Serviços Personalizados
Journal
Artigo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Acessos
Acessos
Links relacionados
-
 Similares em
SciELO
Similares em
SciELO
Compartilhar
GE-Portuguese Journal of Gastroenterology
versão impressa ISSN 2341-4545
GE Port J Gastroenterol vol.24 no.3 Lisboa jun. 2017
https://doi.org/10.1159/000452692
REVIEW ARTICLE
Acute Pancreatitis Etiology Investigation: A Workup Algorithm Proposal
Investigação Etiológica da Pancreatite Aguda: Proposta de Protocolo de Atuação
Sílvia da Silva, Mónica Rocha, João Pinto-de-Sousa
General Surgery Department, Centro Hospitalar do Tâmega e Sousa, Penafiel, Portugal
* Corresponding author.
ABSTRACT
Background: Acute pancreatitis represents a significant number of hospital admissions. Most of the patients are admitted in an acute setting. Early identification of its etiology is an essential step toward the rational approach, both for its implications in the immediate therapy and the prevention of recurrence. Although often obvious, the etiological workup of acute pancreatitis can be challenging. Conclusion: There are several studies emphasizing the multiple etiologies underlying acute pancreatitis but lacking structured diagnostic workups to allow a rational and organized study. The main goal of this work is to develop an algorithm proposal, which aims to serve as a guide for the investigation of the etiology of acute pancreatitis based on a review of already published literature, adjusted to the reality of our hospital and the available resources.
Keywords: Acute disease; Pancreatitis; Etiology
RESUMO
Introdução: A pancreatite aguda é responsável por um número significativo de internamentos hospitalares. A maioria dos doentes é admitido pelo Serviço de Urgência. A identificação precoce do fator etiológico é essencial, quer pelas suas implicações na terapêutica imediata, quer na prevenção das recorrências. Apesar de frequentemente linear, a investigação etiológica pode constituir um desafio considerável. Conclusão: Vários são os estudos que enfatizam as múltiplas etiologias, no entanto são raras orientações diagnósticas estruturadas que permitam encetar um estudo racional e orientado por critérios bem estabelecidos. O objetivo deste trabalho é a elaboração de um protocolo de atuação, que pretende servir como um guia para a investigação etiológica das pancreatites agudas, baseado numa revisão dos trabalhos publicados, adaptada à realidade deste Hospital e meios disponíveis.
Palavras-Chave: Doença aguda; Pancreatite; Etiologia
Introduction
The incidence of acute pancreatitis (AP) has been rising over the years in western countries [1–3] and, in fact, this disease represents a significant cause of morbidity and mortality regardless of its etiology [2].
This increasing incidence seems to be related to several factors. On the one hand, life expectancy has risen, and the mean age of the first episode reaches his peak around the sixth decade. On the other hand, in this century, we face the obesity epidemic, and, as it is known, overweight is an individual risk factor for biliary gallstones, one of the main causes of AP [1].
The effort to identify the cause of AP is a worthy process mainly for 2 reasons: to guide the therapy in the acute setting and to prevent recurrences [3]. In fact, the overall relapse risk is very high, reaching 50% for alcoholic pancreatitis and 32–61% for nontreated gallstones after the initial episode [4].
According to the guidelines published in 2007 by the American Gastroenterological Association [2], the diagnostic etiology should be established in at least 75% of the cases.
It is therefore meaningful to establish an etiological diagnosis in order to reduce the morbidity and mortality of this disease so prevalent among us.
In the Centro Hospitalar do Tâmega e Sousa, which is a tertiary center in the north of Portugal, resources are narrow. It is located in a rural area, with a high prevalence of gallstone disease and a high alcohol intake. Annually, about 250 admissions for AP are registered, making this one of the most common admission diagnoses. Although often simple, in some cases a clarifying etiology can be challenging, highlighting the need to adopt reasonable means based on literature evidence in order to achieve it in a cost-effective manner.
