Serviços Personalizados
Journal
Artigo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Acessos
Acessos
Links relacionados
-
 Similares em
SciELO
Similares em
SciELO
Compartilhar
GE-Portuguese Journal of Gastroenterology
versão impressa ISSN 2341-4545
GE Port J Gastroenterol vol.27 no.3 Lisboa jun. 2020
https://doi.org/10.1159/000503073
CLINICAL CASE STUDY
De novo Gastrinoma: A Case Report
Gastrinoma de novo: A propósito de um caso clínico
Arsénio Barbosaa, Filipa Gomesa, Luísa Fonsecaa, Tiago Maiab, Jorge Almeidaa
aInternal Medicine Department, Centro Hospitalar São João, Porto, Portugal; bDepartment of Anatomic Pathology, Centro Hospitalar São João, Porto, Portugal
* Corresponding author.
ABSTRACT
Gastrinomas are neuroendocrine tumors characterized by gastrin overexpression – 80% are sporadic and 20% are associated with multiple endocrine neoplasia type 1. A 75-year-old male patient, surgically treated at the age of 50 years for gastrinoma, followed on an outpatient basis because of chronic non-bloody diarrhea, was admitted to our hospital because of abdominal pain, watery diarrhea, and nonbiliary vomits. He was hypotensive and showed no response to fluids. Blood cultures were positive for Salmonella, and a diagnosis of septic shock due to Salmonella infection was made. The patients condition improved, but the history of chronic diarrhea was still not explained. To investigate chronic diarrhea, gastrinoma recurrence was considered. Serum gastrin measurement was five times higher than the upper limit of the normal range (536 pg/mL). A positive somatostatin receptor scintigraphy was diagnostic for neuroendocrine tumor. Metastases were excluded. The patient was proposed to curative surgery, and a diagnosis of a well-differentiated neuroendocrine tumor was made.
Keywords: Gastrinoma, Zollinger-Ellison syndrome ,Neuroendocrine tumor, Multiple endocrine neoplasia
RESUMO
Gastrinomas são tumores neuroendócrinos caracterizados por hipersecreção de gastrina – 80% são esporádicos e 20% associados a neoplasia endócrina múltipla tipo 1 (MEN1). Apresenta-se o caso de um homem, de 75 anos, tratado cirurgicamente aos 50 anos por gastrinoma, seguido em consulta de Medicina Interna por diarreia crónica não-sanguinolenta, internado no nosso hospital por dor abdominal, diarreia aquosa e vómitos não biliares. À admissão, apresentava-se hipotenso e pouco responsivo a fluidoterapia. As hemoculturas foram positivas para Salmonella e foi feito o diagnóstico de choque séptico por Salmonella. O doente melhorou, mas a história prévia de diarreia crónica não estava esclarecida. Para investigar a diarreia crónica, foi colocada como hipótese recidiva de gastrinoma. A medição de gastrina sérica foi cinco vezes superior ao limite superior do normal (536 pg/mL). A cintigrafia com receptores de somatostatina foi diagnóstica para tumor neuroendócrino. Foram excluídas metástases. O doente foi proposto para cirurgia curativa e foi feito o diagnóstico de tumor neuroendócrino bem diferenciado.
Palavras-Chave: Gastrinoma, Síndrome Zollinger-Ellison, Neoplasia endócrina múltipla
Introduction
Gastrinomas are neuroendocrine tumors characterized by gastrin overexpression resulting in Zollinger-Ellison syndrome [1]. Approximately 80% are sporadic, and 20% are associated with multiple endocrine neoplasia type 1 (MEN1). The annual incidence is 0. 5–2 per million, with a higher incidence among males [1, 2]. The diagnosis is frequently made between the ages of 20 and 50 years [3].
Typical symptoms include abdominal pain and chronic diarrhea (up to two-thirds of the patients), but gastroesophageal reflux and peptic ulcer disease can also be identified.
