Serviços Personalizados
Journal
Artigo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Acessos
Acessos
Links relacionados
-
 Similares em
SciELO
Similares em
SciELO
Compartilhar
GE-Portuguese Journal of Gastroenterology
versão impressa ISSN 2341-4545
GE Port J Gastroenterol vol.24 no.6 Lisboa dez. 2017
https://doi.org/10.1159/000461589
CLINICAL CASE STUDY
Type 2 Autoimmune Pancreatitis: A Challenge in the Differential Diagnosis of a Pancreatic Mass
Pancreatite Autoimune Tipo 2: Desafio no Diagnóstico Diferencial de uma Massa Pancreática
Cláudio Martinsa, Paula Lagoc, Paula Sousab, Tarcísio Araújoc, José Davided,e, Fernando Castro-Poçasc–e, Isabel Pedrotoc,e
aDepartment of Gastroenterology, Centro Hospitalar de Setúbal – Hospital de São Bernardo, Setúbal, bDepartment of Gastroenterology, Centro Hospitalar Tondela-Viseu – Hospital de São Teotónio, Viseu, and cDepartment of Gastroenterology, and dHepatobiliopancreatic Unit, Centro Hospitalar do Porto – Hospital Geral de Santo António, and eInstitute of Biomedical Sciences Abel Salazar, University of Porto, Porto, Portugal
* Corresponding author.
ABSTRACT
Introduction: Autoimmune pancreatitis is a rare entity of unknown etiology that can mimic pancreatic cancer and whose diagnosis involves clinical, serological, imagiological, and histological findings. There are two types of autoimmune pancreatitis: type 1, in which the pancreas is involved as one part of a systemic immunoglobulin G4-related disease, and type 2, generally without immunoglobulin G4-positive cells and without systemic involvement. Case: We report the case of a 45-year-old female, who underwent an abdominal magnetic resonance imaging for etiological study of a solid liver lesion, which revealed a tail pancreatic mass. Laboratory analyses showed normal levels of immunoglobulin G4 and negative antinuclear antibodies. Endoscopic ultrasound revealed a homogeneous and hypoechogenic lesion in the pancreatic tail with a sausage-like appearance. Endoscopic ultrasound-guided fine needle aspiration was inconclusive and the patient underwent a laparoscopic distal pancreatectomy. Histopathology examination confirmed the diagnosis of type 2 autoimmune pancreatitis. Conclusion: This case highlights the challenge in the diagnostic approach of a pancreatic mass, particularly in distinguishing benign from malignant disease.
Keywords: Autoimmune pancreatitis, Immunoglobulin G4, Pancreatic cancer
RESUMO
Introdução: A pancreatite autoimune é uma entidade rara, de etiopatogenia desconhecida, que pode simular cancro do pâncreas e cujo diagnóstico envolve a integração de dados clínicos, serológicos, imagiológicos e histológicos. Descrevem-se dois tipos de pancreatite autoimune: tipo 1, cujo envolvimento pancreático integra o espetro das doenças associadas à imunoglobulina G4, e tipo 2, geralmente sem evidência de células imunoglobulina G4 positivas e sem manifestações sistémicas. Caso: Apresentamos o caso de uma mulher de 45 anos, submetida a ressonância magnética abdominal para esclarecimento etiológico de lesão nodular hepática que revelou uma massa na cauda do pâncreas. Analiticamente apresentava imunoglobulina G4 normal e anticorpos antinucleares negativos. A ultrassonografia endoscópica revelou uma lesão homogénea e hipoecogénica na cauda pancreática com morfologia em salsicha. A punção aspirativa por agulha fina foi inconclusiva tendo a doente sido submetida a pancreatectomia distal por via laparoscópica. O exame histopatológico confirmou o diagnóstico de pancreatite autoimune tipo 2. Conclusão: Este caso destaca o desafio na abordagem diagnóstica da massa pancreática, particularmente na diferenciação entre patologia benigna e maligna.
Palavras-Chave: Pancreatite autoimune, Imunoglobulina G4, Cancro pancreático
Introduction
Autoimmune pancreatitis (AIP) is a fibro-inflammatory disease that accounts for 4.6–5% of chronic pancreatitis and comprises two entities, currently designated as type 1 and type 2, which have unique histopathological patterns and differ significantly in their demographic profiles, clinical presentation, and natural history [1, 2]. Type 1 has been recognized as the pancreatic manifestation of a multiorgan disease, named immunoglobulin G4 (IgG4)-related disease, while type 2 is a pancreatic specific disorder not associated with IgG4.