Etiological Factors
Several causes can lead to an acute inflammatory process in the pancreas (Table 1). Among those identified, gallstone disease and alcohol abuse predominate in western countries and together are responsible for 70–80% of all cases. [1, 5–8] However, the risk of developing AP in the presence of these factors is extremely low, which suggests that other factors might be involved and emphasizes the role of individual susceptibility [4]. Whenever investigating AP etiology, and after the exclusion of these 2 main causes, less frequent factors must be considered. In fact, in the course of the last decade, other numerous causes have been identified, representing as a whole about 10% of all cases, among which the following stand out: anatomic and functional abnormalities, metabolic conditions, drugs, trauma, infections, and vascular and genetic causes.

It is a matter of fact that, despite all efforts with a thorough investigation, that in about 10% of the episodes it is not possible to achieve a causal factor, and therefore these must be classified as idiopathic (Table 2). Idiopathic AP remains a challenge, and it is expected that in 10% of the cases of a single episode, and in more than 30% of the recurrent ones, the etiology remains unknown [5].
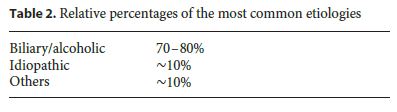
Etiological Workup
Reasonable efforts to obtain an etiological diagnosis must be undertaken during the hospitalization period for AP, with particular focus on the identification of causes that may influence the approach in the acute phase.
In this respect, investigation should start by a detailed clinical history and a complete physical examination, which should lead clinicians to identify or to exclude the most frequent causes [9]. Relevant information must include a previous diagnosis of biliary disease or gallstones, former cholecystectomy, history of alcohol or drug abuse, personal history of dyslipidemia, recent abdominal surgery, trauma or endoscopic retrograde cholangiopancreatography (ERCP), the presence of consumptive symptoms suggesting malignancy, or a family history of recurrent pancreatitis.
However, despite the information obtained with this simple inquiry, investigation should always aim at excluding the 2 most frequent causes: gallstones and alcohol.
Current guidelines recommend blood samples to test for liver function (bilirubin, transaminases, and alkaline phosphatase), calcium, and triglycerides for all patients during the first 24 h after admission. Similarly, abdominal ultrasound (US), given its unquestionable value in achieving an etiology rather than just a diagnosis of AP, should be performed in all patients upon admission [2, 8, 10].
Serum levels of triglycerides should be measured at an initial phase of the disease. Rather than representing a major cause of AP, it is to be remembered that these levels sharply decrease after 24–48 h of fasting [11]. Therefore, as a direct consequence, the diagnosis may be difficult to establish. In order to consider hypertriglyceridemia as the most probable etiology of AP, values above 1,000 mg/dL must be present [4, 5, 11]
The main purpose of serum calcium evaluation should be considered a prognostic tool rather than a diagnostic one. Indeed, a comparison between the basal values of serum calcium and those obtained at 48 h can help stratifying AP gravity according to the Ranson criteria [11].
The demonstration of gallstones or choledocolithiasis makes a biliary etiology probable, and values of aspartate alanine transferase >150 IU/L during the first 48 h seem to have a positive predictive value higher than 85% for this condition [9]. Whenever US performed at admission is inconclusive, all efforts shall be done to do it again. Repeating the examination might increase its sensitivity for microlithiasis (stones <3 mm) identification, which is only 50% at the initial workup [12, 13]. Indeed, US can be done in the follow-up context after patient discharge.
The presence of alcohol-drinking habits should never be forgotten, particularly in some geographic areas. Although the exact mechanism inducing pancreatic injury is not well understood, the link between AP and alcohol has been known for a long time [11]. It is assumed that individuals with heavy drinking (>60 g/day) are at a greater risk for developing AP; therefore, this etiology should always be considered in such individuals. Some authors argue that continued consumption over several years is required [4], though others state that a single large intake will be enough to trigger the inflammatory process [11]. Similarly, it is common knowledge that heavy consumption is a risk factor for other diseases, namely gallstones, obesity (in turn a risk factor for gallstones), and hypertriglyceridemia. In some patients these factors might coexist and so it may not be easily possible to securely identify the causal agent. In these circumstances more than 1 etiologic diagnosis should be considered.
