27 In each organ (upper GI tract, small bowel, colon/rectum and anus) the score ranges from 0.0 (absence of damage) to 10.0 (complete resection).27 As expected, the median Lémann Index increases with disease duration. There are numerous future potential applications for this index, including the assessment of disease progression and patients heterogeneity, the identification of parameters at diagnosis associated with a high risk of rapid progression, and the impact of therapy strategies on longterm outcomes.27,28
Another emerging endpoint is disability, as an objective way of measuring the impairments, activity limitations, and participation restrictions that are experienced by patients suffering from IBD in different areas or health domains.29 In the years to come, it is likely that predictors of bowel damage and disability will be identified, as well as the impact of new drugs and therapeutic strategies in preventing them.
4. New therapeutic strategies - tight control monitoring and the treat-to-target strategy
The traditional symptom-oriented management has shown not to improve long-term outcomes in IBD.7 Therefore a more proactive attitude is emerging in IBD, similar to other chronic diseases.7
The treat-to-target strategy is based on frequent assessment of disease activity, using objective markers of inflammation and subsequently adjusting therapy accordingly to reach the pre-established target.7,28 The ultimate objective of this approach is to modify the natural course of the disease.28 The ideal target to achieve in IBD is still a matter of debate and likely to evolve. So far, mucosal healing (MH) seems to be the best established therapeutic endpoint both in UC and CD.30 The definition of MH used in most clinical trials is the complete absence of all inflammatory and ulcerative lesions.30,31 Several clinical trials and population-based studies have demonstrated that MH achievement is associated with better outcomes, such as a lower incidence of disease relapse, hospitalization and surgery.30,32-35
Due to the invasive nature and cost of endoscopy, several other treatment goals have emerged, such as the use of cross-sectional imaging and inflammation biomarkers.36 Although new endoscopic and therapeutic techniques, as deep enteroscopy, have a specific role in diagnosis and treatment in IBD, magnetic resonance enterography (MRE), computed tomography (CT) and ultrasound (US) may be alternatives to ileocolonoscopy.37 They have the advantages of being less invasive, able to assess the small bowel and providing information about the transmural nature of inflammation.36 CT has a similar accuracy to MRE for assessing disease activity in CD, but the risk of radiation exposure limits its use as a monitoring tool.36 Rimola et al38 suggested and validated a Magnetic Resonance Index of Activity (MaRIA) score to quantify CD activity based on MRI findings in each ileocolonic segment, and found a strong correlation with CDEIS (Crohns Disease Endoscopic Index of Severity). In a systematic review by Panes et al39, including 16 studies with a total of 1629 segments assessed, the overall sensitivity and specificity of MRE for the assessment of disease activity was 80% and 89%, respectively. However, the accuracy of MRI to monitor therapeutic responses has not yet been fully ascertained. In a recent multicentre prospective study with 48 patients receiving corticosteroids or anti-TNFa, the disease activity was assessed before and after treatment using MRE (MaRIA score) and ileocolonoscopy (CDEIS). The authors found a significant correlation between imaging activity index and CDEIS scores, with MRE determining ulcer healing with 90% accuracy.40
Non-invasive biomarkers of intestinal and systemic inflammation may represent an attractive alternative for evaluating disease activity, independently of disease location. The most broadly used and extensively studied biomarkers are faecal calprotectin (FC) and serum C-reactive protein (CRP).36 There is a correlation between these markers concentration and endoscopic lesions, risk of relapse, risk of complications and response to therapy.41 Elevated biomarker levels are associated with endoscopic and histologic bowel inflammation. In general, this correlation is higher when using faecal biomarkers, such as calprotectin.41 Nonetheless, in CD patients with small bowel involvement, FC seems to have a weaker correlation with endoscopic activity. In such cases, CRP levels may better reflect the transmural inflammatory process.36 The association between non-invasive biomarkers levels and intestinal inflammation is not always accurate. Up to one-third of patients with intestinal inflammation may have normal CRP levels.42 However, in CD patients who have a raised CRP concentration at baseline, changes in CRP levels may provide useful information in monitoring response to treatment.7 Jürgens et al43 showed that in CD patients treated with infliximab, early normalization of CRP levels correlated with sustained long-term response to therapy, while a persisting increase in CRP under therapy was associated with a high likelihood of relapse. Biomarkers have been studied less in the postoperative setting. The Poccer study was a prospective study aiming at identifying the best strategy for prevention of post-operative CD. As a part of this study several biomarkers were measured. No correlation between increased levels of CRP and disease recurrence was observed.44 Whereas, following surgery, levels of FC >100 mg/g indicated endoscopic recurrence with 89% sensitivity and 58% specificity, and a negative predictive value of 91%.45
CRP and faecal calprotectin may be best used as screening tool to decide the ideal timing for endoscopic or cross-sectional imaging reassessment.36 A recent metastudy analysing data from six studies (551 CD patients) found that in patients with symptoms (CDAI > 220), CRP≤5 mg/l or calprotectin≤200 mg/g predicted a CDEIS≤6 with a sensitivity and specificity of 83% and 71%, respectively.46 Therefore with the use of appropriate threshold levels, these biomarkers could help to make decisions about the ideal timing of endoscopy.41
Still, the reliability of these biomarkers to predict MH is not fully established. Depending on the calprotectin concentration threshold used, its sensitivity and specificity in predicting absence of MH were 70-100% and 44-100%, respectively.36 Recently, DHaens et al47 suggested a faecal calprotectin cut-off value of 250 mg/g levels above this concentration predicted large ulcers in CD (sensitivity 60%, specificity 80%) and active mucosal disease activity (Mayo >0) in UC (sensitivity 71%, specificity 100%). In CD, a concentration <250 mg/g predicted MH (CDEIS <3) with a sensitivity of 94% and a specificity of 62%.47 In a subanalysis of the STORI cohort the combination of faecal calprotectin (at a threshold of 250 mg/g) and CRP (at a threshold of 5 mg/l) has shown to improve the ability to predict mucosal healing (sensitivity 70%, specificity 70%).48
There are some issues that need to be defined, namely the optimal time frame for assessment of treatment goals in a treat-to-target strategy. Using MH as the target, recent trials in CD found that in patients under treatment with immunosuppressives, anti-TNFa or a combination of both, the time frame for achieving MH ranged from 10 to 26 weeks.49-52 Allen and Peyrin-Biroulet28 suggested that, independently of the method used (endoscopy, MRE, CRP and/or faecal calprotectin), the treatment goal should be first assessed at 3-6 months after introduction of diseasemodifying agents. Bouguen et al7 proposed that, in CD patients at high risk for disease-related progression or complications, MH should be assessed every 6 months until the disappearance of ulceration and every 1-2 years thereafter.
