Serviços Personalizados
Journal
Artigo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Acessos
Acessos
Links relacionados
-
 Similares em
SciELO
Similares em
SciELO
Compartilhar
GE-Portuguese Journal of Gastroenterology
versão impressa ISSN 2341-4545
GE Port J Gastroenterol vol.24 no.2 Lisboa abr. 2017
https://doi.org/10.1159/000450901
ORIGINAL ARTICLE
Adenoma Detection Rate: I Will Show You Mine if You Show Me Yours
Taxa de Deteção de Adenomas: Revelo a Minha Se Revelares a Tua
Alexandre Oliveira Ferreira, Catarina Fidalgo, Carolina Palmela, Maria Pia Costa Santos, Joana Torres, Joana Nunes, Rui Loureiro, Rosa Ferreira, Elídio Barjas, Luísa Glória, António Alberto Santos, Marília Cravo
Gastroenterology Department, Hospital Beatriz Ângelo, Loures, Portugal
* Corresponding author.
ABSTRACT
Background: Colorectal cancer (CRC) is the first cause of cancer-related mortality in Portugal. CRC screening reduces disease-specific mortality. Colonoscopy is currently the preferred method for screening as it may contribute to the reduction of CRC incidence. This beneficial effect is strongly associated with the adenoma detection rate (ADR). Aim: Our aim was to evaluate the quality of colonoscopy at our unit by measuring the currently accepted quality parameters and publish them as benchmarking indicators. Methods: From 5,860 colonoscopies, 654 screening procedures (with and without previous fecal occult blood testing) were analyzed. Results: The mean age of the patients was 66.4 ± 7.8 years, and the gender distribution was 1: 1. The overall ADR was 36% (95% confidence interval [CI] 32–39), the mean number of adenomas per colonoscopy was 0.66 (95% CI 0.56–0.77), and the sessile serrate lesion detection rate was 1% (95% CI 0–2). The bowel preparation was rated as adequate in 496 (76%) patients. The adjusted cecal intubation rate (CIR) was 93.7% (95% CI 91.7–95.8). Most colonoscopies were performed under monitored anesthesia care (53%), and 35% were unsedated. The use of sedation (propofol or midazolam based) was associated with a higher CIR with an odds ratio of 3.60 (95% CI 2.02–6.40, p < 0.001). Conclusion: Our data show an above-standard ADR. The frequency of poor bowel preparation and the low sessile serrated lesion detection rate were acknowledged, and actions were implemented to improve both indicators. Quality auditing in colonoscopy should be compulsory, and while many units may do so internally, this is the first national report from a highthroughput endoscopy unit.
Keywords: Adenoma; Colonoscopy; Colorectal neoplasms; Quality of health care; Quality health care indicators
RESUMO
O cancro do colon e reto (CCR) é a primeira causa de cancro e de morte por cancro em Portugal. O rastreio reduz a mortalidade específica por CCR. A colonoscopia é o método preferencial para o rastreio uma vez que pode contribuir para a redução da incidência do CCR. Este efeito está fortemente associado à taxa de deteção de adenomas (TDA). O nosso objetivo foi avaliar e dar a conhecer a qualidade da colonoscopia na nossa unidade, através da medição dos principais indicadores de qualidade e torná-los públicos como indicadores de aferição para outras unidades. De um total de 5,860 colonoscopias foram selecionadas para análise 654 de rastreio (com ou sem pesquisa de sangue oculto prévia). A idade média foi de 66.4 ± 7.8 anos e a distribuição por género de 1: 1. A TDA global foi de 36% (95% CI 32–39), o número médio de adenomas por colonoscopia foi de 0.66 (95% CI 0.56–0.77) e a taxa de deteção de lesões serreadas sésseis foi 1% (95% CI 0–2). A preparação intestinal foi considerada adequada em 496 (76%). A taxa de intubação cecal ajustada foi de 93.7% (95% CI 91.7–95.8). A maioria das colonoscopias foi realizada sob sedação profunda/anestesia por anestesista (53%) e 35% foram sem sedação. A utilização de sedação está associada a uma maior taxa de intubação cecal (OR 3.60; 95% CI 2.02–6.40, p < 0.001). Estes dados revelam uma TDA superior ao mínimo definido para colonoscopia de qualidade. A frequência de preparações intestinais inadequadas e a baixa taxa de deteção de lesões serreadas sésseis são indicadores importantes que foram reconhecidos e levaram a medidas de melhoria de qualidade na nossa unidade. A auditoria de qualidade em colonoscopia deve ser realizada de forma contínua e embora muitas unidades façam auditorias internas, esta é a primeira publicação com os dados de uma unidade de endoscopia nacional.
