Serviços Personalizados
Journal
Artigo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Acessos
Acessos
Links relacionados
-
 Similares em
SciELO
Similares em
SciELO
Compartilhar
GE-Portuguese Journal of Gastroenterology
versão impressa ISSN 2341-4545
GE Port J Gastroenterol vol.23 no.5 Lisboa out. 2016
CLINICAL CASE
Treating Gastric Antral Vascular Ectasia – When Argon Therapy Is Not Enough
Tratamento da Ectasia Vascular do Antro Gástrico – Quando a Terapêutica com Árgon É Insuficiente
Francisca Dias de Castro a, * , Pedro Boal Carvalho a, Tiago Cúrdia Gonçalves a, Joana Magalhães a, Maria João Moreira a, Carla Marinho a, José Cotter a, b, c
a Gastroenterology Department, Hospital da Senhora da Oliveira, Centro Hospitalar do Alto Ave, Guimarães, Portugal
b Life and Health Sciences Research Institute (ICVS), School of Health Sciences, University of Minho, Braga, Portugal
c ICVS/3B's, PT Government Associate Laboratory, Guimarães, Portugal
* Corresponding author.
ABSTRACT
Gastric antral vascular ectasia (GAVE) is a capillary-type vascular malformation of the gastric antrum and an infrequent cause of chronic gastrointestinal blood loss and iron deficiency anemia.
The authors describe a case report of GAVE in a female cirrhotic patient presenting with severe symptomatic iron deficiency anemia. After failure of argon plasma coagulation (APC), the patient was treated with endoscopic band ligation (EBL) with resolution of anemia, without new episodes of rebleeding and no need for further hospitalizations or transfusion requirements.
Even though APC is the current treatment of choice for GAVE recurrence-free survival at one year is achieved in less than 50% of the patients and failed therapy has been described in up to 14% of the patients. EBL has been reported to be a relatively easy technique for GAVE therapy and has been shown to be safe and effective with lower complication rates in comparison with APC. This technique may in the future be used as the initial endoscopic treatment to eradicate GAVE.
Keywords: Endoscopy Gastrointestinal. Gastric Antral Vascular Ectasia. Argon Plasma Coagulation. Ligation.
RESUMO
A ectasia vascular do antro gástrico (GAVE) é uma malformação vascular e uma causa infrequente de anemia por défice de ferro. Os autores descrevem um caso de doente do sexo feminino com antecedentes de cirrose hepática alcoólica com diagnóstico de GAVE após estudo etiológico de anemia ferropénica sintomática. Após falência do tratamento endoscópico com árgon plasma (APC), a doente foi submetida a laqueação elástica da GAVE com resolução da anemia, sem novos episódios de hemorragia e sem necessidade de re-internamentos ou suporte transfusional.
Apesar do APC ser o tratamento de primeira linha para a GAVE a taxa de doentes sem recorrência em um ano é alcançada em menos de 50% dos doentes, para além de falências primárias ao tratamento rondarem os 14%. A laqueação elástica tem sido descrita como uma técnica de fácil aplicação no tratamento da GAVE e tem-se revelado segura, eficaz e com baixa taxa de complicações em comparação com o APC. Esta técnica pode, no futuro, ser utilizada como tratamento endoscópico inicial para a erradicação da GAVE.
Palavras-chave: Endoscopia Gastrointestinal. Ectasia Vascular do Antro Gástrico. Coagulação Àrgon plasma. Laqueação.
1. Introduction
Gastric antral vascular ectasia (GAVE) is a capillary-type vascular malformation of the gastric antrum1 and an infrequent cause of chronic gastrointestinal blood loss and iron deficiency anemia. GAVE is responsible for up to 4% of nonvariceal upper gastrointestinal (GI) bleeding and 6% of upper GI bleeding in cirrhotic patients.2,3
Most cases are idiopathic, but it is frequently associated with other clinical conditions, such as cirrhosis and autoimmune diseases,4 with a prevalence reported in cirrhotic patients of 12%3 and reaching 30% in patients with liver failure.5
At oesophagogastroduodenoscopy (OGD), GAVE may appear as multiple longitudinal streaks that converge at the pyloric orifice, with a “watermelon” pattern (stripe type), or as multiple erythematous spots (diffuse or granular type).6 This last form occurring more frequently in patients with cirrhosis.6
The current treatment of choice for GAVE is endoscopic intervention with argon plasma coagulation (APC).7 However recurrence-free survival at one year is achieved in less than 50% of the patients8 and in addition rates of failed therapy of up to 14% have been reported.3 Equally important to refer is the complication rate which is high (20–30%).3
The authors describe a case report of GAVE in a female cirrhotic patient presenting with severe symptomatic iron deficiency anemia. After failure of APC, the patient was treated with endoscopic band ligation (EBL) with resolution of anemia, without new episodes of rebleeding and no need for further hospitalizations or transfusion requirements.