Another controversy issue in this treat-to-target strategy is the management of insufficiently healed patients.36 It is not yet clear whether patients in clinical remission and with residual mucosal inflammation have a worse prognosis than patients with complete MH.7
The minimal degree of mucosal improvement required to alter outcomes is not yet known. Ferrante et al53, in a post hoc analysis of the SONIC trial data, showed that a decrease of at least 50% in CDEIS from baseline to week 26 (endoscopic response) was associated with steroid-free remission at week 50, with a similar specificity and sensibility to mucosal healing (absence of ulcers). The impact of this finding on long-term outcomes needs to be clarified.
5. New drugs in IBD
An important step in the pathogenesis of IBD involves the migration of leukocytes into the gut. The recruitment of Tcells and other inflammatory cells to the intestinal mucosa leads to pro-inflammatory cytokine production, affecting the endothelial barrier and inducing cell apoptosis.54 Integrin antagonists are anti-adhesion molecules which block the interaction between inflammatory and endothelial cells, thereby disrupting trafficking of T-lymphocytes into the inflamed gut.55
The first anti-adhesion agent that proved to be effective in the treatment of IBD was natalizumab. Natalizumab is an anti-a4-integrin IgG4 non-gut-selective antibody, also used for treating multiple sclerosis, which binds to a4b1 and a4b7 integrins, inhibiting T-lymphocyte adhesion to vascular cell adhesion molecule-1 (VCAM-1) and mucosal addressin cell adhesion molecule-1 (MAdCAM-1).56,57 The initial enthusiasm surrounding its success was halted by reports of the aggressive progressive multifocal leukoencephalopathy.58,59 Nevertheless, this agent provided proof of concept and paved the way for the development of new anti-adhesion agents, such as vedolizumab (VDZ).1 VDZ is a humanised anti-a4b7 integrin-IgG1 monoclonal antibody that binds to MAdCAM-1. Unlike natalizumab, VDZ is gut specific, providing gut-selective immunosuppression with similar efficacy, with a lower risk of systemic infection.60 A recent study using the anti-MAdCAM-1 antibody showed that treatment with this drug does not affect the populations of CD4 and CD8 lymphocytes in the cerebrospinal fluid.61
The results of the phase III, randomized, placebocontrolled trials (RCT) on the induction and maintenance of VDZ in UC and CD (the GEMINI I and II studies) have recently been published.62,63 In the UC induction study,62 374 patients were randomized to VDZ at a dose of 300 mg or placebo at week 0 and week 2. During induction, at week 6, 47.1% of the patients on VDZ versus 25.5% of the patients on placebo achieved clinical response (p < 0.001). Rates of mucosal healing (Mayo score 0 or 1) were 40.9% for VDZ and 24.8% for placebo (p = 0.001). A second cohort of 521 patients received open-label VDZ at weeks 0 and 2, with disease evaluation at week 6. Responders from both cohorts were then randomized to receive drug every 4 or 8 weeks or placebo. A total of 41.8% and 44.8% maintained remission when receiving medication every 8 weeks and every 4 weeks, respectively; patients on placebo had maintenance of remission at a rate of 15.9% (p < 0.001 for the two VDZ groups, respectively, vs. placebo).62 Adverse events were similar in both groups.62
In the CD induction study,63 368 patients were randomized to 300 mg of VDZ or placebo at weeks 0 and 2. A second cohort of 747 patients received open-label VDZ. All patients were followed through 6 weeks. Patients on VDZ had a statistically significant difference in clinical remission of 14.5 versus 6.8% in placebo (p = 0.02) at week 6, but no difference in CDAI-100 response or reduction in mean Creactive protein (CRP) levels at week 6.63 Patients from both cohorts with a clinical response at week 6 (461 patients) entered the maintenance trial and were randomized to receive VDZ every 8 weeks, or every 4 weeks, or placebo for up to 52 weeks. In the VDZ groups, 39% and 36.4% of those assigned to VDZ every 8 and 4 weeks, respectively, were in clinical remission at week 52, as compared with 21.6% assigned to placebo (p < 0.001 and p = 0.004 for the two VDZ groups, respectively, vs. placebo). There was also statistical significance in CDAI-100 response, and steroidfree remission at week 52 for both VDZ groups versus placebo (p < 0.001 and 0.004, respectively). Patients with prior anti-TNFa exposure had lower clinical response and remission rates at week 6 and week 52.63 In the CD studies, VDZ was associated with higher degree of adverse events than placebo.