The diagnostic criteria for AIP are a combination of imagiological, laboratorial, and histopathological findings. International criteria for the diagnosis of AIP have been defined, such as the HISORt (histology, imaging, elevated serum IgG4 levels, other organ involvement, and response to steroids) criteria [3] from the Mayo Clinic and, most recently, International Consensus Diagnostic Criteria from International Association of Pancreatology [4]. Despite this, the definitive diagnosis of AIP, particularly type 2, is challenging, as this disorder commonly presents as a pancreatic mass mimicking pancreatic cancer. Misdiagnosis has the potential to be catastrophic as an undiagnosed cancer may cause delay or loss of the opportunity for potential curative surgery. The opposite scenario of a pancreatic surgery performed for benign disease, with its high risk of morbidity and mortality, is also unsatisfactory.
Herein, we report an uncommon case of type 2 AIP presented as a pancreatic mass illustrating the challenge in the differential diagnosis between AIP and pancreatic cancer.
Case Report
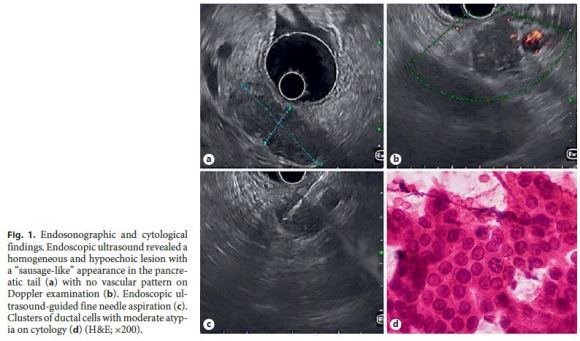
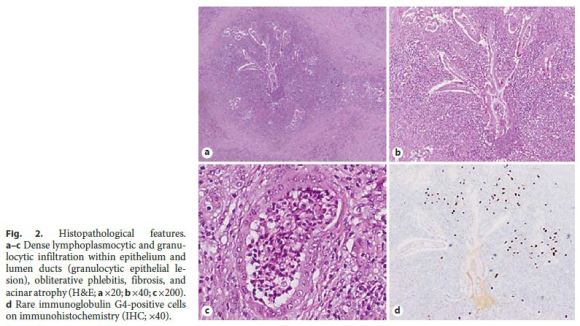
A 45-year-old Caucasian female underwent an abdominal magnetic resonance imaging (MRI) for etiological study of a solid liver lesion (focal nodular hyperplasia), detected on routine abdominal ultrasound, which revealed a tail pancreatic solid lesion of undetermined etiology. She had a medical history of arterial hypertension and ulcerative colitis treated with bisoprolol and mesalazine. She reported no history of smoking or alcohol consumption. There was no evidence of fever, jaundice, abdominal pain, anorexia, weight loss, or other gastrointestinal symptoms. Physical examination was unremarkable. Laboratory analysis revealed no anemia and liver tests, amylase, lipase, C reactive-protein, erythrocyte sedimentation rate, CA 19-9, and IgG4 were normal. Antinuclear antibodies were negative. In order to clarify the nature of the referred lesion, an endoscopic ultrasound (EUS) was performed, which revealed a homogeneous and hypoechogenic lesion, with 40 mm of greater diameter, in the pancreatic tail with a sausage-like appearance (Fig. 1a). This lesion showed no vascular pattern on Doppler examination (Fig. 1b). There were no pancreatic calcifications and common biliary duct, pancreatic duct, and splenoportal axis were normal. An EUS-guided fine needle aspiration (FNA) with a 19-gauge needle was performed (Fig. 1c), whose cytological analysis showed abundant inflammatory infiltrate and ductal epithelium with mild to moderate atypia, apparently reactive (Fig. 1d). Given that the pancreatic adenocarcinoma hypothesis could not be excluded, the patient underwent a laparoscopic distal pancreatectomy. Histopathology examination confirmed the diagnosis of a type 2 autoimmune pancreatitis (Fig. 2). At the 6-month follow-up, the patient remained asymptomatic with no evidence of recurrence.