Whenever gallstones, alcohol and dyslipidemia can be securely ruled out, the investigation must be directed to other, less prevalent causes of AP. Extensive or invasive investigations are not recommended for patients at a first episode and l<40 years of age [1, 2]. Whenever a diagnosis is not achieved after the first episode of AP and after conclusion of the basic study, patients should be classified as having an undetermined or unclear etiology. Indeed, trying to catalogue these cases as idiopathic seems to be inappropriate at this time, mainly because an extensive study has not been carried out yet. For patients without an identifiable cause, although in the context of a single episode of AP, if they are older than 40 years, it is recommended to proceed to an abdominal CT scan due to the
underlying risk of malignancy [2]. More aggressive investigation is advised regardless of patient age in those suffering more than 1 episode of AP, taking into consideration the greater risk of morbidity and possible progression to chronic pancreatitis [3].
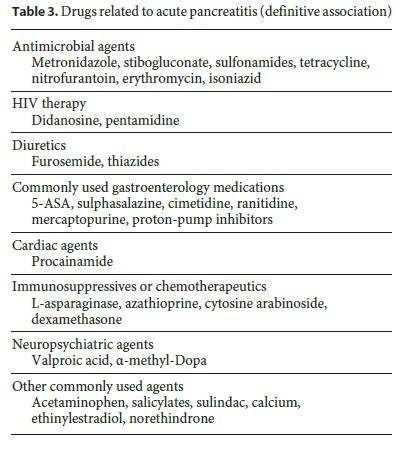
As shown in Table 3, several drugs are able to induce AP. Those with a higher incidence and well-established relationship are azathioprine, mercaptopurine, and didanosine. Most drugs act by an idiosyncratic effect or by direct toxicity. With others, such as valproic acid or didanosine, episodes typically occur weeks to months after starting the therapy, which suggests that the mechanism appears to be linked to toxic metabolite accumulation. Hypersensitivity reactions that usually develop after the first month seem to be linked to drugs such as azathioprine, mercaptopurine, metronidazole, aminosalicylates, and sulphonamides [3].
To the best of our knowledge, a clear cause-effect relation between infection and AP has not yet been established. Moreover, facing the multiple agents connected to the AP etiology (Table 4), a routinely exhaustive investigation is not advised, unless a strong clinical suspicious exists [3]. AP in connection with HIV is well established, occurs frequently, and often has an increased risk of event-related disease progression. In this particular case, 2 mechanisms seem to be engaged in AP: the first one is the infectious process itself and the subsequent patient immunosuppression, and the second one is the antiretroviral therapy, which is one of the most frequent causes of drug-induced pancreatitis [3, 4]. Another infectious agent, Ascaris , represents the second-leading cause of AP in India, and it should therefore be valued within the adequate epidemiological context [3].

When facing recurrent episodes of AP, an autoimmune etiology should always be considered, and though representing a rare condition, it is an important basis of chronic pancreatitis. Elevated serum IgG4 levels, antibodies to carbonic anhydrase II antigens, and lactoferrin are some markers of this disorder [3]. Quite often, this condition appears either as pancreatic or biliary stenosis, or in a focal form, therefore mimicking malignancy.
When facing a differential diagnosis with malignancy, by adopting a cutoff for IgG4 >135 mg/dL, sensitivities and specificities of 95 and 97%, respectively, can be achieved, which almost enables the diagnosis [5]. In these circumstances and after excluding malignancy, it is essential to start with corticoid-based directed therapy.
Hereditary pancreatitis, another rare condition, is supposed to be suspected whenever the first episode arises inyoung patients (typically <20 years), often with a family history of recurrent pancreatitis going back at least 2 consecutives generations. Several genetic mutations have been associated with episodes of AP. Mutation of the cystic fibrosis transmembrane conductance regulator (CFTR) gene is associated with multiple conditions related to cystic fibrosis, including pancreatitis, although the exact mechanism is still largely unknown [3, 5]. Mutation in the gene encoding the serine protease inhibitor Kazal type 1 (SPINK1) , a pancreatic trypsin inhibitor, has also been linked to recurrent pancreatitis, although it seems to be a co-factor that lowers the threshold for triggering mechanisms of AP related to other factors rather than the cause. Furthermore, mutations of the gene encoding cationic trypsinogen (PRSS1) are linked to the disease, following an autosomal dominant inheritance with 80% penetrance [3, 5]. Despite this evidence, the indication for the genetic study remains controversial. In fact, after obtaining the diagnosis, not much can be offered to patients in terms of treatment, prevention, or delay of progression. Under these circumstances, at present, a search of genetic alterations associated with episodes of AP is not routinely recommended [2]. Whenever other possible causes can be excluded, the presence of a suggestive family history will be sufficient for the diagnosis. If any doubt persists, genetic testing might be endorsed as a way to avoid other, more invasive tests.