Based on these studies, VDZ has received approval for marketing in CD and UC for patients not responding to one or more standard therapies (corticosteroids, immunomodulators, or tumour necrosis factor blocker medications) and is expected to arrive in the clinical practice very soon. How will VDZ be positioned in the treatment of IBD? So far no head-to-head trials are available comparing anti-TNFa to VDZ. For CD patients it is apparent from the RCT, that VDZ is not a good induction drug, because it may require a longer induction period to achieve a clinical response. Based on the pivotal trials, it will probably be reserved for CD patients with moderate to severe luminal disease not responding or loosing response to anti-TNFa agents. Its use in perianal fistulising disease and as induction first-line agent will require further studies.
In UC, a meta-analysis including 7 double-blind placebo-controlled trials, with a total of 1502 patients (720 given placebo), showed that in patients with moderate to severe active UC naive to biological therapy, VDZ has similar efficacy to the anti-TNFa antibodies infliximab, adalimumab, and golimumab for induction of response and remission and for maintenance of response and remission.64 This has however never been tested in real life. Likely VDZ will be reserved for the treatment of ulcerative colitis patients who fail one anti-TNFa, given its success in this population of patients and the low efficacy of a second anti-TNFa agent to induce a clinical response in this setting.65
Many more promising treatments are expected to arrive in the next years. Etrolizumab, which is also an anti-integrin, is currently undergoing phase III trials. Tofacitinib, an oral JAK inhibitor, ustekinumab, and anti-IL12/IL23 are also undergoing clinical trials. Hopefully some of these new drugs will reach the clinic and become part of the therapeutic arsenal in IBD.
6. Therapeutic drug monitoring – optimizing what we currently have
Loss of response (LOR) is an important issue of anti-TNFa therapy.66 Empiric escalation of the drug either by increasing the dose or shortening the interval has been used in the past to manage loss of response but it is increasingly being replaced by therapeutic drug monitoring (TDM). In a significant number of patients, loss of response is mediated through formation of anti-drug antibodies (ADA).67 Scheduled anti-TNF administration, rather than episodic, as well as association with an immunosuppressive agent, are the main strategies to reduce ADA formation.50
Commercially available assays for measuring drug metabolites and anti-drug antibodies (ADA) for infliximab (IFX) and adalimumab are now available and may be helpful in guiding treatment strategies in a more customized, economic and rational way.68,69
Among the risk factors for LOR, immunogenicity is by far the most important. Despite the well known association between ADA formation, lower trough levels and LOR, only recently has the kinetics of anti-drug antibody formation been described.70 In this study by Ungar et al, among 125 patients (98 CD, 27 UC) treated with IFX, sera were collected before each infusion and analyzed for ADA and IFX trough levels. Around 46% of patients developed ADA, and 90% did so in the first 12 months of therapy. The formation of ADA heralded LOR in 54% of patients with a median delay of 2 months. Interestingly, the median time to ADA formation was 1.5 months (range 0.5-31 months), meaning that 75% of patients by week 22 of treatment had developed ADA.
Based on ADA and trough levels most authorities recommend the following approach to patients presenting with LOR71: (1) patients with low drug levels should have dose intensification, (2) patients with high concentrations of ADA should switch to other anti-TNFa molecule, and (3) patients who present a therapeutic drug concentration without ADA should discontinue anti-TNFa therapy and switch to another class of medications or consider surgical options, after active disease is objectively confirmed. We must bear in mind some important aspects of this algorithm. In first place this strategy is mainly based on results obtained with the ELISA methodology, which is limited by the inability to measure ADA in the presence of circulating drug.72 Secondly, the optimal drug level associated with clinical response is still a matter of debate, as detection assays may vary between centres. Nevertheless trough levels for IFX of 3-7 mg/mL73 and 5-10 mg/mL74 have recently been suggested as a target for maintenance therapy for both UC and CD, respectively.75 For adalimumab 5.85 mg/mL has been suggested as an adequate trough level.76 Finally, this algorithm may not be applicable to almost one-third of patients with results outside the defined cut-off values.77
7. Therapy de-escalation
If a patient reaches sustained remission, there is an opportunity for treatment de-escalation. De-escalation strategies are increasingly being considered in clinical practice because of safety concerns (opportunistic infections and cancer78) and economic issues.79 However, proper de-escalation criteria are not yet defined. De-escalation strategies may include discontinuation of the immunosuppressant (IS), withdrawal of the anti-TNFa, or reducing the dose of one or both drugs.80
A randomized, controlled study evaluated the impact of IS discontinuation in CD patients receiving combination therapy with IFX and in remission for at least six months.81 The authors found that although patients who discontinued IS had higher C-reactive protein levels and lower IFX trough levels, there were similar proportions of patients needing a change in IFX regimen over 24 months in the two groups (60% and 55% in those who continued and interrupted IS, respectively).81
The STORI trial from the GETAID evaluated the impact of IFX discontinuation in 115 CD patients treated with IFX plus IS for more than one year and with steroid-free remission for more than six months.16 The authors found relapse rates of 44% and 52% over 1 year and 2 years after IFX discontinuation, respectively.16 In a multivariable analysis, risk factors for relapse included male gender, absence of surgical resection, leucocyte counts > 6.0×109 L−1, levels of haemoglobin≤145 g/L, C-reactive protein≥5.0 mg/l, faecal calprotectin≥300 mg/g and absence of mucosal healing (CDEIS > 0).16 Re-treatment with IFX was found to be effective and well tolerated in 88% of patients who had relapsed.16 No prospective data in UC are available so far.
