Serviços Personalizados
Journal
Artigo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Acessos
Acessos
Links relacionados
-
 Similares em
SciELO
Similares em
SciELO
Compartilhar
GE-Portuguese Journal of Gastroenterology
versão impressa ISSN 2341-4545
GE Port J Gastroenterol vol.27 no.3 Lisboa jun. 2020
https://doi.org/10.1159/000503010
CLINICAL CASE STUDY
Pancreas- and Pylorus-Preserving Duodenectomy for Advanced Familial Duodenal Polyposis
Duodenectomia com conservação do pâncreas e do piloro na polipose duodenal severa
Júlio S. Leitea, José Guilherme Tralhãoa, António Mansoa, Miguel Fernandesa, Inês Cunhab, Pedro Amarob
aServiço de Cirurgia Geral, Centro Hospitalar e Universitário de Coimbra, Coimbra, Portugal; bServiço de Gastrenterologia, Centro Hospitalar e Universitário de Coimbra, Coimbra, Portugal
* Corresponding author.
ABSTRACT
Most patients with familial adenomatous polyposis (FAP) will develop duodenal polyps and 5% progress to cancer. Those with Spigelman stage IV have a 36% risk of cancer at 10 years. Endoscopic surveillance is necessary with local ablation for early disease. Unresectable duodenal disease and severe dysplasia are an indication for prophylactic radical surgery by pancreaticoduodenectomy or pancreas-sparing duodenectomy. Some preliminary results have shown better outcomes with duodenectomy. A 45-year-old female with FAP had restorative proctocolectomy at 24 years, desmoid of the mesentery with regression after sulindac, two pregnancies, and at the age of 37 years had duodenal polyposis stage III carpeting the periampullary region. Endoscopic papillectomy and extensive piecemeal mucosectomy was performed but was unsuccessful due to recurrence. After 7 years of regular endoscopic surveillance, focal high-grade dysplasia was diagnosed at the last evaluation. Some diminutive polyps were seen in the small-bowel capsule endoscopy. MRCP showed a normal biliary and pancreatic duct without visualization of the Santorini duct. A pancreas and pylorus-preserving duodenectomy was performed with 3 main steps: (1) duodenectomy with preservation of the pancreas and the pylorus; (2) reconstruction with an advanced jejunal limb and duodenojejunostomy; (3) reimplantation of the biliary and pancreatic duct in the jejunal loop. The patient was discharged on the 11th postoperative day without complications. In conclusion, pancreas- and pylorus-preserving duodenectomy is a promising alternative to pancreaticoduodenectomy for advanced duodenal polyposis that allows complete endoscopic surveillance.
Keywords: Pancreas-sparing duodenectomy, Pancreaticoduodenectomy, Familial adenomatous polyposis, Duodenal polyposis
RESUMO
A maioria dos doentes portadores de polipose adenomatosa familiar (FAP) vem a desenvolver pólipos duodenais que poderão degenerar em 5% dos casos. Os casos que apresentem um estádio IV de Spigelman têm um risco de degenerescência de 36% ao fim de 10 anos. É necessária vigilância endoscópica e excisão das lesões iniciais. Os pólipos considerados irressecáveis e com displasia de alto grau têm indicação para exérese cirúrgica radical através de duodenopancreatectomia ou de duodenectomia com conservação do pâncreas. Existem alguns resultados preliminares a revelar melhores resultados com a duodenectomia. Uma doente de 45 anos portadora de FAP efetuou proctocolectomia reconstrutiva aos 24 anos. Desenvolveu tumor desmoide mesentérico após um ano e que regrediu com sulindac, teve dois filhos e aos 37 anos apresentou polipose duodenal, em toalha periampular, com estádio III. Foi submetida a papilectomia endoscópica e mucosectomia fragmentada da lesão circundante tendose verificado recorrência. Durante 7 anos procedeu-se a vigilância endoscópica regular com presença de displasia de alto grau focal na última avaliação. Na cápsula endoscópica foram observados alguns pólipos diminutos no intestino delgado. A CPRM revelou normalidade nos canais pancreático e biliar, sem evidência do Santorini. Foi efetuada uma duodenectomia com conservação do pâncreas e do piloro cujos passos cirúrgicos principais foram: (a) duodenectomia com conservação do pâncreas e do piloro; (b) reconstrução com ansa jejunal e duodenojejunostomia; (c) reimplantação dos canais biliar e pancreático à ansa jejunal. A doente teve alta ao 11º dia pós-operatório sem complicações. Em conclusão, a duodenectomia com conservação do pâncreas e do piloro constitui uma boa alternativa à duodenopancreatectomia permitindo vigilância endoscópica completa.