Most gastrinomas (up to 80%) are localized in the gastrinoma triangle, comprising the head of the pancreas and the duodenal sweep [1, 4].
A gastrin level 10 times higher than the upper limit range is considered diagnostic of gastrinoma, but lower levels are frequent – especially when associated with MEN1. However, the final diagnosis depends on pathological and immunohistochemical analysis. Surgery is the only curative treatment [2, 4–6].
Clinical Case
We present the case of a 75-year-old Caucasian male diagnosed with Zollinger-Elisson syndrome. He underwent partial gastroduodenopancreatectomy at the age of 50 years, was followed on an outpatient basis in another center for almost 15 years with no evidence of the disease, and was medicated with a proton-pump inhibitor. Gastrinoma was confirmed by the first biopsy, and negative margins too. No information about the surgical procedure was known. He was asymptomatic until 6 months before admission when non-bloody diarrhea started (3–4 times per day, mostly at night, and occasionally awaking him), and involuntary weight loss was observed. Stool specimens showed high leukocyte levels, no fat, and no ova or parasites; bacteriological stool culture was negative. A course of albendazole was administered for 3 days, but the symptoms did not improve. Both upper and lower gastrointestinal endoscopic examination were performed and showed no abnormalities.
He attended our emergency department due to worsening symptoms with abdominal pain, watery diarrhea (4–6 times a day), nausea, non-biliary vomit, anorexia, and asthenia. Fever was denied. These clinical manifestations worsened 1 week prior to admission, and his son and grandson had had the same symptoms the week before. On examination, the patient was alert and oriented. His blood pressure was 103/53 mm Hg, heart rate 103 bpm, respiratory rate 22 cycles per minute, and temperature 36.8 ° C. His heart sounds and lungs were normal to auscultation. He had no palpable liver, lymph nodes, or abdominal pain on examination. He was dehydrated and presented bilateral lower-limb edema. The neurological examination was normal.
The initial workup revealed mild anemia (hemoglobin 12.5 g/dL), leukocytosis (15.50 × 109/L), thrombocytopenia (111 × 109/L), mild acute kidney injury (creatinine 1.76 mg/dL), and elevated Creactive protein (238.8 mg/L). The abdominal ultrasound and the chest X-ray were normal. Blood cultures were collected, and ceftriaxone was empirically started for a presumed bacterial gastroenteritis. Fluid depletion was also corrected. He was admitted to the ward. In the first 12 h after admission, the patient developed shock with hypotension refractory to fluids, obnubilation, oliguria, and respiratory failure. He was admitted to the intensive care unit for mechanical ventilation and vasopressor support. Gram-negative bacilli were isolated in the blood cultures, and the antibiotic was changed to piperacillin-tazobactam.
On the day after, the initial blood tests were positive for Salmonella spp., and ceftriaxone was reintroduced. The computed tomography (CT) scan showed pulmonary embolism and bilateral pleural effusion. An EcoDoppler of the lower limbs was performed, and deep vein thrombosis was diagnosed. Anticoagulation with low-molecular-weight heparin was started.
Due to Salmonella bacteremia, endocarditis was excluded by transthoracic and transesophageal echocardiography. A CT scan of the spine excluded spondylodiscitis.
The patient improved, blood cultures were negative, and he was discharged from the intensive care unit to the ward. He was treated with ceftriaxone for 21 days. Although there was a clear diagnosis for the acute setting, it could not explain the history of diarrhea in the previous 6 months, so the investigation was pursued, and gastrinoma recurrence was considered. The upper and lower endoscopies with biopsies were repeated, and no abnormalities were shown. Serum gastrin measurement was 536 pg/mL (five times the upper limit of the reference range). Since positron emission tomography is not available in our center, a somatostatin receptor scintigraphy was performed, and a retroperitoneal preaortic focus of intense radiopharmaceutical uptake was identified in relation with an overexpressed somatostatin receptor lesion compatible with neuroendocrine tumor (Fig. 1, 2). An abdominal CT angiography, done to localize the lesion more precisely, showed a solid hypervascular lesion of 20 × 17 mm in the body of the pancreas. A diagnosis of pancreatic gastrinoma was made (Fig. 3).