Palavras-Chave: Adenoma; Colonoscopia; Neoplasias colorrectais; Indicadores de qualidade em assistência à saúde; Qualidade de cuidados de saúde
Introduction
Colorectal cancer (CRC) is the first cause of cancer-related mortality in Portugal and a leading cause of cancer deaths in the world [1]. CRC screening reduces diseasespecific mortality [2–10]. Colonoscopy is currently the preferred method for screening [11, 12] as it allows for the detection and removal of premalignant lesions and may contribute for the reduction of CRC incidence [2, 4, 5, 13, 14], which is still increasing in Portugal [1, 15]. However, this beneficial effect is strongly associated with the adenoma detection rate (ADR) [2], which is the single most important quality surrogate for screening colonoscopy [16]. Besides ADR, the European Society of Gastrointestinal Endoscopy (ESGE) published a set of indicators and the recommended quality thresholds to ensure effective screening in Europe [17].
In Portugal, there is no organized CRC screening program yet. Screening colonoscopy is performed in hospitals, ambulatory centers, or office-based endoscopy clinics, and there is no systematic audit of colonoscopy quality in place.
Health services research is increasingly being valued as a means to study the outcome of specific interventions and to establish benchmarking criteria to healthcare providers. It also enables to detect organizational underperformance in order to undertake conscientious changes.
Our aim was to evaluate the quality of colonoscopy at the Hospital Beatriz Angelo in Loures, Portugal, in the first 3 years since its opening in 2012, having as comparators the established indicator thresholds when available.
Methods
We conducted a single-center, cross-sectional study in the secondary care hospital Hospital Beatriz Angelo (HBA) between January 2012 and December 2014. The data were retrospectively collected.
Patients
We selected all patients ≥ 50 years of age who were referred to HBA directly for colonoscopy screening or following a positive fecal occult blood test (FOBT). Patients referred for colonoscopy for other indications, including surveillance after resection of colorectal lesions and a family history of CRC or adenomas, were excluded from the analysis.
All patients were pre-evaluated at a gastroenterology appointment where the written informed consent for the procedure was obtained.
Bowel preparation was accomplished using verbal and written information. Patients were informed to take a 3-day low-residue diet, a low-volume (2 L) polyethylene glycol bowel preparation (Moviprep ® ; Norgine Limited, Hengoed, UK), and 2 tablets of bisacodyl 5 mg in the evening prior to the procedure for morning patients and a split-dose regimen for those in the afternoon schedule.
Setting
The Endoscopy Unit at HBA is integrated in a surgical ambulatory care center and comprises 3 endoscopy rooms equipped with Olympus Evis-Exera II (Olympus, Tokyo, Japan) video processors and endoscopes of the 160 and 180 series. The electrosurgical units are VIO 200D and 200S models (Erbe Elektromedizin GmbH, Tubingen, Germany). All rooms are equipped with an anesthesia workstation (Fabius Tiro; Drager, Vienna, Austria).
The recovery room has a total capacity of 26 patients, 10 of which are attributed to the Endoscopy Unit.
During the study period, the unit was staffed by 8 gastroenterology consultants and 1 gastroenterology resident.
Each room is staffed by an endoscopist, a nurse, and a staff assistant. For the cases performed under propofol sedation – monitored anesthesia care (MAC) – an anesthesiologist and a second nurse were also staffing the room.