2. Case presentation
A 69-years-old female presented with a three-week history of fatigue. She denied hematemesis and abdominal pain, and there was no change in appetite, weight, bowel pattern, or stool color. She had a medical history of alcoholic cirrhosis (Child Pugh A, MELD 7 points) and was on furosemide, had no known allergies and no family history of gastrointestinal pathology. She denied consumption of nonsteroidal anti-inflammatory and smoking habits.
Physical examination revealed paleness of the skin and mucosae and sinus tachycardia with normal blood pressure. Rectal examination did not reveal blood in stools.
Laboratory data evidenced severe anemia with hemoglobin level of 4.0 g/dL, low platelet count, normal coagulation parameters and inflammatory markers were normal. Iron study revealed a serum iron level of 14 μg/dL (reference range 50–170 μg/dL) with a total iron binding capacity of 398 μg/dL (reference range 250–450 μg/dL) and ferritin of 2.8 ng/mL (reference range 8–252 ng/mL). Transfusion of red blood cells was initiated with hemoglobin level increasing to 7.7 g/dL.
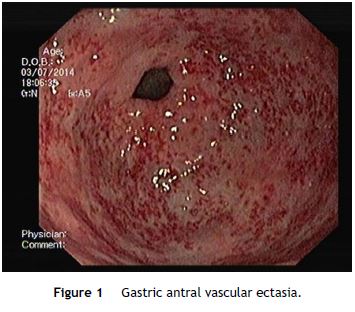
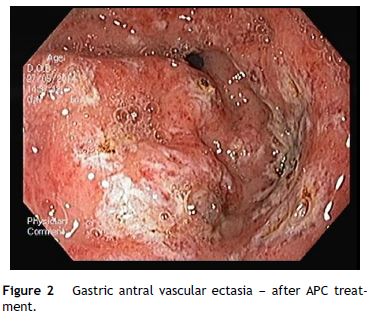
An OGD was performed and revealed multiple erythematous spots in the antrum compatible with GAVE, diffuse type (Fig. 1), and mild portal hypertensive gastropathy, without active bleeding. There was no evidence of esophageal or gastric varices. GAVE was treated with APC without complications (Fig. 2). A colonoscopy with ileoscopy was also performed, but it was negative for blood or hemorrhagic lesions. Her hemoglobin levels remained stable and the patient was discharged and started on propranolol (for portal hypertensive gastropathy), with a target of 55 beats per minute, 40 mg of pantoprazole every 24 h and performed iron intravenous supplementation in ambulatory.
During the following 4 months, the patient had 3 new episodes of severe symptomatic anemia requiring transfusion. During these episodes she underwent OGD that revealed persistence of GAVE and APC treatments were performed.
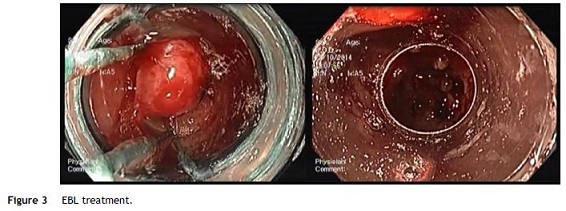
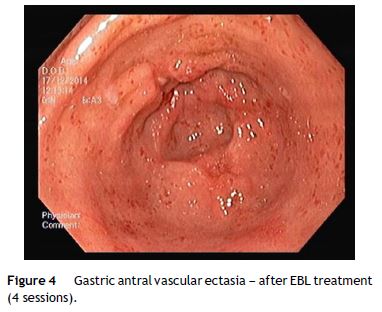
Nevertheless the patient presented with a new episode of symptomatic anemia, and an hemoglobin level of 3.7 g/dL was observed. After transfusion of red blood cells an OGD was performed. At that time, since GAVE persisted despite treatment sessions using APC, and taking into consideration the severity and short recurrence intervals of symptomatic anemia, a decision was made to perform EBL (multi-band ligator, Ezy Shoot, G-Flex®). Six bands were applied, starting from lesions adjacent to the pylorus, then continued proximally in the antrum until most of the GAVE affected areas were treated (Fig. 3). No complications of the procedure were reported, and the patient was discharged after 24 h, maintaining propranolol and pantoprazole. Endoscopic evaluation was performed every 4 weeks and another 3 sessions of EBL were performed with almost complete eradication of GAVE (Fig. 4). Following the first EBL treatment the patient remained asymptomatic with stable hemoglobin level of 12 g/dL and no need of red cells blood transfusion or iron supplementation.