If the etiologic investigation taken this far allows us to exclude the aforementioned causes and despite this diagnosis still remains occult, 2 major essential groups must be considered: biliopancreatic anatomic alterations and microlithiasis. Indeed, according to some studies, microlithiasis is assumed to be the most frequent cause of AP initially labeled as undetermined, with a frequency that, although variable, can be as high as 80% in some series [3, 14, 15].
If not performed yet, a CT scan should be done at this stage of the etiology investigation, mainly focused on structural or anatomic alterations, which, if present, will be very useful in guiding subsequent studies.
In ordinary conditions, 3 diagnostic tests should be considered directed to the biliary tract study with ERCP, magnetic resonance cholangiopancreatography (MRCP), and endoscopic ultrasonography (EUS). Because of its inherent complications, high costs, and associated radiation dose as well as by the emergence of less expensive and less invasive procedures, ERCP is currently playing a major role in a therapeutic setting rather than in the traditional diagnostic approach [16, 17].
EUS and MRCP diagnostic accuracy seems to be redundant overlapping [17]. EUS sensitivity seems to be higher regarding the diagnosis of microlithiasis. Indeed, the stones EUS can detect (as small as 0.1 mm) are much smaller than those identified by MRCP (usually not smaller than 1.5 mm) [17]. Besides that, EUS avoids gas or abdominal fat interposition, important determinants of the effectiveness of traditional US, and therefore allows better visualization of the distal bile duct [17, 18]. Additionally, the sensitivity of EUS to identify pancreatic masses as small as 2–3 cm is very high (about 95%) [18, 19]. Compared to MRCP, EUS owns the advantage of being a dynamic examination but is also highly operatordependent and invasive. Furthermore, due to technical limitations, it is more difficult to visualize the intrahepatic tree, and, indeed, is not free of complications too and is available only in a small number of very specialized centers. MRCP has the advantage of being completely noninvasive and widespread, therefore easily accessible, allowing the study of both extra- and intrahepatic bile ducts. Sensitivity and specificity values for stone detection overlap those achieved by ERCP. However, its sensitivity for the detection of masses, chronic pancreatitis, and microlithiasis is smaller than with EUS [16, 17].
According to guidelines published by the IAP/APA group in 2013 [9], after an initial inconclusive study (including 2 negative US), EUS should be the next step in the investigation, mainly due to its higher sensitivity detecting microlithiasis. Nevertheless, when comparing MRCP and EUS, no statistically significant differences were found [17]. MRCP might probably be preferred in highrisk patients in whom the invasive nature of EUS is not indicated [17], but in general, the option will depend on the examination accessibility and available experience for its interpretation.
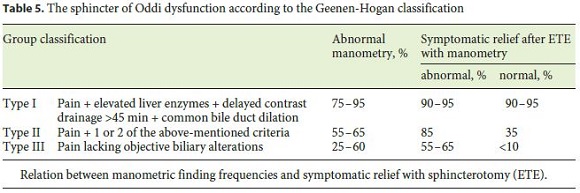
By this far on investigations, virtually all anatomical promoters of AP phenomena have been identified, such as anomalous biliopancreatic duct junction, pancreas divisum, neoplasms, and lithiasis not visible on US. Despite this, functional alterations of the sphincter of Oddi (SOD) might yet be missed, which can represent 25–65% of the idiopathic pancreatitis. According to some studies, the gold standard test for the SOD is manometry [3, 20, 21]. So far, this test is not available, but whenever possible, diagnosing these functional abnormalities allows the rational treatment, consisting of sphincterotomy. However, as stated by some available data, results are not uniform for all types of dysfunctions, that is, symptomatic relief ranges between 90–95% in type I SOD, 85% in type II, and 55–60% in type III (Table 5) [21]. Furthermore, in these patients, many complications can occur after ERCP, including pancreatitis, possibly due to the sphincter hypertonicity [20, 21]. In an effort to minimize these consequences, some noninvasive strategies such as low-fat diet and specific medication might be undertaken in order to reduce the biliopancreatic stimulation. The last category includes analgesics for the acute setting and nifedipine, nitrates, and antispasmodics to reduce basal sphincter pressure [3, 20]. Indeed, some studies concluded that nifedipine could achieve symptomatic relief in about 75% of the patients with SOD. In view of the benign nature of the disease and the safety of the mentioned drugs, medical therapy should be the first-line treatment of patients with suspected type III SOD and mild-to-moderate type II before adopting invasive measures [21]. ERCP with manometry and sphincterotomy should be reserved for patients with highly debilitating symptoms and without response to the previous measures [21].