Outcomes
We used the institutions electronic health record to collect individual patient demographic characteristics as well as colonoscopy quality indicators, which were as follows: ADR (calculated as the number of colonoscopies with histologically confirmed adenomas over the total number of colonoscopies); the mean number of histologically confirmed adenomas per colonoscopy; lesion detection rate (number of colonoscopies with endoscopically detected lesions over the total number of colonoscopies); number of endoscopic detected lesions per colonoscopy; advanced ADR (lesion size ≥ 10 mm, high-grade dysplasia, or villous histology); CRC detection rate; lesion attack rate (number of lesions removed over number of lesions detected); cecal intubation rate (CIR), crude and adjusted for stenosis); rate of cecal intubation photographic documentation; bowel preparation quality as rated by the endoscopist as adequate (good and fair) or inadequate; type of sedation (propofol based, midazolam based, or none); written surveillance recommendation rate, and complication rate (clinically significant bleeding and perforation or post-polypectomy syndrome) that involved admittance of the patient or a subsequent emergency room episode.
As quality thresholds for CIR, we used the bowel preparation quality and informed consent rate for those set by the ESGE [17], and for the remainders, we used the thresholds set by the American Society for Gastrointestinal Endoscopy (ASGE) [16].
Statistical Analysis
The mean and standard deviations are shown for continuous variables with a normal distribution. These were compared using an independent t test. The other continuous variables were compared using the Mann-Whitney U test.
Categorical variables are presented as proportions (%) and compared with the Fisher or χ 2 tests. For the estimation of the confidence intervals (CIs), the simple asymptotic method was used. Logistic regression was used for dichotomous outcomes in order to determine the effect estimates that are presented as odds ratio (OR) and 95% CIs. Missing data were dealt by pairwise deletion.
Results
From a total of 5,860 colonoscopies performed during the study period, 736 were included for review. After individual review of each patients electronic health record, 82 were excluded as they were considered to be diagnostic procedures for symptomatic patients. The final sample was composed of 654 colonoscopies, and the demographic and procedural characteristics are depicted in Table 1 .
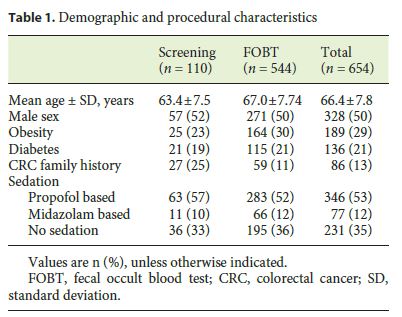
The mean age was 66.4 ± 7.8 years, and the gender ratio was 1: 1. Colonoscopy quality indicators are shown in Table 2. The overall ADR is 36% (95% CI 32–39) 45.8 and 25.1% for the male and female patients, respectively. The mean adenoma number per colonoscopy was 0.66 (95% CI 0.56–0.77).
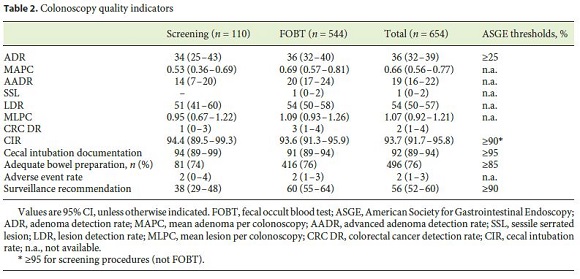
The bowel preparation was rated by the endoscopist as adequate (excellent, good, or fair) in 496 (76%) patients. 236 (35%) patients were submitted to unsedated colonoscopy, while the majority (53%) were offered propofolbased deep sedation under anesthesiologist care.
The crude CIR was 92% (95% CI 89–94) and 93.7% (95% CI 91.7–95.8) after adjusting for stenosis and poor bowel preparation. Table 3 shows the adjusted CIR according to the sedation type. The use of sedation (propofol or midazolam based) was associated with a higher CIR with an OR of 3.60 (95% CI 2.02–6.40, p < 0.001). Concerning CIR, there was no statistically significant difference between propofol- or midazolam-based sedation (OR 0.88, 95% CI 0.29–2.72, p = 0.831).

To increase the CIR by 1, the number that needs to be sedated is 18.9.
With these data, we can calculate a number needed to diagnose of 2.8 for colorectal adenomas and 50 colonoscopies for CRC. In the subgroup of positive FOBT, the numbers were 2.8 and 33, respectively.