3. Discussion
GAVE is a poorly understood entity, of unknown etiology, and an increasingly identifiable cause of chronic iron deficiency anemia. The pharmacological management of GAVE has been met with disappoint results, and the mainstay of GAVE management remains endoscopic therapy.3,9
APC is a modality of non-contact electrocoagulation that applies high-frequency energy into tissue to cause thermal effects, which can be used for hemostasis.10 APC has been used successfully to treat GAVE with an effective transient response, but primary failure rates of therapy of up to 14% have been reported.3 Some authors11–13 suggest that this endoscopic modality is insufficient in order to achieve medium and long-term treatment success, as it has been associated with a high recurrence rate (40–100%), with less than 50% of the patients achieving a recurrence-free survival at one year.8 In addition to this limitation, APC results in complications, such as sepsis, pyloric stenosis and gastric outlet obstruction syndrome, in 20–33% of the patients.3
EBL has been reported to be a relatively easy technique for GAVE therapy, has been shown to be safe and effective with lower complication rates in comparison with APC.14,15 A recent prospective study showed endoscopic improvement with the use of EBL in 91% of the patients, associated with a significant improvement in the hemoglobin and ferritin levels. EBL in GAVE has been associated with transient abdominal pain in a minority of the patients but no major complications have been reported in the literature.14
Another new option therapy for GAVE is radiofrequency ablation, which allows larger mucosal surfaces to be treated. A recent study16 suggests that radiofrequency ablation is a safe and effective treatment for recurrent bleeding from GAVE. However this technique requires additional training, is more expensive than other options and is not widely available.16 Potential complications are the same as for APC and includes perforation and bleeding.16
In this case, the patient was initially treated with APC after being diagnosed with GAVE as the cause of severe iron deficiency anemia, however this strategy was not successful. APC treatment might not be effective in some cases and this could be explained by the limited depth of thermal injury.10
Even tough portal hypertensive gastropathy was mild and probably not the cause of anemia, because of the severe presentation without active bleeding, the patient started on propranolol which is the first line treatment to reduce portal pressure.9
EBL is widely employed as an effective treatment for esophageal varices because of its capacity to obliterate submucosal varices. With respect to GAVE, since the histological changes are present in the mucosa and submucosal layer, EBL may be more effective because of its ability to obliterate the submucosal vascular plexus.10 In this patient, the presence of extensive areas of the antrum affected with a high-density of both mucosal and submucosal vascular malformations is a likely explanation for the primary failure of the APC treatment. Given the fast recurrence of severe anemia, often within 4 weeks, the decision to perform EBL yielded positive results, with eradication of GAVE with 4 sessions of EBL with no complications, namely abdominal pain.
Even though our case report is in accordance with recent literature some issues still need to be elucidated in order to improve the treatment of GAVE with EBL. The optimal interval between treatment sessions, the potential role of combined therapy with APC in patients with poor response and the need of endoscopic surveillance for recurrence after eradication since the etiology of GAVE are still unknown.
Some studies14,15 suggest that the number of sessions required for GAVE eradication is inferior when using EBL compared to APC, resulting in inferior health care costs for that group of patients. However, randomized controlled trials are lacking to determine whether EBL is more cost effective than APC as the primary endoscopic therapy for GAVE.
In summary, GAVE is a rare cause of chronic gastrointestinal blood loss and iron deficiency anemia, often associated with chronic diseases such as cirrhosis. We report an unusual case of GAVE with severe symptomatic anemia, with no response to primary endoscopic therapy with APC despite correct intravenous iron supplementation between endoscopic treatments. After initiating GAVE eradication with EBL the patient became asymptomatic. EBL may in the future be used as the initial endoscopic treatment to eradicate GAVE, but larger studies are needed to clarify this issue.