Therefore, in patients receiving combination therapy, relapse rate after IS withdrawal seems lower than after anti-TNFa discontinuation, therefore the former is considered preferable by some authors.79
A step-down approach based on IS or anti-TNFa dose reduction according to disease activity has been studied in rheumatology with conflicting results.82,83 Prospective studies in IBD are needed to assess the benefit of this approach.
Finally, complete drug withdrawal would be the ultimate goal of therapy de-escalation in IBD. However, to date, treatment discontinuation in patients receiving monotherapy (with either IS or anti-TNFa) is not universally recommended because of a high clinical relapse rate (30-60% at 12 months after IS withdrawal in IBD patients under IS alone).79
In conclusion, de-escalation treatment strategies should be mainly considered in highly selected CD patients with a high risk of severe adverse events and low risk of relapse after treatment withdrawal (deep remission: including clinical remission, biomarker normalization and complete mucosal healing). In the future, the ultimate target in IBD will probably be complete remission, including also histological and molecular healing. Nonetheless the prognostic value of histological remission as a treatment target in IBD needs to be confirmed and validated histological scores are needed.84
8. Conclusions
New trends are emerging in IBD (Fig. 1), with the hope of changing natural history of disease. Among the new drugs expected to arrive in the next years, vedolizumab, etrolizumab, ustekinumab and tofacitinib appear to be the most promising.
Figure 1
A new paradigm moving towards disease modification in CD is emerging which includes early intensive (if necessary) treatment and tight disease control. Early treatment with immunosuppressants and/or biologics in patients with poor prognostic factors should be considered.
A treat-to-target strategy, based on frequent assessment of disease activity and subsequent adjustment of treatment accordingly, may modify the natural course of IBD. The use of monitoring tools such as endoscopy, cross-sectional imaging and biomarkers has not been prospectively validated and remains empirical. Mucosal healing appears to be an important treatment endpoint but there is a need for a validated universal definition and to use a standardized validated endoscopic score both in UC and CD. The use of non-invasive inflammatory biomarkers as well as blood drug levels may improve the efficacy of therapy monitoring. Further prospective studies to assess benefits and potential risks of a treat-to-target approach on long-term outcomes in IBD are needed.
Additionally, the increased use of immunomodulators, administered earlier and for longer periods, raised consideration about treatment de-escalation and safety of chronic immunosuppression. Predictors for relapse following therapy discontinuation in IBD are needed.
In conclusion, new endpoints are arising and new strategies aiming at the sustained suppression of inflammation, and involving disease activity monitoring in a treat-totarget approach are becoming increasingly incorporated in the daily clinical practice of treating IBD.
References
1. Danese S. New therapies for inflammatory bowel disease: from the bench to the bedside. Gut. 2012;61:918-32. [ Links ]
2. Ordas I, Feagan BG, Sandborn WJ. Early use of immunosuppressives or TNF antagonists for the treatment of Crohns disease: time for a change. Gut. 2011;60:1754-63. [ Links ]
3. Mandel MD, Miheller P, Mullner K, Golovics PA, Lakatos PL. Have biologics changed the natural history of Crohns disease. Dig Dis. 2014;32:351-9. [ Links ]
4. Solberg IC, Lygren I, Jahnsen J, Aadland E, Hoie O, Cvancarova M, et al. Clinical course during the first 10 years of ulcerative colitis: results from a population-based inception cohort (IBSEN Study). Scand J Gastroenterol. 2009;44:431-40. [ Links ]
5. Solberg IC, Vatn MH, Hoie O, Stray N, Sauar J, Jahnsen J, et al. Clinical course in Crohns disease: results of a Norwegian population-based ten-year follow-up study. Clin Gastroenterol Hepatol. 2007;5:1430-8. [ Links ]
6. Magro F, Rodrigues-Pinto E, Coelho R, Andrade P, Santos-Antunes J, Lopes S, et al. Is it possible to change phenotype progression in Crohns disease in the era of immunomodulators. Predictive factors of phenotype progression. Am J Gastroenterol. 2014;109:1026-36. [ Links ]