To calculate the cost-effectiveness ratio for CRC and adenoma detection, we used the value payed by the national health system to private units, which is set at EUR 101.23 for a colonoscopy. Accordingly, the cost for the detection and removal of an adenoma is EUR 283.44, and EUR 5,061.50 for 1 diagnosis of CRC.
Discussion
HBA is a newly built hospital (2012), and its management places an important focus on quality improvement and innovation. The current study results from this need and aims at promoting the design and implementation of specific measures to improve the outcomes.
The single most important outcome to measure the effectiveness of colonoscopy is the ADR, since it is associated with the future risk of CRC incidence and mortality [2]. The ADR at our unit (36%, 95% CI 32–39) is well above the quality threshold set by the endoscopy societies, which is currently 25% [16]. The benefit of knowing our own ADR may also motivate quality improvement, as has been shown in several interventional studies with the implementation of scheduled personalized ADR report cards. Endoscopists thrive when they are aware of their own quality metrics [18, 19].
It is our intention to maintain the audit in order to promote a continuous incentive to improve the yield of colonoscopy.
Nevertheless, although ADR is considered the best surrogate marker of colonoscopy quality, it is associated with several shortcomings such as allowing itself to be gamed while inducing a one-and-done performance by the practicing endoscopist [16]. To overcome this limitation, the mean number of adenomas per colonoscopy is an alternative indicator that is gaining acceptance as it possesses more information than the ADR [16, 20]. The mean number of histologically confirmed adenomas per colonoscopy in our cohort (0.66 [95% CI 0.56–0.77]) is well above the threshold of 0.5 lesions per colonoscopy proposed by the Indiana Group [20].
The very low sessile serrated lesion (SSL) detection has led us to discuss the issue with our pathologists, and we are currently performing a large-scale randomized trial to evaluate a specific intervention to improve SSL detection. Participation in research protocols may constitute by itself an incentive to overperform, and this is a hypothesis that we will be evaluated in the future.
The CIR (93.7%) was also within the established quality threshold. The use of sedation seems to have been valuable in this regard as it was used in the majority of procedures and was associated with a higher CIR.
Regarding procedural sedation, our unit has a higher rate of propofol sedation than the reported in Portuguese public hospitals in a recent national survey [21]. The ESGE evidence-based guideline endorses the use of nonanesthesiology-administered propofol sedation [22], but the Portuguese National Health Administration (Direção Geral de Saúde) recommends the routine use of MAC for screening colonoscopy. Sedation has been extensively studied and mainly improves patients comfort and acceptance with little (if any) added risk with anesthesia services [23].
The data presented herein support sedation use, either as moderate/traditional sedation or as MAC. The usage of sedation was associated with a higher CIR with a number need to treat of 18.9, and although we did not evaluate safety, we have previously studied propofol-based deep sedation during colonoscopy in a strictly controlled clinical trial, and there were no serious adverse events [24].
Still, there is ongoing and renewed discussion on the benefits of sedation since the NordICC trial exposed some evidence failing to associate a benefit in comfort, CIR, and ADR with sedation [25]. Moreover, concern over the potential for the increase in adverse events due to sedation reemerged with the analysis of over 3 million colonoscopy administrative claims in the USA (patients aged 40–64 years) by Wernli et al. [23], where 34% were performed with anesthesia services. In that study, anesthesia was associated with a significant increased risk for complications (OR 1.13 [95% CI 1.12–1.14]), albeit a low absolute risk. A safe and very cost-effective alternative to MAC is nonanesthesiologist administration of propofol as we have shown previously in a noninferiority randomized controlled trial, which enrolled 277 low-risk (American Society of Anesthesiologists [ASA] <3) patients [24]. Currently, we offer the option of moderate sedation to all our patients and MAC to selected patients.