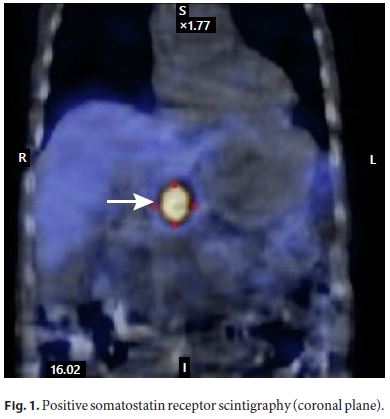
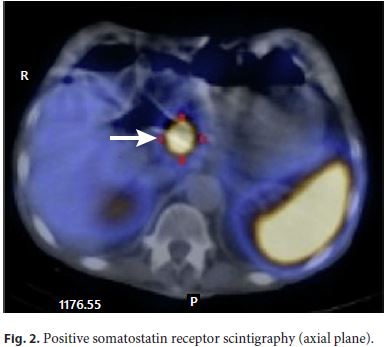
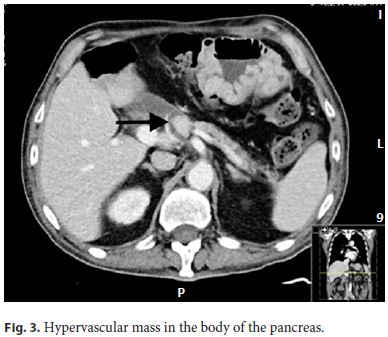
To rule out MEN1-associated gastrinoma, measurement of serum PTH, calcium, and vitamin D were done, revealing elevated PTH (104.4 pg/mL), normal calcium (2.42 mEq/L), and low vitamin D (4 ng/mL). The parathyroid ultrasound was normal. Vitamin D supplementation was started, and normalization of PTH and vitamin D levels were observed. Hyperparathyroidism was interpreted as not related to MEN1 but secondary to the low levels of vitamin D.
Pituitary tumor was ruled out by normal pituitary MRI and normal serum levels of ACTH, prolactin, FSH, and LH.
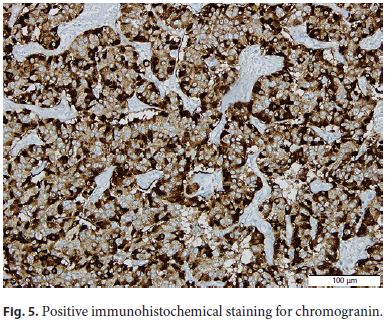
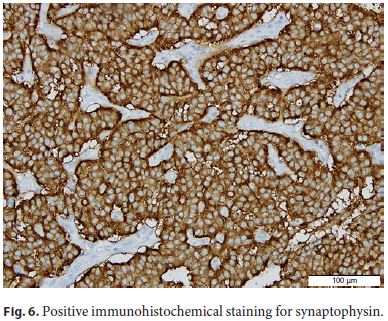
The patient was submitted to surgical resection of pancreatic gastrinoma, and the histological examination confirmed a welldifferentiated neuroendocrine tumor (Fig. 4, Fig. 5, Fig. 6).
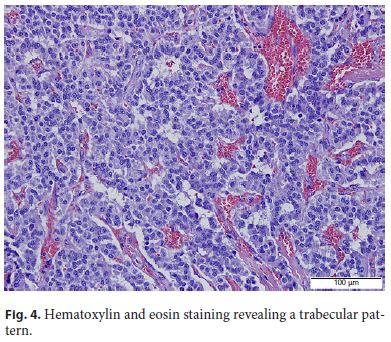
Discussion
Gastrinoma could be sporadic or associated with MEN1. MEN1 is characterized by parathyroid, pancreatic, and pituitary tumors, developing primary hyperparathyroidism, pancreatic neuroendocrine tumors, and pituitary adenomas, respectively [5].