Palavras-Chave: Duodenectomia com conservação do pâncreas, Duodenopancreatectomia, Polipose adenomatosa familiar, Polipose duodenal
Introduction
Familial adenomatous polyposis (FAP) is characterized by the development of multiple adenomas along the gastrointestinal tract. The most frequent genetic mutations responsible for adenomatous polyposis are the APC mutation, associated with FAP, and the MYH mutations, the MUTYH-associated polyposis [1, 2].
Prophylactic proctocolectomy decreased the incidence of colorectal cancer in these patients, leaving desmoid tumors and duodenal cancers as the leading causes of death in FAP patients [3]. In contrast to colonic polyps, no more than 5% of duodenal polyps are likely to progress to duodenal malignancy [4]. The Spigelman classification [5] segregates patients based on the number, size, histology, and grade of dysplasia of the duodenal lesions (Table 1). The stages range from I to IV, with IV representing the most advanced stage, and 36% developed cancer at 10 years in a prospective study of 114 patients from St Marks Hospital [6]. Local endoscopic or surgical therapy may be appropriate for early disease, but with a high recurrence rate [7, 8]. Now, with lower mortality following pancreatic surgery and better understanding of duodenal disease in FAP as a result of surveillance, prophylactic resectional duodenal surgery has become a reasonable option. Pancreaticoduodenectomy or pancreas-sparing duodenectomy offer definitive therapy in preventing duodenal carcinoma. Although pancreas-sparing duodenectomy has been used less frequently, it offers the potential advantage of preserving a normal pancreas and some preliminary results are promising [9]. This surgical option was performed at our hospital, and as far as we know also in Portugal, for the first time.

Clinical Case
A 45-year-old female with FAP with no mutation found, but with a family history in her father and brothers, was submitted to prophylactic restorative proctocolectomy at 24 years. After 1 year she presented with abdominal pain and a 5-cm desmoid of the mesentery was diagnosed by CT scan. Partial regression was observed with sulindac. She had two pregnancies.
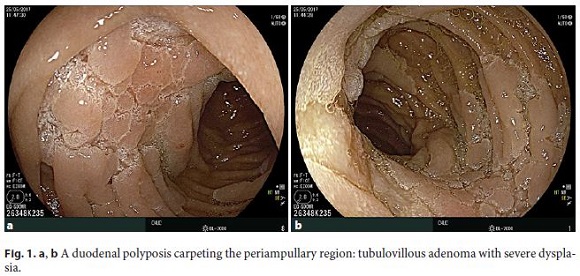
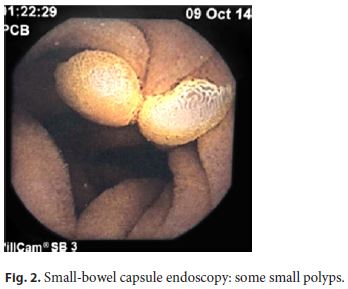
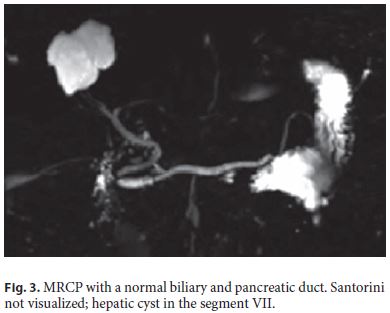
