Serviços Personalizados
Journal
Artigo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Acessos
Acessos
Links relacionados
-
 Similares em
SciELO
Similares em
SciELO
Compartilhar
GE-Portuguese Journal of Gastroenterology
versão impressa ISSN 2341-4545
GE Port J Gastroenterol vol.24 no.1 Lisboa fev. 2017
https://doi.org/10.1159/000450878
CLINICAL CASE STUDIES
Acute Liver Failure due to Trazodone and Diazepam
Insuficiência Hepática Aguda Induzida pela Trazodona e Diazepam
Sofia Carvalhanaa, Ana Oliveirab, Pedro Ferreirab, Margarida Resendeb, Rui Perdigotoc, Eduardo Barrosoc
a Gastroenterology and Hepatology Department, Hospital de Santa Maria, Centro Hospitalar de Lisboa Norte, and b Intensive Care Unit and c Transplantation Unit, Hospital Curry Cabral, Centro Hospitalar de Lisboa Central, Lisbon, Portugal
* Corresponding author.
ABSTRACT
Most antidepressant agents have the potential to cause liver injury, even at therapeutic doses. Nevertheless, drug-induced liver injury (DILI) from antidepressant agents is a rare event. There is no way to prevent idiopathic DILI, but the severity of the reaction may be minimized with prompt recognition and early withdrawal of the agent. We describe a rare case of a 63-year-old man presenting with acute liver failure after 3 months of trazodone and diazepam administration at normal therapeutic doses, requiring liver transplantation. This report should increase physicians awareness of this complication and call attention to the regular monitoring of liver tests in patients taking trazodone, in order to prevent life-threatening complications.
Keywords: Antidepressive agents; Diazepam; Liver failure, acute/chemically induced; Trazodone
RESUMO
A maioria dos antidepressivos tem o potencial de causar lesão hepática, mesmo em doses terapêuticas. Contudo, a hepatite tóxica por antidepressivos é um evento raro. Não existe forma de prevenir a hepatite tóxica, mas a sua gravidade pode ser minimizada com o diagnóstico precoce e a retirada antecipada do fármaco. Apresentamos um caso raro de um homem de 63 anos com insuficiência hepática aguda após 3 meses de terapêutica com trazodona e diazepam em doses terapêuticas, com necessidade de transplante hepático. Com este caso pretende-se alertar os clínicos para a possibilidade desta entidade e da necessidade de monitorização regular das provas hepáticas em doentes sob tratamento com trazodona, de forma a prevenir morbilidade e mortalidade.
Palavras-Chave: Antidepressivos; Diazepam; Insuficiência hepática aguda/induzida quimicamente; Trazodona
Introduction
Trazodone is a second-generation, nontricyclic antidepressant that has been linked to rare cases of hepatic injury. The onset of injury is usually several months after continuous ingestion and the presentation pattern is commonly one of hepatocellular serum enzyme elevation, although cases with a shorter latency and with a mixed or cholestatic form have also been described [1]. There is no way to prevent idiopathic drug-induced liver injury (DILI), but the severity of the reaction may be minimized with prompt recognition and early withdrawal of the agent. We describe a rare case of acute liver failure (ALF) after 3 months of trazodone and diazepam administration at normal therapeutic doses, requiring liver transplantation.
Clinical Case
A 57-year-old man with no previous medical history was started on diazepam (5 mg/day) and trazodone (200 mg/day) for depression. Three months later, he developed increased jaundice and was admitted. Laboratory test results included marked elevations in serum aminotransferase levels (ALT 4,638 U/L, AST 2,745 U/L), with increases in GGT (603 U/L), alkaline phosphatase (122 U/L), bilirubin levels (12.7 mg/dL), and international normalized ratio (1.84). His liver enzymes were known to be normal in the past. All medications were stopped. Over the next week, he developed encephalopathy, his international normalized ratio (2.37) and bilirubin (25.2 mg/dL) increased, and his factor V fell (from 51 to 29%). He was transferred to a liver transplant center and was listed for urgent liver transplantation. Test results for hepatitis A, B, and C were negative, as were serologic tests for acute cytomegalovirus, herpes simplex, and Epstein-Barr virus infection. Iron and copper studies were normal and autoantibodies were negative. An abdominal ultrasound ruled out biliary obstruction and occlusion of the liver vessels. A liver biopsy showed confluent necrosis involving most of the liver parenchyma, intrahepatic cholestasis, modest inflammation, and evidence of feathery degeneration and ballooning of hepatocytes in parenchyma, but no fat or fibrosis, compatible with toxic ALF (Fig. 1). In order to evaluate the causality between liver manifestation and drug treatment, the Council for International Organizations of Medical Sciences/Roussel Uclaf Causality Assessment Method (CIOMS/RUCAM) scale has been used. The CIOMS/RUCAM scale indicated a probable relationship for trazodone and a possible relationship for diazepam in causing hepatotoxicity. While the patient was waiting for liver transplantation, encephalopathy worsened requiring mechanical ventilation. He underwent liver transplantation 2 weeks after medication stoppage and he fully recovered.