References
1. Regula J, Wronska E, Pachlewski J. Vascular lesions of the gastrointestinal tract. Best Pract Res Clin Gastroenterol. 2008;22:313-28. [ Links ]
2. Dulai G.S, Jensen DM, Kovacs TO, Gralnek IM, Jutabha R. Endoscopic treatment outcomes in watermelon stomach patients with and without portal hypertension. Endoscopy. 2004;36:68-72. [ Links ]
3. Swanson E, Mahgoub A, MacDonald R, Shaukat A. Medical and endoscopic therapies for angiodysplasia and gastric antral vascular ectasia: a systematic review. Clin Gastroenterol Hepatol. 2014;12:571-82. [ Links ]
4. Kar P, Mitra S, Resnick JM, Torbey CF. Gastric antral vascular ectasia: case report and review of the literature. Clin Med Res. 2013;11:80-5. [ Links ]
5. Ward E.M, Raimondo M, Rosser BG, Wallace MB, Dickson RD. Prevalence and natural history of gastric antral vascular ectasia in patients undergoing orthotopic liver transplantation. J Clin Gastroenterol. 2004;38:898-900. [ Links ]
6. Ito M, Uchida Y, Kamano S, Kawabata H, Nishioka M. Clinical comparisons between two subsets of gastric antral vascular ectasia. Gastrointest Endosc. 2001;53:764-70. [ Links ]
7. Naidu H, Huang Q, Mashimo H. Gastric antral vascular ectasia: the evolution of therapeutic modalities. Endosc Int Open. 2014;2:E67-73. [ Links ]
8. Nakamura SMA, Konishi H, Oi I, Shiratori K, Suzuki S. Long-term follow up of gastric antral vascular ectasia treated by argon plasma coagulation. Dig Endosc. 2006;18:128-33. [ Links ]
9. Patwardhan VR, Cardenas A. Review article: the management of portal hypertensive gastropathy and gastric antral vascular ectasia in cirrhosis. Aliment Pharmacol Ther. 2014;40:354-62. [ Links ]
10. Sato T, Yamazaki K, Akaike J. Endoscopic band ligation versus argon plasma coagulation for gastric antral vascular ectasia associated with liver diseases. Dig Endosc. 2012;24:237-42. [ Links ]
11. Roman S, Saurin JC, Dumortier J, Perreira A, Bernard G, Ponchon T. Tolerance and efficacy of argon plasma coagulation for controlling bleeding in patients with typical and atypical manifestations of watermelon stomach. Endoscopy. 2003;35:1024-8. [ Links ]
12. Yusoff I, Brennan F, Ormonde D, Laurence B. Argon plasma coagulation for treatment of watermelon stomach. Endoscopy. 2002;34:407-10. [ Links ]
13. Chaves DM, Sakai P, Oliveira CV, Cheng S, Ishioka S. Watermelon stomach: clinical aspects and treatment with argon plasma coagulation. Arq Gastroenterol. 2006;43:191-5. [ Links ]
14. Zepeda-Gomez S, Sultanian R, Teshima C, Sandha G, Van S, Montano-Loza AJ. Gastric antral vascular ectasia: a prospective study of treatment with endoscopic band ligation. Endoscopy. 2015;47:538-40. [ Links ]
15. Keohane J, Berro W, Harewood GC, Murray FE, Patchett SE. Band ligation of gastric antral vascular ectasia is a safe and effective endoscopic treatment. Dig Endosc. 2013;25:392-6. [ Links ]
16. Dray X, Repici A, Gonzalez P, Fristrup C, Lecleire S, Kantsevoy S, et al. Radiofrequency ablation for the treatment of gastric antral vascular ectasia. Endoscopy. 2014;46:963-9. [ Links ]
Ethical disclosures
Protection of human and animal subjects.
The authors declare that no experiments were performed on humans or animals for this study.
Confidentiality of data. The authors declare that they have followed the protocols of their work center on the publication of patient data.
Right to privacy and informed consent. The authors declare that no patient data appear in this article.
Conflicts of interest
The authors have no conflicts of interest to declare.
* Corresponding author.
E-mail address: franciscacastro@chaa.min-saude.pt (F. Dias de Castro).
Received 4 December, 2015; accepted 3 January 2016