A duodenal polyposis stage III carpeting the periampullary region was observed when the patient was 37 years old. Endoscopic papillectomy and piecemeal mucosectomy was performed in one session; bleeding occurred during resection and was managed by coagulation with the tip of the snare and argon plasma. After the operation, no obvious bleeding or perforation was seen and the procedure was considered complete with no apparent residual adenoma. However, the early postprocedure period, about 12 h later, was complicated by hemorrhage originating from the resection scar. A decision to intervene surgically was made but a duodenotomy showed that the bleeding had stopped spontaneously and no hemostasis was needed. Unfortunately, endoscopic re-evaluation several months after surgery showed recurrent adenoma; due to the perceived risk of perforation in the context of fibrosis due to previous extensive mucosectomy, no further endoscopic resection was tried and a few attempts of ablation with argon plasma were also unsuccessful. Thereafter, regular endoscopic surveillance was maintained with the presence of a tubulovillous adenoma with low-grade dysplasia over several years but, eventually, high-grade dysplasia was diagnosed (Fig. 1a, b). Some diminutive polyps were also seen in the small-bowel capsule endoscopy (Fig. 2). The CT scan and the endoscopic ultrasonography did not demonstrate parietal or nodal involvement and the MRCP showed normal biliary and pancreatic ducts without visualization of the Santorini duct (Fig. 3). A pancreas- and pylorus-preserving duodenectomy was performed in 3 main steps (Fig. 4):
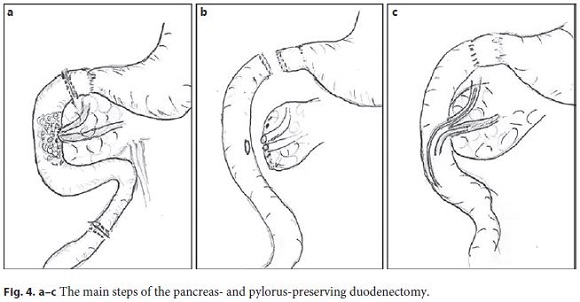
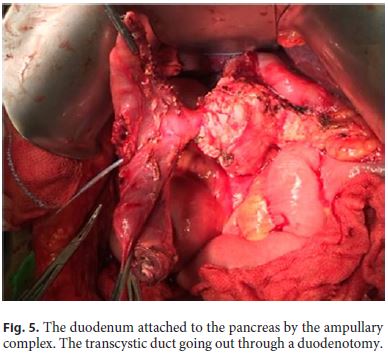
1. A cholecystectomy was carried out with a catheter introduced through the cystic duct to identify the papilla. The carpeting periampullary lesion was confirmed by duodenotomy without evidence of degeneration or other polyps in the proximal or distal duodenum and the transition to the jejunum was divided. The distal duodenum was progressively separated from the pancreas to the ampulla using Ligasure. The pylorus and the proximal duodenum were identified, separated from the pancreas without evidence of the Santorini duct. The duodenum was transected 2 cm distal to the pylorus. The dissection was completed leaving only the ampullary complex attached to the duodenum (Fig. 5).
2. The reconstruction was then performed using an advanced jejunal limb. The stapled duodenum was sutured with the stapled jejunum and a latero-lateral duodenojejunostomy was performed with separated stitches.