7. Bouguen G, Levesque BG, Feagan BG, Kavanaugh A, Peyrin-Biroulet L, Colombel JF, et al. Treat to target: a proposed new paradigm for the management of Crohns disease. Clin Gastroenterol Hepatol. 2013, pii: S1542-3565(13)01301-3. [ Links ]
8. Dias CC, Rodrigues PP, da Costa-Pereira A, Magro F. Clinical prognostic factors for disabling Crohns disease: a systematic review and meta-analysis. World J Gastroenterol. 2013;19:3866-71. [ Links ]
9. Beaugerie L, Seksik P, Nion-Larmurier I, Gendre JP, Cosnes J. Predictors of Crohns disease. Gastroenterology. 2006;130:650-6. [ Links ]
10. Loly C, Belaiche J, Louis E. Predictors of severe Crohns disease. Scand J Gastroenterol. 2008;43:948-54. [ Links ]
11. Yang CH, Ding J, Gao Y, Chen X, Yang ZB, Xiao SD. Risk factors that predict the requirement of aggressive therapy among Chinese patients with Crohns disease. J Dig Dis. 2011;12:99-104. [ Links ]
12. Zallot C, Peyrin-Biroulet L. Clinical risk factors for complicated disease: how reliable are they? Dig Dis. 2012;30 Suppl. 3:67-72. [ Links ]
13. Romberg-Camps MJ, Dagnelie PC, Kester AD, Hesselink-van de Kruijs MA, Cilissen M, Engels LG, et al. Influence of phenotype at diagnosis and of other potential prognostic factors on the course of inflammatory bowel disease. Am J Gastroenterol. 2009;104:371-83. [ Links ]
14. Allez M, Lemann M, Bonnet J, Cattan P, Jian R, Modigliani R. Long term outcome of patients with active Crohns disease exhibiting extensive and deep ulcerations at colonoscopy. Am J Gastroenterol. 2002;97:947-53. [ Links ]
15. Lakatos PL, Czegledi Z, Szamosi T, Banai J, David G, Zsigmond F, et al. Perianal disease, small bowel disease, smoking, prior steroid or early azathioprine/biological therapy are predictors of disease behavior change in patients with Crohns disease. World J Gastroenterol. 2009;15:3504-10. [ Links ]
16. Louis E, Mary JY, Vernier-Massouille G, Grimaud JC, Bouhnik Y, Laharie D, et al. Groupe Detudes Therapeutiques Des Affections Inflammatoires D. Maintenance of remission among patients with Crohns disease on antimetabolite therapy after infliximab therapy is stopped. Gastroenterology. 2012;142:63-70, e5; quiz e31. [ Links ]
17. Jess T, Simonsen J, Jorgensen KT, Pedersen BV, Nielsen NM, Frisch M. Decreasing risk of colorectal cancer in patients with inflammatory bowel disease over 30 years. Gastroenterology. 2012;143:375-81, e1; quiz e13-4. [ Links ]
18. Sandborn WJ, Hanauer S, Van Assche G, Panes J, Wilson S, Petersson J, et al. Treating beyond symptoms with a view to improving patient outcomes in inflammatory bowel diseases. J Crohns Colitis. 2014;8:927-35. [ Links ]
19. Hefti MM, Chessin DB, Harpaz NH, Steinhagen RM, Ullman TA. Severity of inflammation as a predictor of colectomy in patients with chronic ulcerative colitis. Dis Colon Rectum. 2009;52:193-7. [ Links ]
20. Kuriyama M, Kato J, Fujimoto T, Nasu J, Miyaike J, Morita T, et al. Risk factors and indications for colectomy in ulcerative colitis patients are different according to patients clinical background. Dis Colon Rectum. 2006;49:1307-15. [ Links ]
21. Targownik LE, Singh H, Nugent Z, Bernstein CN. The epidemiology of colectomy in ulcerative colitis: results from a population-based cohort. Am J Gastroenterol. 2012;107:1228-35. [ Links ]
22. Arnott ID, Landers CJ, Nimmo EJ, Drummond HE, Smith BK, Targan SR, et al. Sero-reactivity to microbial components in Crohns disease is associated with disease severity and progression, but not NOD2/CARD15 genotype. Am J Gastroenterol. 2004;99:2376-84. [ Links ]
23. Dubinsky MC. Serologic and laboratory markers in prediction of the disease course in inflammatory bowel disease. World J Gastroenterol. 2010;16:2604-8. [ Links ]
24. Cleynen I, Gonzalez JR, Figueroa C, Franke A, McGovern D, Bortlik M, et al. Genetic factors conferring an increased susceptibility to develop Crohns disease also influence disease phenotype: results from the IBDchip European Project. Gut. 2013;62:1556-65. [ Links ]
25. Prideaux L, De Cruz P, Ng SC, Kamm MA. Serological antibodies in inflammatory bowel disease: a systematic review. Inflamm Bowel Dis. 2012;18:1340-55. [ Links ]
26. Jung C, Colombel JF, Lemann M, Beaugerie L, Allez M, Cosnes J, et al. Genotype/phenotype analyses for 53 Crohns disease associated genetic polymorphisms. PLoS ONE. 2012;7:e52223. [ Links ]
27. Pariente B, Mary JY, Danese S, Chowers Y, De Cruz P, DHaens G, et al. Development of the Lemann index to assess digestive tract damage in patients with Crohns disease. Gastroenterology. 2015;148, 52-63.e3. [ Links ]