Until now, neither EUS nor MRCP seemed to be of any significance in the diagnosis of SOD, but recently, the addition of secretin to MRCP has offered promising results according to some studies [5, 22–24]. Intravenous delivery of secretin promotes the stimulation and consequent production of pancreatic enzymes, and thus, by highlighting a delay in ducts emptying secondary to SOD, allows stressing their persistent dilation, thereby making diagnosis possible in an indirect way. Diagnosis is made in situations where the diameter of the main pancreatic duct, recorded 15 min after stimulation with secretin, exceeds 1 mm in comparison to the baseline values [22]. In addition, secretin allows a better definition of the biliopancreatic duct system in both tests, MRCP and EUS [23, 24]. This technique seems promising; however, the addition of secretin to MRCP is not yet accepted as a substitute for manometry of the sphincter of Oddi, which is mainly due to the numerous false-negative results reported in some data [22, 25].
Conclusions and Algorithm Proposal
As previously stated, guiding an etiologic study for AP depends on multiple factors, which include the frequency of each cause, personal history, epidemiology, and available resources.
Dealing with the reality of the Centro Hospitalar do Tâmega e Sousa and based on the available data presented before, the authors propose the algorithm introduced in Figure 1 for the etiological study of AP.
This paper aims to represent a general guiding for the study of the AP etiology and should be flexible enough to adjust to each patient. We recommend MRCP for bile duct study, taking into account its greater availability and lower invasiveness facing EUS.
References
1 Spanier BW, Dijkgraaf MG, Bruno MJ: Epidemiology, aetiology and outcome of acute and chronic pancreatitis: an update. Best Pract Res Clin Gastroenterol 2008;22:45–63. [ Links ]
2 American Gastroenterological Association (AGA) Institute on Management of Acute Pancreatits Clinical Practice and Economics Committee; AGA Institute Governing Board: AGA Institute medical position statement on acute pancreatitis. Gastroenterology 2007;132:2019–2021. [ Links ]
3 Lee JK, Enns R: Review of idiopathic pancreatitis. World J Gastroenterol 2007;13:6296–313. [ Links ]
4 Sekimoto M, Takada T, Kawarada Y, Hirata K, Mayumi T, Yoshida M, et al: JPN Guidelines for the management of acute pancreatitis: epidemiology, etiology, natural history, and outcome predictors in acute pancreatitis. J Hepatobiliary Pancreat Surg 2006;13:10–24. [ Links ]
5 Al-Haddad M, Wallace MB: Diagnostic approach to patients with acute idiopathic and recurrent pancreatitis, what should be done? World J Gastroenterol 2008;14:1007–1010. [ Links ]
6 Steer ML, Perides G: Pathogenesis and pathophysiology of acute pancreatitis; in: Beger HG, Matsuno S, Cameron JL: Diseases of the Pancreas: Current Surgical Therapy. Berlin Heidelberg, Springer-Verlag, 2008; pp 153–162. [ Links ]
7 Wang GJ, Gao CF, Wei D, Wang C, Ding SQ: Acute pancreatitis: etiology and common pathogenesis. World J Gastroenterol 2009;15:1427–1430. [ Links ]
8 Pongprasobchai S, Thamcharoen R, Manatsathit S: Changing of the etiology of acute pancreatitis after using a systematic search. J Med Assoc Thai 2009;92:S38–S42. [ Links ]
9 Working Group IAP/APA Acute Pancreatitis Guidelines: IAP/APA evidence-based guidelines for the management of acute pancreatitis. Pancreatology 2013;13:e1–e15. [ Links ]