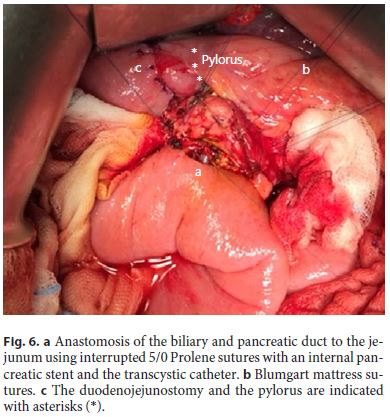
3. Afterwards, the ampullary complex was transected with scissors: the pancreatic duct was cut and a stent introduced. Next, the biliary duct was transected with preservation of the transcystic catheter and the duodenum removed. No induration or suspicious degenerescence was observed. Both ducts were joined with a suture. After a small enterotomy, the anastomosis of the biliary and pancreatic duct was performed using interrupted 5/0 Prolene sutures with an internal pancreatic stent and the transcystic catheter (Fig. 6). The pancreaticojejunal anastomosis was reinforced with Blumgart mattress sutures. The omentum was placed around the anastomosis with a closed suction drain. A nasojejunal feeding tube was placed and secured.
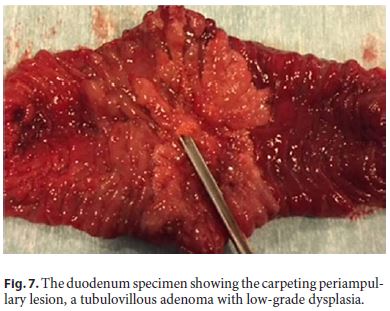
The specimen showed the carpeting periampullary lesion, a tubulovillous adenoma with low-grade dysplasia (Fig. 7). The histopathology of the surgical specimen did not confirm the previous diagnosis of high-grade dysplasia.
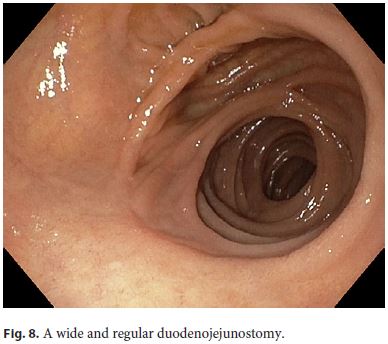
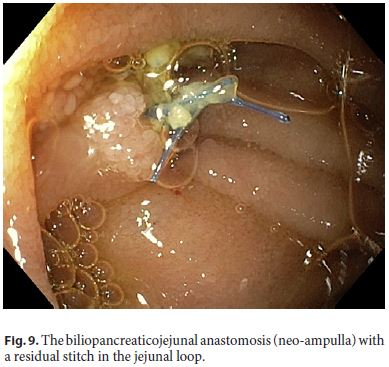
The patient was discharged on the 11th postoperative day without complications and the transcystic tube was removed at 2 months. Follow-up endoscopy during the first postoperative year showed a wide and regular duodenojejunostomy (Fig. 8) and a discrete biliary anastomosis (neo-ampulla) in the jejunum loop (Fig. 9).
Discussion
With enhanced survival as a consequence of screening and prophylactic colectomy, the significance of duodenal disease has increased [10]. The aim of endoscopic surveillance was to identify patients before the transition from adenoma to carcinoma or when only small foci of carcinomatous change were present. Once the advanced malignant change is established the prognosis after surgery is poor [11].
The selection of patients for the prophylactic surgical procedure is not easy. In a recent cohort study of 437 patients with endoscopic surveillance according to the Spigelman criteria, 52 underwent radical surgical treatment and 11 (21%) were found to have adenocarcinoma, with a further 2 cases of conservative transduodenal ampullectomy [12]. Of these 13 patients with carcinoma, only 5 were classified as Spigelman IV. The authors [12] concluded that the Spigelman score must dictate the rhythm of endoscopic surveillance whereas the presence of highgrade dysplasia or of a large unresectable ampullary lesion is an indication for prophylactic radical surgery. These risk factors were present in our patient, supporting the radical surgical option.