28. Allen PB, Peyrin-Biroulet L. Moving towards disease modification in inflammatory bowel disease therapy. Curr Opin Gastroenterol. 2013;29:397-404. [ Links ]
29. World Health Organization. International Classification of Functioning, Disability and Health (ICF). Geneva. Geneva: WHO; 2001. Available at: http://www.who.int/classifications/icf/en [ Links ]
30. Walsh A, Palmer R, Travis S. Mucosal healing as a target of therapy for colonic inflammatory bowel disease and methods to score disease activity. Gastrointest Endosc Clin N Am. 2014;24:367-78. [ Links ]
31. DHaens G, Sandborn WJ, Feagan BG, Geboes K, Hanauer SB, Irvine EJ, et al. A review of activity indices and efficacy end points for clinical trials of medical therapy in adults with ulcerative colitis. Gastroenterology. 2007;132:763-86. [ Links ]
32. Froslie KF, Jahnsen J, Moum BA, Vatn MH, Group I. Mucosal healing in inflammatory bowel disease: results from a Norwegian population-based cohort. Gastroenterology. 2007;133:412-22. [ Links ]
33. Schnitzler F, Fidder H, Ferrante M, Noman M, Arijs I, Van Assche G, et al. Mucosal healing predicts long-term outcome of maintenance therapy with infliximab in Crohns disease. Inflamm Bowel Dis. 2009;15:1295-301. [ Links ]
34. Baert F, Moortgat L, Van Assche G, Caenepeel P, Vergauwe P, De Vos M, et al. Belgian Inflammatory Bowel Disease Research G North-Holland Gut C. Mucosal healing predicts sustained clinical remission in patients with early-stage Crohns disease. Gastroenterology. 2010;138:463-8, quiz e10-1. [ Links ]
35. Hanauer SB, Feagan BG, Lichtenstein GR, Mayer LF, Schreiber S, Colombel JF, et al. Maintenance infliximab for Crohns disease: the ACCENT I randomised trial. Lancet. 2002;359:1541-9. [ Links ]
36. Benitez JM, Meuwis MA, Reenaers C, Van Kemseke C, Meunier P, Louis E. Role of endoscopy, cross-sectional imaging and biomarkers in Crohns disease monitoring. Gut. 2013;62:1806-16. [ Links ]
37. Annese V, Daperno M, Rutter MD, Amiot A, Bossuyt P, East J, et al. European evidence based consensus for endoscopy in inflammatory bowel disease. J Crohns Colitis. 2013;7:982-1018. [ Links ]
38. Rimola J, Ordas I, Rodriguez S, Garcia-Bosch O, Aceituno M, Llach J, et al. Magnetic resonance imaging for evaluation of Crohns disease: validation of parameters of severity and quantitative index of activity. Inflamm Bowel Dis. 2011;17:1759-68. [ Links ]
39. Panes J, Bouzas R, Chaparro M, Garcia-Sanchez V, Gisbert JP, Martinez de Guerenu B, et al. Systematic review: the use of ultrasonography, computed tomography and magnetic resonance imaging for the diagnosis, assessment of activity and abdominal complications of Crohns disease. Aliment Pharmacol Ther. 2011;34:125-45. [ Links ]
40. Ordas I, Rimola J, Rodriguez S, Paredes JM, Martinez-Perez MJ, Blanc E, et al. Accuracy of magnetic resonance enterography in assessing response to therapy and mucosal healing in patients with Crohns disease. Gastroenterology. 2014;146:374-82, e1. [ Links ]
41. Lewis JD. The utility of biomarkers in the diagnosis and therapy of inflammatory bowel disease. Gastroenterology. 2011;140:1817-26, e2. [ Links ]
42. Denis MA, Reenaers C, Fontaine F, Belaiche J, Louis E. Assessment of endoscopic activity index and biological inflammatory markers in clinically active Crohns disease with normal C-reactive protein serum level. Inflamm Bowel Dis. 2007;13:1100-5. [ Links ]
43. Jürgens M, Mahachie John JM, Cleynen I, Schnitzler F, Fidder H, van Moerkercke W, et al. Levels of C-reactive protein are associated with response to infliximab therapy in patients with Crohns disease. Clin Gastroenterol Hepatol. 2011;9:421-70. [ Links ]
44. De Cruz P, Kamm MA, Hamilton AL, Ritchie KJ, Krejany EO, Gorelik A, et al. Crohns disease management after intestinal resection: a randomised trial. Lancet. 2015;385:1406-17. [ Links ]
45. Wright EK, Kamm MA, De Cruz P, Hamilton AL, Ritchie KJ, Krejany EO, et al. Measurement of fecal calprotectin improves monitoring and detection of recurrence of Crohns disease following surgery. Gastroenterology. 2015;148, 938-947.e1. [ Links ]
46. Bondjemah V, Mary JY, Jones J, Sandborn W, Schoepfer A, Louis E, et al. P133 fecal calprotectin and CRP as biomarkers of endoscopic activity in Crohns disease: a meta-study. J Crohns Colitis. 2012;6:S63. [ Links ]
47. DHaens G, Ferrante M, Vermeire S, Baert F, Noman M, Moortgat L, et al. Fecal calprotectin is a surrogate marker for endoscopic lesions in inflammatory bowel disease. Inflamm Bowel Dis. 2012;18:2218-24. [ Links ]
