Serviços Personalizados
Journal
Artigo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Acessos
Acessos
Links relacionados
-
 Similares em
SciELO
Similares em
SciELO
Compartilhar
GE-Portuguese Journal of Gastroenterology
versão impressa ISSN 2341-4545
GE Port J Gastroenterol vol.24 no.2 Lisboa abr. 2017
https://doi.org/10.1159/000450872
CLINICAL CASE STUDY
Paraduodenal Pancreatitis: Three Cases with Different Therapeutic Approaches
Pancreatite Paraduodenal: Três Casos Com Diferentes Abordagens Terapêuticas
Diana Carvalho, Rafaela Loureiro, Verónica Pavão Borges, Pedro Russo, Carlos Bernardes, Gonçalo Ramos
Gastroenterology and Hepatology Department, Hospital Santo António dos Capuchos, Centro Hospitalar de Lisboa Central, Lisbon, Portugal
* Corresponding author.
ABSTRACT
Background: Paraduodenal pancreatitis is a rare cause of chronic abdominal pain characterized by an inflammatory process and scarring in the groove area between the pancreatic head and the duodenal wall. Besides abdominal pain, symptoms such as vomiting and weight loss are common. Currently, advances in radiological and endoscopic diagnostic methods allow it to be identified without histological confirmation, although the differentiation from pancreatic adenocarcinoma could be challenging in some cases. Many therapeutic options are available nowadays including pharmacological, endoscopic, or surgical treatment. Methods: We report 3 cases of paraduodenal pancreatitis that had different therapeutic approaches. Results and Conclusion: They show that this pathology should be taken into account in the differential diagnosis of pancreatic masses with duodenal infiltration, and that its management should be individualized and judicious.
Keywords: Pancreatitis; Chronic abdominal pain; Pancreaticoduodenectomy; Somatostatin; Duodenal obstruction
RESUMO
Introdução: A pancreatite paraduodenal constitui uma causa rara de dor abdominal crónica, caracterizada por processo inflamatório com consequente cicatrização no sulco entre a cabeça do pâncreas e a parede duodenal. Para além de dor abdominal, sintomas como vómitos e perda de peso são frequentes. Actualmente, com o desenvolvimento dos métodos diagnósticos imagiológicos e endoscópicos, pode ser identificada sem necessidade de confirmação histológica, embora a diferenciação com o adenocarcinoma pancreático possa ser desafiante. Diversas opções terapêuticas encontram-se disponíveis incluindo tratamento farmacológico, endoscópico ou cirúrgico. Métodos: Os autores descrevem três casos de pancreatite paraduodenal com diferentes atitudes terapêuticas. Resultados e Conclusão: Estes demonstram que esta patologia deve ser considerada no diagnóstico diferencial de massas pancreáticas com infiltração duodenal e que a sua abordagem deve ser individualizada e judiciosa.
Palavras-Chave: Pancreatite crónica; Dor abdominal crónica; Pancreaticoduodenectomia; Somatostatina; Obstrução duodenal
Introduction
Paraduodenal pancreatitis (PP) is a pathological condition resulting from fibrotic inflammation that involves the duodenal wall closely to the minor papilla, the adjacent pancreatic parenchyma, and the area between them [1]. Its pathophysiology remains unclear, although it is believed that alcohol consumption and the presence of ectopic pancreatic tissue in the duodenal wall may play a major role in its development [2]. Through the last years, its treatment has been a topic of discussion in the literature, with the proposal of a wide range of conservative and surgical approaches [3]. Nowadays, the consensus is to initially prefer medical or endoscopic treatment, surgery being reserved to patients with uncertain diagnosis or symptoms that do not improve with other measures [4].
We present 3 cases of PP that were managed with 3 different therapeutics procedures: supportive treatment, use of octreotide, and surgery with pancreaticoduodenectomy.
Case 1
A 55-year-old male patient, with daily consumption of 130 g of alcohol and tobacco, was admitted to the Gastroenterology Department for severe epigastric abdominal pain accompanied by anorexia, nausea, vomiting, and weight loss (3 kg). An upper gastrointestinal endoscopy was performed, revealing a bulb deformed by a vegetating lesion, not allowing visualization of the second duodenal portion (D2); histology was negative for malignancy.