Pancreatic surgery may be more technically demanding in patients with FAP because of previous colectomy, the adhesions and desmoplastic changes in the mesentery with more difficult mobilizations, anastomosis of nondilated ducts, and prolonged postoperative ileus with additional stresses on the surgical anastomosis. The St. Marks group presented the operative outcome of the pylorus-preserving pancreatoduodenectomy in 16 FAP patients with Spigelman IV, including mortality in 2 (12%) and morbidity in 8 (50%), and a median hospital stay of 36 days [13]. The experience of the surgeons at the Cleveland Foundation with pancreas- and pylorus-sparing duodenectomy [9] is, in 21 cases with no mortality, a 38% rate of morbidity, 19% leakage rate, and a length of stay of 14 days. These results are comparable with the experience of the same group in performing pylorus-preserving pancreatoduodenectomy in 238 cases, with 1.3% mortality, 43% morbidity, a 9% leakage rate, and a length of postoperative stay of 14 days. The published experience of this group with pancreas-sparing duodenectomy now totals 42 cases [14]. Another Asian group has reported good outcomes in 21 patients who underwent pancreassparing duodenectomy in comparison with 44 cases with pancreaticoduodenectomy associated with a soft pancreas [15].
The morbidity of the pancreas- and pylorus-sparing duodenectomy is principally related with delayed gastric emptying and a leak at the pancreaticobiliary anastomosis. The delayed gastric emptying is multifactorial and can also occur in the classic Whipple procedure or in the pylorus-preserving pancreatoduodenectomy. For this reason, a nasojejunal tube for postoperative enteral feeding is recommended for this procedure. Our patient did not tolerate clamping of the transcystic biliary decompression catheter in the first postoperative week and it was only removed in the second postoperative month.
The anastomotic leak is the most dangerous complication and is more frequent with a soft pancreas and a small pancreatic duct [16]. The incidence of pancreatic leak is decreasing with experience and attention to technical details such as a wide duct to mucosa anastomosis and the dunking effect of the Blumgart stitches, which was the anastomotic technique used in this patient. However, until now, no differences have been found in several metaanalyses in the postoperative pancreatic fistulas related to the comparison of different techniques of pancreaticojejunostomy [17].
The pancreas-preserving duodenectomy has the potential disadvantage of mixing the bile and pancreatic secretions at one anastomotic site [9]. However, by using an open transcystic tube, we believe this risk can be decreased, as was shown in our case. Another issue relates to patients with pancreas divisum or with a large Santorini duct. To identify these cases, it is necessary to carry out a preoperative MRCP to define the pancreatic ductal anatomy with the additional advantage of identifying eventual mesenteric desmoid tumors. In these cases it is recommended to reimplant the minor papilla through a separate anastomosis to the jejunum [9].
Pancreas- and pylorus-preserving duodenectomy has an important advantage related to the possibility of complete endoscopic postoperative assessment of the small duodenal cuff, the neo-ampulla, and the proximal jejunum, in which recurrence of adenomatous tissue or carcinoma can occur. The long-term outcome of this procedure, at a mean follow-up of 79 months, has proven that it is effective in preventing duodenal carcinoma, even at the ampullary complex [9]. The resection of the duodenum shifts the carcinoma risk to the remaining small bowel and the duodenal cuff, if it is preserved. The requirement for complete surveillance would be more difficult with a Whipple procedure.
In conclusion, pancreas- and pylorus-preserving duodenectomy is a promising alternative to pancreaticoduodenectomy for advanced duodenal polyposis that enables complete endoscopic surveillance.
References
1 Bodmer WF, Bailey CJ, Bodmer J, Bussey HJ, Ellis A, Gorman P, et al. Localization of the gene for familial adenomatous polyposis on chromosome 5. Nature. 1987 Aug;328(6131):614–6.
2 Leite JS, Isidro G, Martins M, Regateiro F, Albuquerque O, Amaro P, et al. Is prophylactic colectomy indicated in patients with MYHassociated polyposis? Colorectal Dis. 2005 Jul;7(4):327–31.
3 Belchetz LA, Berk T, Bapat BV, Cohen Z, Gallinger S. Changing causes of mortality in patients with familial adenomatous polyposis. Dis Colon Rectum. 1996 Apr;39(4):384–7.