10 Banks PA, Freeman ML; Practice Parameters Committee of the American College of Gastroenterology: Practice guidelines in acute pancreatitis. Am J Gastroenterol 2006;101:2379–2400. [ Links ]
11 Nordback I, Sand J, Andren-Sandberg A: Criteria for alcoholic pancreatitis. Results of an international workshop in Tampere, Finland, June 2006. Pancreatology 2007;7:100–104. [ Links ]
12 Signoretti M, Baccini F, Piciucchi M, Iannicelli E, Valente R, Zerboni G, et al: Repeated transabdominal ultrasonography is a simple and accurate strategy to diagnose a biliary etiology of acute pancreatitis. Pancreas 2014;43:1106–1110. [ Links ]
13 Working Party of the British Society of Gastroenterology; Association of Surgeons of Great Britain and Ireland; Pancreatic Society of Great Britain and Ireland; Association of Upper GI Surgeons of Great Britain and Ireland: UK guidelines for the management of acute pancreatitis. Gut 2005;54:iii1–ii9. [ Links ]
14 van Brummelen SE, Venneman NG, van Erpecum KJ, VanBerge-Henegouwen GP: Acute idiopathic pancreatitis: does it really exist or is it a myth? Scand J Gastroenterol Suppl 2003;239: 117–122. [ Links ]
15 Elta GH: Sphincter of Oddi dysfunction and bile duct microlithiasis in acute idiopathic pancreatitis. World J Gastroenterol 2008;14:1023–1026. [ Links ]
16 Hurter D, De Vries C, Potgieter PH: Accuracy of MRCP compared with ERCP in the diagnosis of bile duct disorders. SA J Radiol 2008;12:14–22. [ Links ]
17 Verma D, Kapadia A, Eisen GM, Adler DG: EUS vs MRCP for detection of choledocholithiasis. Gastrointest Endosc 2006;64:248–254. [ Links ]
18 Byrne MF, Jowell PS: Gastrointestinal imaging: endoscopic ultrasound. Gastroenterology 2002;122:1631–1648. [ Links ]
19 Rodrigues CG, Pereira E, Caldeira A, Santos A, Sousa H, Banhudo A: Ecoendoscopia digestiva na prática clínica parte IIa – utilidade na avaliação do pâncreas. GE J Port Gastrenterol 2014;21:60–74. [ Links ]
20 Somogyi L, Martin SP, Venkatesan T, Ulrich CD 2nd: Recurrent acute pancreatitis: an algorithmic approach to identification and elimination of inciting factors. Gastroenterology 2001;120:708–717. [ Links ]
21 Staritz M: Sphincter of Oddi physiology and pathophysiology; in Beger HG, Matsuno S, Cameron JL: Diseases of the Pancreas: Current Surgical Therapy. Berlin Heidelberg, Springer-Verlag, 2008, pp 55–58. [ Links ]
22 Kapetanos DJ: Endoscopic management of acute recurrent pancreatitis. Ann Gastroenterol 2010;23:31–37. [ Links ]
23 Mariani A, Arcidiacono PG, Curioni S, Giussani A, Testoni PA: Diagnostic yield of ERCP and secretin-enhanced MRCP and EUS in patients with acute recurrent pancreatitis of unknown aetiology. Dig Liver Dis 2009;41:753–758. [ Links ]
24 Hellund JC, Skattum J, Buanes T, Geitung JT: Secretin-stimulated magnetic resonance cholangiopancreatography of patients with unclear disease in the pancreaticobiliary tract. Acta Radiol 2007;48:135–141. [ Links ]
25 Mariani A, Curioni S, Zanello A, Passaretti S, Masci E, Rossi M, et al: Secretin MRCP and endoscopic pancreatic manometry in the evaluation of sphincter of Oddi function: a comparative pilot study in patients with idiopathic recurrent pancreatitis. Gastrointest Endosc 2003;58:847–852. [ Links ]
Disclosure Statement
The authors have no conflicts of interest to declare.
* Corresponding author.
Dr. Sílvia da Silva
General Surgery Department, Centro Hospitalar do Tâmega e Sousa
Lugar do Tapadinho, Guilhufe
PT–4564-007 Penafiel (Portugal)
E-Mail silviaraquelcsilva@gmail.com
Received: June 29, 2016; Accepted after revision: September 26, 2016