One of the most important results obtained was the acknowledgement of a high proportion of patients with a bowel preparation quality considered inadequate. Almost a quarter of all procedures were deemed poorly prepared by the endoscopist. Poor bowel preparation has been associated with a lower CIR and ADR [26], as well as higher rates of adverse events and repeat procedures. This study allowed us to acknowledge the underperformance and to determine bowel preparation as a priority issue in our unit. An intervention to optimize it is now in place. The intervention consists of adopting split-dose and same-day regimens for morning and evening procedures and a newly designed written document with emphasis on simplicity. Split-dose for morning procedures has been shown to improve ADR and especially the quality of the preparation in the right bowel [27, 28]. Although, this scheme is advocated by major societies [29, 30], its uptake has been suboptimal due to factors such as fear of increased aspiration risk, fecal incontinence, and low patient education [31], even with the ASA guidelines advocating a 2-hour clear liquid fast for all forms of anesthesia in patients without risk factors for aspiration [32].
The aim of the intervention is to lower the inadequate preparations to a value <15% in order to comply with the quality metrics and improve our ADR while decreasing the number of repeat procedures. We have implemented an ongoing auditing strategy to measure the impact of the intervention, which will soon be reported.
Studies such as the present one show a commitment to quality that should be mandatory in all endoscopy units. We believe that the reports of critical quality indicators such as the ADR should be made public and wish to contribute by taking a first step towards transparency and benchmarking in colonoscopy in Portugal.
Moreover, the continuous audit of quality parameters and the comparison of benchmarks may contribute to implement proven interventions or hypothesize new interventions that may contribute to the increase in effectiveness (or safety) of colonoscopy. As this was an internal audit conducted by the endoscopy unit personnel, an obvious conflict of interest has to be acknowledged. The ideal option would be to have an external audit or natural language software to calculate the quality indicators autonomously
As a limitation we must acknowledge the fact that most patients included were not screening naive as 83% had a positive FOBT as the indication for colonoscopy. However, the ADR in both screening and FOBT groups was remarkably similar, and the estimated CIs were above 25% in both groups.
Another important limitation is the lack of a validated bowel preparation quality scale in our analysis. This is due to the retrospective design of the study and because only one-third of the procedures had reported values for each segment of the colon using a validated scale (the Boston Bowel Preparation Score). All reports used a subjective scale of poor/fair/good preparation determined by an endoscopist. Following this study, it became mandatory in our unit to systematically assess and include the preparation quality in the colonoscopy report.
The presented data also prompted us to implement a proven strategy to improve the bowel preparation quality and designed a multicenter randomized controlled trial to test a specific intervention to improve the SSL detection rate.
The ultimate goal of this study is to increase the public acceptance for colonoscopy by showing data to support its effectiveness and to decrease the incidence- and CRCassociated mortality in Portugal. Moreover, the Portuguese government recently issued an executive document in order to implement a CRC screening strategy in Portugal by 2017. Such a program has to bear in its core the awareness of the importance of quality colonoscopy. We urge colonoscopists to embrace quality metrics and make them public while external audit is not in place. Such transparency will hopefully contribute to make colonoscopy the most cost-effective screening strategy in Portugal.