4 Arvanitis ML, Jagelman DG, Fazio VW, Lavery IC, McGannon E. Mortality in patients with familial adenomatous polyposis. Dis Colon Rectum. 1990 Aug;33(8):639–42.
5 Spigelman AD, Williams CB, Talbot IC, Domizio P, Phillips RK. Upper gastrointestinal cancer in patients with familial adenomatous polyposis. Lancet. 1989 Sep;2(8666):783–5.
6 Groves CJ, Saunders BP, Spigelman AD, Phillips RK. Duodenal cancer in patients with familial adenomatous polyposis (FAP): results of a 10 year prospective study. Gut. 2002 May;50(5):636–41.
7 Norton ID, Geller A, Petersen BT, Sorbi D, Gostout CJ. Endoscopic surveillance and ablative therapy for periampullary adenomas. Am J Gastroenterol. 2001 Jan;96(1):101–6.
8 Penna C, Phillips RK, Tiret E, Spigelman AD. Surgical polypectomy of duodenal adenomas in familial adenomatous polyposis: experience of two European centres. Br J Surg. 1993 Aug;80(8):1027–9.
9 Mackey R, Walsh RM, Chung R, Brown N, Smith A, Church J, et al. Pancreas-sparing duodenectomy is effective management for familial adenomatous polyposis. J Gastrointest Surg. 2005 Nov;9(8):1088–93.
10 Galle TS, Juel K, Bülow S. Causes of death in familial adenomatous polyposis. Scand J Gastroenterol. 1999 Aug;34(8):808–12.
11 Beckwith PS, van Heerden JA, Dozois RR. Prognosis of symptomatic duodenal adenomas in familial adenomatous polyposis. Arch Surg. 1991 Jul;126(7):825–7.
12 Sourrouille I, Lefèvre JH, Shields C, Colas C, Bellanger J, Desaint B, et al. Surveillance of duodenal polyposis in familial adenomatous polyposis: should the Spigelman score be modified? Dis Colon Rectum. 2017 Nov;60(11):1137–46.
13 Gallagher MC, Shankar A, Groves CJ, Russell RC, Phillips RK. Pylorus-preserving pancreaticoduodenectomy for advanced duodenal disease in familial adenomatous polyposis. Br J Surg. 2004 Sep;91(9):1157–64.
14 Augustin T, Moslim MA, Tang A, Walsh RM. Tailored surgical treatment of duodenal polyposis in familial adenomatous polyposis syndrome. Surgery. 2018 Mar;163(3):594–9.
15 Nakayama Y, Konishi M, Gotohda N, Kato Y, Aizawa H, Kudo M, et al. Comparison of postoperative early and late complications between pancreas-sparing duodenectomy and pancreatoduodenectomy. Surg Today. 2017 Jun;47(6):705–11.
16 Kantor O, Talamonti MS, Pitt HA, Vollmer CM, Riall TS, Hall BL, et al. Using the NSQIP pancreatic demonstration project to derive a modified fistula risk score for preoperative risk stratification in patients undergoing pancreaticoduodenectomy. J Am Coll Surg. 2017 May;224(5):816–25.
17 Wang W, Zhang Z, Gu C, Liu Q, Liang Z, He W, et al. The optimal choice for pancreatic anastomosis after pancreaticoduodenectomy: a network meta-analysis of randomized control trials. Int J Surg. 2018 Sep;57:111–6.
Statement of Ethics
This study did not require informed consent or review approval by the appropriate ethics committee.
Disclosure Statement
The authors report no potential conflicts of interest.
* Corresponding author.
Júlio S. Leite
Rua D Sancho I, 64
PT–3030-396 Coimbra (Portugal)
E-Mail julio.s.leite@gmail.com
Received: June 23, 2019; Accepted after revision: August 29, 2019
Author Contributions
All the authors read and approved the manuscript being submitted. All authors listed contributed significantly to the work.