Discussion
A rare form of idiopathic chronic pancreatitis was first described in 1961 by Sarles et al. [5]. The term autoimmune pancreatitis was introduced by Yoshida et al. [6] in 1995 to describe a steroid-responsive disease associated with autoimmune features. In 2009, two subtypes of AIP, called type 1 (lymphoplasmacytic sclerosing pancreatitis) and type 2 (idiopathic duct-centric pancreatitis), were formally recognized [1, 2].
AIP is a rare disorder, with a reported prevalence in Japan of 0.82/100,000. Concerning subtypes, type 1 is the most prevalent worldwide while type 2 is more common in Europe and North America. In an international study of 1,064 patients, the average age at the time of diagnosis was 61.4 and 39.9 years for types 1 and 2, respectively, with a proportion of male patients significantly higher in type 1 (77 vs. 55%) [7].
The clinical picture of AIP depends on its subtype. Type 1 is more prevalent among older males and belongs to the IgG4-related systemic disease, which is associated with high serum and/or tissue IgG4 and other autoimmune disorders, including IgG4 cholangitis, sclerosing sialadenitis, interstitial nephritis, and retroperitoneal fibrosis. The classic clinical presentation in type 1 is painless obstructive jaundice (up to 75% of cases) mimicking pancreatic cancer. Other clinical features include chronic or recurrent abdominal pain (68%), acute pancreatitis, and steatorrhea. On the other hand, type 2 AIP affects mainly younger patients, without a gender predilection, and often is not associated with hyper-IgG4. As in type 1, type 2 also frequently presents with obstructive jaundice (50% of cases) but without systemic involvement, except for inflammatory bowel disease which is present in about 30% of cases, particularly ulcerative colitis [8].
Overall, the pancreatic disease in patients with AIP can present in different ways including (1) a focal mass or diffuse enlargement on imaging that can be confused with pancreatic cancer (85% of cases), (2) mild abdominal pain with or without acute or chronic pancreatitis, (3) strictures of the pancreatic duct, and (4) peripancreatic vascular complications (23%) [9]. We report a case of a solid pancreatic mass incidentally detected on abdominal MRI in an asymptomatic young female patient with history of ulcerative colitis.
Diagnosis of AIP is challenging, particularly in type 2. Correct diagnosis can help avert the consequences of progressive disease and unnecessary surgery. In the last decade, several diagnostic criteria were proposed including clinical, serological, imagiological, and histological findings. In order to unify the heterogeneity of diagnostic criteria, a multinational group convened in 2011 and developed International Consensus Diagnostic Criteria for AIP [4].
Due to its ability to provide high-resolution imaging, EUS emerged as an important diagnostic tool. Most common endosonographic findings are focal or diffuse sausage-shaped pancreatic enlargement, homogeneous echo pattern, stranding, and calcifications. A long and/or multiple strictures of the pancreatic duct, without significant associated dilation, is also highly characteristic of AIP. The other advantage of EUS is the possibility to perform FNA or core biopsy, which provides tissue samples for cytological or histological diagnosis. Despite the poor sensitivity (up to 37.5%) and specificity of EUSguided FNA, the recent advent of spring-loaded biopsy needles provides a high diagnostic accuracy (about 85%) [10, 11]. Elastography and contrast-enhanced EUS are newer noninvasive technologies that may help differentiate benign from malignant solid pancreatic lesions. Although both of these techniques may increase the diagnostic yield of EUS, there are a considerable number of issues that remain to be solved, requiring additional studies.
The definitive diagnosis of type 2 AIP always requires histology, revealing an idiopathic duct-centric pancreatitis, the hallmarks of which are granulocytic epithelial lesions. IgG4 plasma cells are absent or in small number. Obliterative phlebitis and fibrosis are less prominent than in type 1 [1, 2].
Unlike other forms of pancreatitis, AIP is highly responsive to steroid therapy [13]. The diagnosis of AIP should be reconsidered in patients who do not respond to steroids. The relapse rate in patients with type 1 AIP ranges from 30–60%, while patients with type 2 typically do not relapse (<5%) [14]. Immunosuppressors, such as azathioprine or mycophenolate mofetil, are used in patients with relapse or steroid-resistant disease. Spontaneous remission is seen in 24–55% of patients [12, 13]. In spite of this, early steroid therapy is recommended because, if untreated, the pancreatic and biliary disease can progress to irreversible pancreatic insufficiency and secondary biliary cirrhosis.