References
1 Ferlay J, Soerjomataram I, Dikshit R, Eser S, Mathers C, Rebelo M, Parkin DM, et al: Cancer incidence and mortality worldwide: sources, methods and major patterns in GLOBOCAN 2012. Int J Cancer 2015;136:E359–E386. [ Links ]
2 Corley DA, Jensen CD, Marks AR, Zhao WK,Lee JK, Doubeni CA, Zauber AG, et al: Adenoma detection rate and risk of colorectal cancer and death. N Engl J Med 2014;370:1298–1306. [ Links ]
3 Shaukat A, Mongin SJ, Geisser MS, Lederle FA, Bond JH, Mandel JS, Church TR: Longterm mortality after screening for colorectal cancer. N Engl J Med 2013;369:1106–1114. [ Links ]
4 Nishihara R, Wu K, Lochhead P, Morikawa T, Liao X, Qian ZR, Inamura K, et al: Long-term colorectal-cancer incidence and mortality after lower endoscopy. N Engl J Med 2013;369:1095–1105. [ Links ]
5 Zauber AG, Winawer SJ, OBrien MJ, Lansdorp-Vogelaar I, van Ballegooijen M, Hankey BF, Shi W, et al: Colonoscopic polypectomy and long-term prevention of colorectal-cancer deaths. N Engl J Med 2012;366:687–696. [ Links ]
6 Schoen RE, Pinsky PF, Weissfeld JL, Yokochi LA, Church T, Laiyemo AO, Bresalier R, et al: Colorectal-cancer incidence and mortality with screening flexible sigmoidoscopy. N Engl J Med 2012;366:2345–2357. [ Links ]
7 Lieberman DA, Weiss DG: One-time screening for colorectal cancer with combined fecal occult-blood testing and examination of the distal colon. N Engl J Med 2001;345:555–560. [ Links ]
8 Winawer SJ, Zauber AG, OBrien MJ, Ho MN, Gottlieb L, Sternberg SS, Waye JD, et al: Randomized comparison of surveillance intervals after colonoscopic removal of newly diagnosed adenomatous polyps. The National Polyp Study Workgroup. N Engl J Med 1993;328:901–906. [ Links ]
9 Winawer SJ, Zauber AG, Ho MN, OBrien MJ, Gottlieb LS, Sternberg SS, Waye JD, et al: Prevention of colorectal cancer by colonoscopic polypectomy. The National Polyp Study Workgroup. N Engl J Med 1993;329:1977–1981. [ Links ]
10 Mandel JS, Bond JH, Church TR, Snover DC, Bradley GM, Schuman LM, Ederer F: Reducing mortality from colorectal cancer by screening for fecal occult blood. Minnesota Colon Cancer Control Study. N Engl J Med 1993;328:1365–1371. [ Links ]
11 Qaseem A, Denberg TD, Hopkins RH Jr, Humphrey LL, Levine J, Sweet DE, Shekelle P: Screening for colorectal cancer: a guidance statement from the American College of Physicians. Ann Intern Med 2012;156:378–386. [ Links ]
12 Rex DK, Johnson DA, Anderson JC, Schoenfeld PS, Burke CA, Inadomi JM: American College of Gastroenterology guidelines for colorectal cancer screening 2009 [corrected]. Am J Gastroenterol 2009;104:739–750. [ Links ]
13 Edwards BK, Noone AM, Mariotto AB, Simard EP, Boscoe FP, Henley SJ, Jemal A, et al: Annual Report to the Nation on the status of cancer, 1975–2010, featuring prevalence of comorbidity and impact on survival among persons with lung, colorectal, breast, or prostate cancer. Cancer 2014;120:1290–1314. [ Links ]
14 Lieberman DA, Weiss DG, Harford WV, Ahnen DJ, Provenzale D, Sontag SJ, Schnell TG, et al: Five-year colon surveillance after screening colonoscopy. Gastroenterology 2007;133:1077–1085. [ Links ]
15 Ferlay J, Soerjomataram I, Ervik M, Dikshit R, Eser S, Mathers C, Rebelo M, et al: Cancer Incidence and Mortality Worldwide: IARC CancerBase No. 11 [Internet]. Lyon, International Agency for Research on Cancer, 2013. GLOBOCAN 2012 v1.0. [ Links ]
16 Rex DK, Schoenfeld PS, Cohen J, Pike IM, Adler DG, Fennerty MB, Lieb JG 2nd, et al: Quality indicators for colonoscopy. Gastrointest Endosc 2015;81:31–53. [ Links ]
17 Rembacken B, Hassan C, Riemann JF, Chilton A, Rutter M, Dumonceau JM, Omar M, et al: Quality in screening colonoscopy: position statement of the European Society of Gastrointestinal Endoscopy (ESGE). Endoscopy 2012;44:957–968. [ Links ]