During hospitalization, he improved with analgesia, intravenous fluids, and bowel rest. Laboratory evaluation showed carbohydrate antigen (CA) 19.9 of 61 ng/mL, with no other changes. Magnetic resonance imaging (MRI) showed a large abdominal lesion involving the bulb and D2, which was predominantly hyperintense on T2 and hypointense on T1, without significant contrast enhancement. In the pancreatic head, a cystic lesion was observed (Fig. 1).
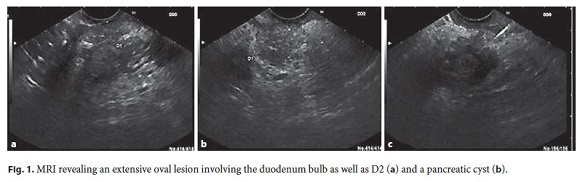
Endoscopic ultrasound (EUS) revealed thickening of D2 (9 mm) with microcysts in the wall, chronic pancreatitis and a cyst (21 mm) in the pancreatic head. Endoscopically, no irregularities of the duodenal mucosa were observed and passage to D2 was possible (Fig. 2). Fine-needle aspiration (FNA) of the pancreatic cyst revealed an amylase of 108,643 U/L and carcinoembryonic antigen (CEA) 44.5 ng/mL on cytochemical examination.
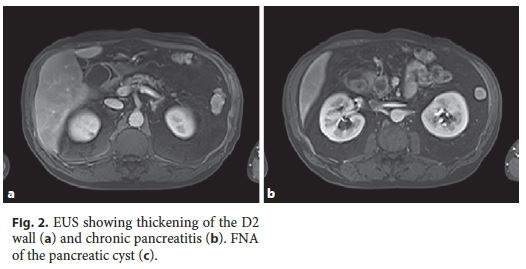
Reassessment imaging by computed tomography exhibited a significant regression of previous duodenal and pancreatic lesions. A presumptive diagnosis of PP was made. After multidisciplinar team discussion, close monitoring was proposed with radiological examination, abstinence of alcohol, and smoking cessation. During the 13 months of follow-up, the patient remains asymptomatic, with no laboratory or radiological abnormalities.
A close follow-up was decided in this case because of the favourable clinical response and the patients adherence to alcohol and tobacco abstinence. It is important to state that all current therapeutic alternatives (medical, endoscopic, or surgical) carry some degree of risk, which was deemed unwarranted in this case.
Case 2
A 58-year-old female patient was referred to the Gastroenterology Department for recurrent epigastric pain not related to food ingestion, with irradiation to the right abdominal flank, followed by nauseas. The patient had no history of addictive behavior. Blood tests revealed cholestatic changes (alkaline phosphatase 222 U/L and gamma-glutamyltransferase 82 U/L) and normal IgG4 levels. She was treated with ursodeoxycholic acid (1,000 mg/day) and pancreatin without clinical improvement.
A MRI was performed, showing parietal concentric thickening of D2 with apparent origin in the submucosal layer. Endoscopically, the duodenal mucosa was edematous, with decreased luminal caliber at this level; biopsies did not reveal any pathological changes. An EUS confirmed a slight hypoechoic thickening of the D2 wall (5 mm), especially for the submucosa, without cystic areas or other abnormalities in the echo structure. The pancreatic parenchyma was regular, but with marked atrophy of the body/tail segments (Fig. 3). PP was diagnosed, and she started therapy with octreotide (20 mg with interval of 4 weeks). She had marked pain reduction, with a sustained clinical response after 1 year of follow-up.
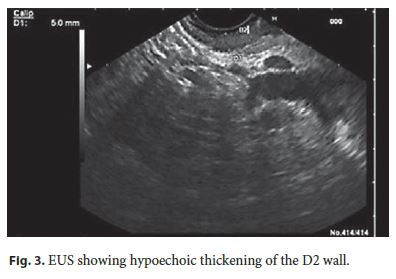
The treatments choice was due to the persistence of pain despite conservative measures (i.e. alcohol abstinence) and the absence of complications that could justify an endoscopic approach. In order to avoid a more invasive approach, the somatostatin analog octreotide was considered the best option for symptomatic relief.