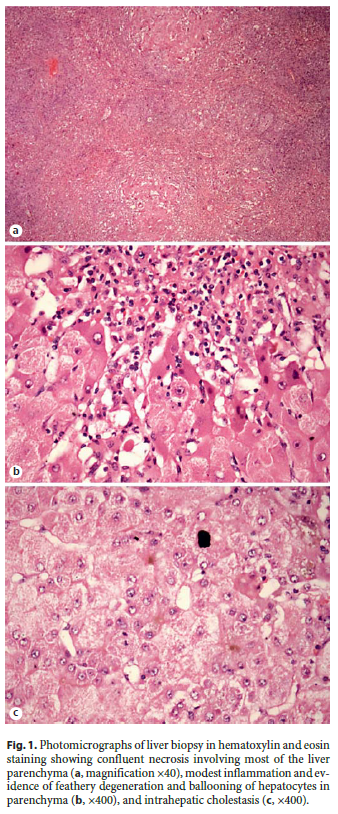
Discussion
Trazodone- and diazepam-induced ALF diagnosiswas supported by (1) the development of hepatocellular liver injury 3 months after the introduction of trazodone and diazepam, (2) hepatotoxicity as a known adverse side effect of each of the drugs, (3) liver biopsy findings, especially confluent necrosis with inflammatory activity, which is the most common pattern seen in idiosyncratic adverse drug reactions complicated with ALF [2], and (4) exclusion of other etiologies of liver injury. Furthermore, when we assessed the causality by the CIOMS/RUCAM scale for trazodone, the score was in the category of possible cause of liver injury.
DILI is a well-recognized problem that accounts for up to 10% of all adverse drug reactions. Two main mechanisms of DILI have been proposed: predictable injury (intrinsic hepatotoxins) and unpredictable injury (idiosyncratic reactions). In our case, an idiosyncratic reaction is likely to be the case. A literature review revealed that both drugs have been known to be implicated in DILI. Trazodone may cause alteration of liver enzyme levels at normal doses, but elevations are usually modest and usually do not require dose modification or discontinuation. Rare instances of ALF and death from trazodone have been described. The onset of liver toxicity may occur a few days or months after drug ingestion and even after the drug has been stopped. The pattern of liver injury is usually hepatocellular, but mixed or cholestatic forms have also been described. Contrarily, increases in liver enzymes during therapy with diazepam are rare and significant hepatotoxicity is uncommon. Only a few cases have been reported and none developed ALF [3]. The onset of injury has ranged from 1 to 6 months, and the pattern of serum enzyme elevations has typically been cholestatic or mixed [3]. The mechanism by which trazodone causes liver damage is unknown. Liver injury from benzodiazepines is probably due to a rarely produced intermediate metabolite.
This case is of special interest for its rarity. To our knowledge, only two other ALF cases associated with trazodone have been reported. Hull et al. [4] reported a 72-year-old woman who developed ALT elevations (107 U/L) 10 weeks after starting trazodone, trifluoperazine, and lithium, developing jaundice at 18 weeks and dying 2 months later. Postmortem liver biopsy revealed acute hepatic necrosis with cholestasis and portal inflammation [4]. The second case occurred in a 48-year-old woman who developed fulminant liver failure after 4 months of trazodone and venlafaxine therapy, requiring liver transplantation [5].
This report should increase physicians awareness of this complication and call attention to the regular monitoring of liver tests in patients taking trazodone, especially if taken in concomitant therapy such as with other antidepressants or antipsychotics, in order to prevent lifethreatening complications.
References
1 Fernandes NF, Martin RR, Schenker S: Trazodone-induced hepatotoxicity: a case report with comments on drug-induced hepatotoxicity. Am J Gastroenterol 2000;95:532–535. [ Links ]
2 Ramachandran R, Kakar S: Histological patterns in drug-induced liver disease. J Clin Pathol 2009;62:481–492. [ Links ]
3 Larrey D, Ripault MP: Benzodiazepines. Hepatotoxicity of psychotropic drugs and drugs of abuse; in Kaplowitz N, DeLeve LD (eds): Drug-Induced Liver Disease, ed 3. Amsterdam, Elsevier, 2013, p 455. [ Links ]
4 Hull M, Jones R, Bendall M: Fatal hepatic necrosis associated with trazodone and neuroleptic drugs. BMJ 1994;309:378. [ Links ]
5 Detry O, Delwaide J, De Roover A, Hans MF, Delbouille MH, Monard J, Honoré P: Fulminant hepatic failure induced by venlafaxine and trazodone therapy: a case report. Transplant Proc 2009;41:3435–3436. [ Links ]
Statement of Ethics
This study did not require informed consent nor review/approval by the appropriate ethics committee.
Disclosure Statement
The authors have no conflicts of interest to declare.
* Corresponding author.
Dr. Sofia Carvalhana
Departamento de Gastrenterologia, Hospital de Santa Maria
Av. Prof. Egas Moniz
PT–1649-028 Lisbon (Portugal)
E-Mail sofiacarvalhana@msn.com
Received: May 10, 2016; Accepted after revision: August 10, 2016
Acknowledgements
All authors contributed equally. They thank Dr. Ana Carvalho for the liver biopsy pictures.