In this case, the patient underwent EUS and the ultrasonographic features were suggestive of AIP. Although the clinical, laboratory, and imaging findings favor the diagnosis of autoimmune pancreatitis, the hypothesis of pancreatic cancer could not be completely excluded dueto the presence of moderate ductal atypia on cytology. Therefore, the case was discussed at a multidisciplinary meeting, and, in order to definitively rule out a more ominous lesion, a surgical approach was decided, allowing a definitive diagnosis.
In conclusion, this report describes an uncommon entity highlighting the challenge in the diagnostic approach of a pancreatic mass. Although the diffuse form of AIP can be easily distinguished from pancreatic cancer on imaging, differentiating focal AIP from pancreatic malignancy is challenging, like in the presented case. Making the correct diagnosis and differentiating AIP from pancreatic cancer is of the utmost importance; an agreed diagnostic pathway should be in place and a multidisciplinary approach taken with each patient.
References
1 Kloppel G, Detlefsen S, Chari ST, et al: Autoimmune pancreatitis: the clinicopathological characteristics of the subtype with granulocytic epithelial lesions. J Gastroenterol 2010;45:787–793. [ Links ]
2 Zhang L, Chari ST, Smyrk TC, et al: Autoimmune pancreatitis (AIP) type 1 and type 2: an international consensus study on histopathologic diagnostic criteria. Pancreas 2011;40:1172–1179. [ Links ]
3 Chari ST, Smyrk TC, Levy MJ, et al: Diagnosis of autoimmune pancreatitis: the Mayo Clinic experience. Clin Gastroenterol Hepatol 2006;4:1010–1016. [ Links ]
4 Shimosegawa T, Chari ST, Frulloni L, et al: International consensus diagnostic criteria for autoimmune pancreatitis: guidelines of the International Association of Pancreatology. Pancreas 2011;40:352–358. [ Links ]
5 Sarles H, Sarles JC, Muratore R, et al: Chronic inflammatory sclerosis of the pancreas – an autonomous pancreatic disease. Am J Dig Dis 1961;6:688–698. [ Links ]
6 Yoshida K, Toki F, Takeuchi T, et al: Chronic pancreatitis caused by an autoimmune abnormality. Proposal of the concept of autoimmune pancreatitis. Dig Dis Sci 1995;40:1561–1568. [ Links ]
7 Hart PA, Kamisawa T, Brugge WR, et al: Long-term outcomes of autoimmune pancreatitis: a multicentre, international analysis. Gut 2013;62:1771–1776. [ Links ]
8 Kamisawa T, Chari ST, Lerch MM, et al: Recent advances in autoimmune pancreatitis: type 1 and type 2. Gut 2013;62:1373–1380. [ Links ]
9 Raina A, Yadav D, Krasinskas AM, et al: Evaluation and management of autoimmune pancreatitis: experience at a large US center. Am J Gastroenterol 2009;104:2295–2306. [ Links ]
10 Mizuno N, Bhatia V, Hosoda W, et al: Histological diagnosis of autoimmune pancreatitis using EUS-guided trucut biopsy: a comparison study with EUS-FNA. J Gastroenterol 2009;44:742–750. [ Links ]
11 Kanno A, Ishida K, Hamada S, et al: Diagnosis of autoimmune pancreatitis by EUS-FNA by using a 22-gauge needle based on the International Consensus Diagnostic Criteria. Gastrointest Endosc 2012;76:594–602. [ Links ]
12 Kamisawa T, Shimosegawa T, Okazaki K, et al: Standard steroid treatment for autoimmune pancreatitis. Gut 2009;58:1504–1507. [ Links ]
13 Sah RP, Chari ST, Pannala R, et al: Differences in clinical profile and relapse rate of type 1 versus type 2 autoimmune pancreatitis. Gastroenterology 2010;139:140–148. [ Links ]
14 Ketwaroo GA, Sheth S: Autoimmune pancreatitis. Gastroenterol Rep (Oxf) 2013;1:27–32. Downloaded [ Links ]
Statement of Ethics
This study did not require informed consent nor review/approval by the appropriate ethics committee.
Disclosure Statement
The authors declare no conflicts of interest.
* Corresponding author.
Dr. Cláudio Martins
Department of Gastroenterology, Centro Hospitalar de Setúbal
Hospital de São Bernardo, Rua Camilo Castelo Branco
PT–2910-446 Setúbal (Portugal)
E-Mail cmartins1@campus.ul.pt
Received: October 10, 2016; Accepted after revision: February 3, 2017