Case 3
A 56-year-old male patient with chronic consumption of alcohol (50 g/day) and tobacco (36 pack-year) was referred to the Gastroenterology Department for intense epigastric postprandial pain with 4 months of evolution. He also mentioned nauseas, vomiting, and weight loss (15 kg). Laboratory tests only showed hyperamylasemia (312 U/L). He was treated with omeprazole 40 mg twice daily, domperidone 10 mg/day, and pregabalin 150 mg/day, without symptomatic relief.
For clarification, an abdominal computed tomography was done, revealing D2 thickening (12 mm) with wall heterogeneity. EUS evidenced a heterogeneous circumferentially thickened duodenal wall (8.7 mm), mainly the third layer (submucosa), without focal lesions and a hypoechoic but homogeneous parenchyma in the pancreatic head (Fig. 4).
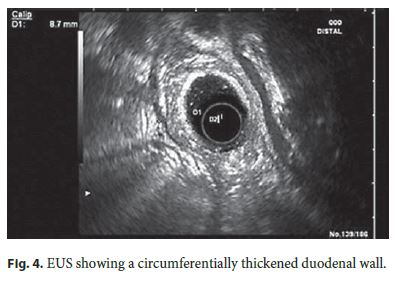
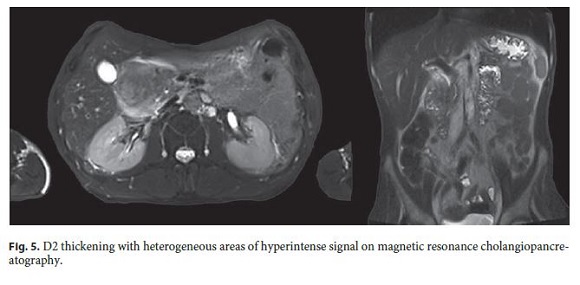
Over the next 2 months, the symptoms worsened, with continuous epigastric pain, uncontrollable vomiting, and weight loss (>20 kg). For this reason, the patient was admitted to the Gastroenterology Department and treated with fluid therapy and analgesia. He showed tolerance to mild oral diet but suffered moderate abdominal pain. Magnetic resonance cholangiopancreatography showed D2 thickening with heterogeneous contrast enhancement, areas of hyperintense signal, remaining cyst or necrosis, and mild heterogeneity of the pancreatic head contour (Fig. 5).
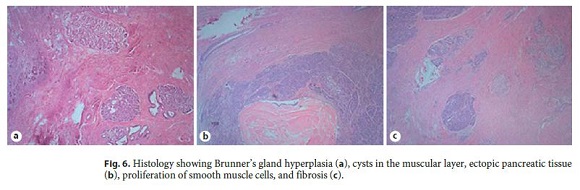
Our diagnostic hypotheses were PP versus duodenal malignancy and, in a multidisciplinary meeting, it was decided to perform a pancreaticoduodenectomy (Transverse-Longmire). The surgery was performed without complications, and histological examination of the surgical specimen revealed cysts in the muscular layer coated with mucinous epithelium in the region of the minor papilla. Ectopic pancreatic tissue, Brunners gland hyperplasia, abscesses, proliferation of smooth muscle cells, and fibrosis were also identified. These aspects confirmed the diagnosis of PP (Fig. 6).
Fifteen months after surgery, the patient remains asymptomatic with progressive increase of weight. In this patient, the definitive diagnosis remained uncertain after the diagnostic work-up, making the surgical approach the best option for both treatment and etiologic confirmation.
Discussion
PP is a rare cause of chronic abdominal pain that has been described for the first time by Becker in 1973 as a segmental pancreatitis [5]. Later, in the early 1990s, it was classified into the following 2 forms: pure, which involves only the groove area (space between the duodenal wall and pancreatic head), and segmental, with inflammation extending to the adjacent pancreatic head. In practice, this differentiation is not always possible to be clearly defined [6].
Its prevalence varies largely from 2.7 to 24.5% in surgical specimens of patients with chronic pancreatitis [5] and is most frequently found in men during the fifth decade [7]. Risk factors include severe alcohol consumption and heavy smoking [8] and in two-thirds, it is associated with chronic pancreatitis [9].