48. Lemann M, Colombel J, Grimaud J, Bouhnik Y, Laharie D, Dupas J. Fecal calprotectin and high sensitivity C-reactive protein levels to predict mucosal healing in patients with Crohns disease. A subanalysis of the STORI study. Gut. 2010;59:A80. [ Links ]
49. Rutgeerts P, Van Assche G, Sandborn WJ, Wolf DC, Geboes K, Colombel JF, et al. Adalimumab induces and maintains mucosal healing in patients with Crohns disease: data from the EXTEND trial. Gastroenterology. 2012;142:1102-11, e2. [ Links ]
50. Colombel JF, Sandborn WJ, Reinisch W, Mantzaris GJ, Kornbluth A, Rachmilewitz D, et al., Group SS. Infliximab, azathioprine, or combination therapy for Crohns disease. N Engl J Med. 2010;362:1383-95. [ Links ]
51. Mantzaris GJ, Christidou A, Sfakianakis M, Roussos A, Koilakou S, Petraki K, et al. Azathioprine is superior to budesonide in achieving and maintaining mucosal healing and histologic remission in steroid-dependent Crohns disease. Inflamm Bowel Dis. 2009;15:375-82. [ Links ]
52. Rutgeerts P, Feagan BG, Lichtenstein GR, Mayer LF, Schreiber S, Colombel JF, et al. Comparison of scheduled and episodic treatment strategies of infliximab in Crohns disease. Gastroenterology. 2004;126:402-13. [ Links ]
53. Ferrante M, Colombel JÄ, Sandborn WJ, Reinisch W, Mantzaris GJ, Kornbluth A, et al. Validation of endoscopic activity scores in patients with Crohns disease based on a post hoc analysis of data from SONIC. Gastroenterology. 2013;145:978-86, e5. [ Links ]
54. Cominelli F. Inhibition of leukocyte trafficking in inflammatory bowel disease. N Engl J Med. 2013;369:775-6. [ Links ]
55. Gilroy L, Allen PB. Is there a role for vedolizumab in the treatment of ulcerative colitis and Crohns disease. Clin Exp Gastroenterol. 2014;7:163-72. [ Links ]
56. Sandborn WJ, Colombel JF, Enns R, Feagan BG, Hanauer SB, Lawrance IC, et al., International Efficacy of Natalizumab as Active Crohns Therapy Trial G, Evaluation of Natalizumab as Continuous Therapy Trial G. Natalizumab induction and maintenance therapy for Crohns disease. N Engl J Med. 2005;353:1912-25. [ Links ]
57. Targan SR, Feagan BG, Fedorak RN, Lashner BA, Panaccione R, Present DH, et al., International Efficacy of Natalizumab in Crohns Disease R, Remission Trial G. Natalizumab for the treatment of active Crohns disease: results of the ENCORE Trial. Gastroenterology. 2007;132:1672-83. [ Links ]
58. Bloomgren G, Richman S, Hotermans C, Subramanyam M, Goelz S, Natarajan A, et al. Risk of natalizumab-associated progressive multifocal leukoencephalopathy. N Engl J Med. 2012;366:1870-80. [ Links ]
59. Wuthrich C, Popescu BF, Gheuens S, Marvi M, Ziman R, Denq SP, et al. Natalizumab-associated progressive multifocal leukoencephalopathy in a patient with multiple sclerosis: a post-mortem study. J Neuropathol Exp Neurol. 2013;72:1043-51. [ Links ]
60. Parikh A, Wyant T, Clifford DB, Berger JR, Feagan BG, Fox I, et al. 1008 No increase in JC viremia lymphocyte count, or circulating CD34+ hematopoietic progenitor cells after treatment with vedolizumab, a humanized monoclonal antibody to a4b7 integrin. Gastroenterology. 2010;138. S-145-S-146. [ Links ]
61. DHaens G, Vermeire S, Cataldi F, Vogelsang H, Allez M, Desreumaux P, et al. OP007 Anti-MAdCAM monoclonal antibody PF-00547659 does not affect immune surveillance in the central nervous system of anti-TNF and immunosuppressant experienced Crohns disease patients who are anti-TNF inadequate responders: results from the TOSCA study. J Crohns Colitis. 2014;8:S4-5. [ Links ]
62. Feagan BG, Rutgeerts P, Sands BE, Hanauer S, Colombel JF, Sandborn WJ, et al. Vedolizumab as induction and maintenance therapy for ulcerative colitis. N Engl J Med. 2013;369:699-710. [ Links ]
63. Sandborn WJ, Feagan BG, Rutgeerts P, Hanauer S, Colombel JF, Sands BE, et al. Vedolizumab as induction and maintenance therapy for Crohns disease. N Engl J Med. 2013;369:711-21. [ Links ]
64. Danese S, Fiorino G, Peyrin-Biroulet L, Lucenteforte E, Virgili G, Moja L, et al. Biological agents for moderately to severely active ulcerative colitis: a systematic review and network metaanalysis. Ann Intern Med. 2014;160:704-11. [ Links ]
65. Sandborn WJ, van Assche G, Reinisch W, Colombel JF, DHaens G, Wolf DC, et al. Adalimumab induces and maintains clinical remission in patients with moderate-to-severe ulcerative colitis. Gastroenterology. 2012;142:257-65, e1-3. [ Links ]