18 Keswani RN, Yadlapati R, Gleason KM, Ciolino JD, Manka M, OLeary KJ, Barnard C, et al: Physician report cards and implementing standards of practice are both significantly associated with improved screening colonoscopy quality. Am J Gastroenterol 2015;110:1134–1139. [ Links ]
19 Kahi CJ, Ballard D, Shah AS, Mears R, Johnson CS: Impact of a quarterly report card on colonoscopy quality measures. Gastrointest Endosc 2013;77:925–931. [ Links ]
20 Kahi CJ, Vemulapalli KC, Johnson CS, Rex DK: Improving measurement of the adenoma detection rate and adenoma per colonoscopy quality metric: the Indiana University experience. Gastrointest Endosc 2014;79:448–454. [ Links ]
21 Ferreira AO, Torres J, Dinis-Ribeiro M, Cravo M: Endoscopic sedation and monitoring practices in Portugal: a nationwide web-based survey. Eur J Gastroenterol Hepatol 2015;27:265–270. [ Links ]
22 Dumonceau JM, Riphaus A, Schreiber F, Vilmann P, Beilenhoff U, Aparicio JR, Vargo JJ, et al: Non-anesthesiologist administration of propofol for gastrointestinal endoscopy: European Society of Gastrointestinal Endoscopy,European Society of Gastroenterology and Endoscopy Nurses and Associates Guideline – Updated June 2015. Endoscopy 2015;47:1175–1189. [ Links ]
23 Wernli KJ, Brenner AT, Rutter CM, Inadomi JM: Risks associated with anesthesia services during colonoscopy. Gastroenterology 2016;150:888–894. [ Links ]
24 Ferreira AO, Torres J, Barjas E, Nunes J, Gloria L, Ferreira R, Rocha M, et al: Non-anesthesiologist administration of propofol sedation for colonoscopy is safe in low risk patients: results of a noninferiority randomized controlled trial. Endoscopy 2016;48:747–753. [ Links ]
25 Bretthauer M, Kaminski MF, Loberg M, Zauber AG, Regula J, Kuipers EJ, Hernan MA, et al: Population-based colonoscopy screening for colorectal cancer: a randomized clinical trial. JAMA Intern Med 2016;176:894–902. [ Links ]
26 Sulz MC, Kroger A, Prakash M, Manser CN, Heinrich H, Misselwitz B: Meta-analysis of the effect of bowel preparation on adenoma detection: early adenomas affected stronger than advanced adenomas. PLoS One 2016;11:e0154149. [ Links ]
27 Martel M, Barkun AN, Menard C, Restellini S, Kherad O, Vanasse A: Split-dose preparations are superior to day-before bowel cleansing regimens: a meta-analysis. Gastroenterology 2015;149: 9–88. [ Links ]
28 Enestvedt BK, Tofani C, Laine LA, Tierney A, Fennerty MB: 4-Liter split-dose polyethylene glycol is superior to other bowel preparations, based on systematic review and meta-analysis. Clin Gastroenterol Hepatol 2012;10:1225–1231. [ Links ]
29 Saltzman JR, Cash BD, Pasha SF, Early DS, Muthusamy VR, Khashab MA, Chathadi KV, et al: Bowel preparation before colonoscopy. Gastrointest Endosc 2015;81:781–794. [ Links ]
30 Hassan C, Bretthauer M, Kaminski MF, Polkowski M, Rembacken B, Saunders B, Benamouzig R, et al: Bowel preparation for colonoscopy: European Society of Gastrointestinal Endoscopy (ESGE) guideline. Endoscopy 2013;45:142–150. [ Links ]
31 Radaelli F, Paggi S, Repici A, Gullotti G, Cesaro P, Rotondano G, Cugia L, et al: Barriers against split-dose bowel preparation for colonoscopy. Gut 2016, Epub ahead of print. [ Links ]
32 Apfelbaum JL, Caplan RA, Connis RT, Epstein BS, Nickinovich DG, Warner MA: Practice guidelines for preoperative fasting and the use of pharmacologic agents to reduce the risk of pulmonary aspiration: application to healthy patients undergoing elective procedures: an updated report by the American Society of Anesthesiologists Committee on Standards and Practice Parameters. Anesthesiology 2011;114:495–511. [ Links ]
Statement of Ethics
Written informed consent for the procedure was obtained by the patients.
Disclosure Statement
The authors have no conflicts of interest to declare.
* Corresponding author.
Dr. Alexandre Oliveira Ferreira
Servico de Gastrenterologia, Hospital Beatriz Angelo
Avenida Carlos Teixeira 3
PT–2674-514 Loures (Portugal)
E-Mail alex.gastrohep@gmail.com
Received: June 29, 2016; Accepted after revision: September 13, 2016