Various factors are implicated in PPs etiology: pancreatic ectopic tissue in the duodenal wall with localized inflammation and scarring; absence or functional obstruction of the minor papilla; increased fluid viscosity, Brunner gland hyperplasia and intraductal pressure caused by chronic consumption of alcohol, as well as modifications of local anatomy secondary to gastroduodenal ulcers, gastrectomy, or biliary diseases [8].
Usually patients present with upper abdominal pain associated with nausea and recurrent postprandial vomiting [10]. With chronicity, significant weight loss appears and eventually jaundice, diarrhea, and diabetes mellitus [4, 11].
In laboratory evaluation, a slight elevation of pancreatic and hepatic enzymes is frequently found [5]. The tumor markers CEA and CA 19.9 are normal, with few cases describing a small raise.
Diverse pathologies are included in the differential diagnosis: pancreatic adenocarcinoma, cystic tumors, autoimmune pancreatitis [8], duodenal carcinoma, cholangiocarcinoma, and acute relapsing pancreatitis. The first one is particularly challenging even with resource to imagiology and endoscopic modalities [1].
Imaging exams, especially MRI, are very important to confirm the diagnosis of PP. The spectrum of findings is large but the typical presentation involves focal thickening of the D2, cystic degeneration, and a sheet-like mass of fibrotic tissue within the groove [3]. Other changes that can be found include duct abnormalities (dilation, atrophy, and irregularity) and signs of chronic pancreatitis [3].
Nowadays, besides MRI, EUS is also considered a gold standard for PP diagnosis [2]. It reveals a hypoechoic area between the duodenal wall and the head of the pancreas, thickening and stenosis of the duodenal wall, intramural cysts and a slightly narrowing of the main biliary duct. Eventually, in cases of segmental PP, it is possible to see infiltration and calcification of pancreatic parenchyma, pseudocysts, and dilation of pancreatic duct [1, 4]. EUSFNA has limited value in this setting and depends on the biopsy site and its interpretation, being difficult to exclude a subjacent tumor [6]. Upper gastrointestinal endoscopy usually shows a swollen and polypoid duodenal mucosa and stenosis; however, biopsies are frequently inconclusive [8]. Endoscopic retrograde cholangiopancreatography can be difficult or even impossible due to duodenal stenosis. When successful, it may reveal distal stenosis of the main biliary duct or irregularities of Wirsung and Santorini ducts [4].
Histologically, the most characteristic findings consist of myofibroblastic and neural expansion into the groove area, thickening of the duodenal wall with fibrosis and Brunners gland and smooth muscle hyperplasia, occurrence of dilated ducts as well as pseudocysts within the duodenal wall, and the presence of hypercellullar granulation tissue and foreign body type giant cells [1, 12]. Cytology from fragments obtained by EUS can show a wide range of results, frequently with observation of spindled stromal cells. Although the majority is interpreted as negative for malignancy, the results can be misinterpreted as a tumor, especially if there is an abundance of Brunner gland hyperplasia and spindle cells [6, 12].
Currently, its treatment is based on a stepwise approach, which can be effective with an acceptable complication rate [9]. Conservative measures involve the abstinence of alcohol and tobacco, pancreatic rest, and analgesics, the first one being crucial for long-term results [8]. Recently, somatostatin analogs were considered an alternative medical option. However, few reports are described in literature and symptomatology recurrence seems to be frequent after treatment cessation [4].
Endoscopic treatment, such as stricture dilation and cysts or pancreatic ductal drainage, is an important nonsurgical approach with good results. In a study conducted by Arvanitakis et al. [9] involving 51 patients, this approach, together with medical treatment, had a high rate of clinical success (nearly 80%) and showed low adverse effects.
If symptoms do not improve with the previous strategies, complications appear, or when a suspicion of a neoplasm is present, surgery must be the treatment of choice. The technique usually performed is the Whipple procedure [4], but some authors sustain that more conservative interventions, such as suprapapillary segmental duodenal resection, should be considered in cases of isolated duodenal dystrophy [13]. Apart of the procedure used, it is proved that surgery highly improves quality of life and contributes to pain cessation [2].
In conclusion, PP should be considered in the differential diagnosis of pancreatic head masses with duodenal infiltration. Its treatment should be individualized to each case, choosing a conservative management whenever possible.