66. Gisbert JP, Panes J. Loss of response and requirement of infliximab dose intensification in Crohns disease: a review. Am J Gastroenterol. 2009;104:760-7. [ Links ]
67. Baert F, Noman M, Vermeire S, Van Assche GGDH, Carbonez A, Rutgeerts P. Influence of immunogenicity on the longterm efficacy of infliximab in Crohns disease. N Engl J Med. 2003;348:601-8. [ Links ]
68. Steenholdt C, Brynskov J, Thomsen OO, Munck LK, Fallingborg J, Christensen LA, et al. Individualised therapy is more cost-effective than dose intensification in patients with Crohns disease who lose response to anti-TNF treatment: a randomised, controlled trial. Gut. 2014;63:919-27. [ Links ]
69. Papamichael K, Gils A, Rutgeerts P, Levesque BG, Vermeire S, Sandborn WJ, et al. Role for therapeutic drug monitoring during induction therapy with TNF antagonists in IBD: evolution in the definition and management of primary nonresponse. Inflamm Bowel Dis. 2015;21:182-97. [ Links ]
70. Ungar B, Chowers Y, Yavzori M, Picard O, Fudim E, Har-Noy O, et al. ABIRISK Consortium. The temporal evolution of antidrug antibodies in patients with inflammatory bowel disease treated with infliximab. Gut. 2014;63:1258-64. [ Links ]
71. Sandborn WJ. Tailoring treatment to the individual patient: drug monitoring. Dig Dis. 2014;32 Suppl. 1:88-91. [ Links ]
72. Ordas I, Feagan BG, Sandborn WJ. Therapeutic drug monitoring of tumor necrosis factor antagonists in inflammatory bowel disease. Clin Gastroenterol Hepatol. 2012;10:1079-87, quiz e85-6.
73. Vande Casteele N, Compernolle G, Ballet V, Van Assche G, Gils A, Vermeire S, et al. OP11 Individualised infliximab treatment using therapeutic drug monitoring: a prospective controlled Trough level Adapted infliXImab Treatment (TAXIT) trial. J Crohns Colitis. 2012;6 Supplement 1:S6. [ Links ]
74. Vaughn BP, Martínez-Vazquez M, Patwardhan V, Moss AC, Sandborn WJ, Cheifetz AS. 209 prospective therapeutic drug monitoring to optimizing infliximab (IFX) maintenance therapy in patients with inflammatory bowel disease (IBD). Gastroenterology. 2014;146. S-54. [ Links ]
75. Kopylov U, Ben-Horin S, Seidman E. Therapeutic drug monitoring in inflammatory bowel disease. Ann Gastroenterol. 2014;27:304-12. [ Links ]
76. Mazor Y, Koplov U, Ben Hur D, Almog R, Waterman M, Ben-Horin S, et al. P517 evaluating adalimumab drug and antibody levels as predictors of clinical and laboratory response in Crohns disease patients. J Crohns Colitis. 2013;7:S217. [ Links ]
77. Yanai H, Lichtenstein L, Assa A, Mazor Y, Weiss B, Levine A, et al. Levels of drug and antidrug antibodies are associated with outcome of interventions after loss of response to infliximab or adalimumab. Clin Gastroenterol Hepatol. 2015;13, 522-530.e2. [ Links ]
78. Pinto Pais T, Fernandes S, Carvalho J. Squamous cell carcinoma in enterocutaneous fistula associated with Crohns disease: first case report. J Crohns Colitis. 2014;8:1142-3. [ Links ]
79. Pariente B, Laharie D. Review article: why, when and how to de-escalate therapy in inflammatory bowel diseases. Aliment Pharmacol Ther. 2014;40:338-53. [ Links ]
80. Cohen BL, Torres J, Colombel JF. Immunosuppression in inflammatory bowel disease: how much is too much. Curr Opin Gastroenterol. 2012;28:341-8. [ Links ]
81. Van Assche G, Magdelaine-Beuzelin C, DHaens G, Baert F, Noman M, Vermeire S, et al. Withdrawal of immunosuppression in Crohns disease treated with scheduled infliximab maintenance: a randomized trial. Gastroenterology. 2008;134:1861-8. [ Links ]
82. van den Bemt BJ, den Broeder AA, Snijders GF, Hekster YA, van Riel PL, Benraad B, et al. Sustained effect after lowering high-dose infliximab in patients with rheumatoid arthritis: a prospective dose titration study. Ann Rheum Dis. 2008;67:1697-701. [ Links ]
83. Maini RN, Breedveld FC, Kalden JR, Smolen JS, Davis D, Macfarlane JD, et al. Therapeutic efficacy of multiple intravenous infusions of anti-tumor necrosis factor alpha monoclonal antibody combined with low-dose weekly methotrexate in rheumatoid arthritis. Arthritis Rheum. 1998;41:1552-63. [ Links ]
84. Bryant RV, Winer S, Travis SP, Riddell RH. Systematic review: histological remission in inflammatory bowel disease. Is complete remission the new treatment paradigm? An IOIBD initiative. J Crohns Colitis. 2014;8:1582-97. [ Links ]
* Corresponding author.
E-mail address: marilia.cravo@sapo.pt (M. Cravo).
Conflicts of interests
The authors have no conflicts of interest to declare.
Received 17 January 2015; accepted 12 March 2015