References
1 Arora A, Dev A, Mukund A, Patidar Y, Bhatia V, Sarin SK: Paraduodenal pancreatitis. Clin Radiol 2014; 69: 299–306. [ Links ]
2 Casetti L, Bassi C, Salvia R, Butturini G, Graziani R, Falconi M, et al: Paraduodenal pancreatitis: results of surgery on 58 consecutives patients from a single institution. World J Surg 2009; 33: 2664–2669. [ Links ]
3 Hungerford JP, Magarik MAN, Hardie AD: The breadth of imaging findings of groove pancreatitis. Clin Imag 2015; 39: 363–366. [ Links ]
4 Pallisera-Lloveras A, Ramia-Ángel JM, Vicens-Arbona C, Cifuentes-Rodenas A: Groove pancreatitis. Rev Esp Enferm Dig 2015; 107: 280–288. [ Links ]
5 Black TP, Guy CD, White RR, Obando J, Burbridge RA: Groove pancreatitis: four cases from a single center and brief review of the literature. ACG Case Rep J 2014; 1: 154–157. [ Links ]
6 Ram an SP, Salaria SN, Hruban RH, FishmanEK: Groove pancreatitis: spectrum of imaging findings and radiology-pathology correlation. AJR Am J Roentgenol 2013; 201:W29–W39. [ Links ]
7 Rebours V, Lévy P, Vullierme MP, Couvelard A, OToole D, Aubert A, et al: Clinical and morphological features of duodenal cystic dystrophy in heterotopic pancreas. Am J Gastro 2007; 102: 871–879. [ Links ]
8 DeSouza K, Nodit L: Groove pancreatitis: a brief review of a diagnostic challenge. Arch Pathol Lab Med 2015; 139: 417–421. [ Links ]
9 Arvanitakis M, Rigaux J, Toussai E, Eisendrath P, Bali MA, Matos C, et al: Endotherapy for paraduodenal pancreatitis: a large retrospective case series. Endoscopy 2014; 46: 580–587. [ Links ]
10 Brosens LAA, Leguit RJ, Vleggaar FP, Veldhuis WB, Van Leeuwen MS, Offerhaus GJA: EUS-guided FNA cytology diagnosis of paraduodenal pancreatitis (groove pancreatitis) with numerous giant cells: conservative management allowed by cytological and radiological correlation. Cytopathology 2015; 26: 122–125. [ Links ]
11 Gravito-Soares M, Gravito-Soares E, Alves A, Gomes D, Almeida N, Tralhão G, et al: Groove pancreatitis with biliary and duodenal stricture: an unusual cause of obstructive jaundice. GE Port J Gastroenterol 2016; 23: 170–174. [ Links ]
12 Chute DJ, Stelow EB: Fine-needle aspiration features of paraduodenal pancreatitis (groove pancreatitis): a report of three cases. Diagn Cytopathol 2012; 40: 1116–1121. [ Links ]
13 Egorov VI, Vankovich AN, Petrov RV, Starostina NS, Butkevich AT, Sazhin AV, et al: Pancreas-preserving approach to paraduodenal pancreatitis treatment: why, when, and how? Experience of treatment of 62 patients with duodenal dystrophy. Biomed Res Int 2014; 2014: 185265. [ Links ]
Statement of Ethics
This study did not require informed consent or review/approval by the appropriate ethics committee.
Disclosure Statement
The authors declare that they have no competing interests.
* Corresponding author.
Dr. Diana Carvalho
Gastroenterology and Hepatology Department, Hospital Santo António dos Capuchos
Centro Hospitalar de Lisboa Central
PT–1169–050 Lisbon (Portugal)
E-Mail dianafbcarvalho@gmail.com
Received: March 31, 2016; Accepted after revision: July 3, 2016
Erratum
In the article by Carvalho et al. entitled Paraduodenal Pancreatitis: Three Cases with Different Therapeutic Approaches [GE Port J Gastroenterol 2017;24:89–94, DOI: 10.1159/000450872], figures 1 and 2 have erroneously been exchanged. Figure legend 1 refers to figure 2 (MRI), and figure legend 2 refers to figure 1 (EUS).