In our patient, we found no association between gastrinoma and MEN1, despite the initial identification of hyperparathyroidism. No pituitary involvement was found.
In this case, infectious diarrhea by Salmonella was a confounding factor. The major causes of infectious diarrhea are viral, bacterial, and Protozoa infections. The severity of this case favored the suspicion of bacterial etiology. Diarrhea, positive blood cultures for Salmonella spp., the epidemiological context, and the diagnosis of septic shock due to Salmonella infection could have stopped our investigation. Despite all that, the previous history of diarrhea led us to proceed the study. Upper endoscopy showed no peptic ulcers, and duodenal biopsy did not show villous atrophy, a pathological feature of celiac disease, but no serological test was performed. Colonoscopy was unremarkable. There was no evidence of inflammatory bowel disease as no blood was found in the stool, and the endoscopic evaluation was normal 6 months before admission.
Testing for gastrinoma started with the measurement of the serum gastrin level as it is elevated virtually in all patients with Zollinger-Ellison syndrome. As our patient was medicated with proton-pump inhibitors (known to raise serum gastrin), a high gastrin level under the diagnostic range for gastrinoma could just represent an overestimation of gastrin levels secondary to this drug. A positive somatostatin receptor scintigraphy was then diagnostic for neuroendocrine tumor, and CT angiography localized the lesion to the head of the pancreas and excluded metastases.
Current guidelines recommend curative surgical resection for all localized tumors, including locoregional lymph node metastasis removal. Follow-up is recommended with CT or MRI every 3–6 months and positron emission tomography 18–24 months after the surgery [6].
The most interesting aspect of this case is related to the appearance of a de novo gastrinoma 25 years after the first episode. Although frequent, as more than 50% of all surgically treated patients relapse, it occurs mostly within the first 2 years [7]. The risk factors are not well known, but size, histological grade, and neural invasion seem to be implicated. Lymph nodes and liver metastases – the most frequent sites for recurrence – are also implicated, but most studies are done in patients with localized tumors with no metastases [8, 9]. We did not find reports of a de novo gastrinoma 25 years after the first surgery in the literature.
References
1 Zhang WD, Liu DR, Wang P, Zhao JG, Wang ZF, Chen LI. Clinical treatment of gastrinoma: A case report and review of the literature. Oncol Lett. 2016 May;11(5):3433–7.
2 Maire F, Sauvanet A, Couvelard A, Rebours V, Vullierme MP, Lebtahi R, et al. Recurrence after surgical resection of gastrinoma: who, when, where and why? Eur J Gastroenterol Hepatol. 2012 Apr;24(4):368–74.
3 Huang SF, Kuo IM, Lee CW, Pan KT, Chen TC, Lin CJ, et al. Comparison study of gastrinomas between gastric and non-gastric origins. World J Surg Oncol. 2015 Jun;13(1):202. [ Links ]
4 Banasch M, Schmitz F. Diagnosis and treatment of gastrinoma in the era of proton pump inhibitors. Wien Klin Wochenschr. 2007;119(19-20):573–8.
5 Öberg K, Knigge U, Kwekkeboom D, Perren A; ESMO Guidelines Working Group. Neuroendocrine gastro-entero-pancreatic tumors: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol. 2012 Oct;23 Suppl 7:vii124–30.
6 Huang C, Zhu X, Han X, Lou W. Metastatic multiple endocrine neoplasia type 1: report of one case. Translational Gastroenterology and Hepatology. 2016. 23: 1-73. J Gastroenterol. 1993;93(1):66–70.
7 Metz DC, Cadiot G, Poitras P, Ito T, Jensen RT. Diagnosis of Zollinger-Ellison syndrome in the era of PPIs, faulty gastrin assays, sensitive imaging and limited access to acid secretory testing. Int J Endocr Oncol. 2017;4(4):167–85.
8 Hain E, Coriat R, Dousset B, Gaujoux S. [Management of gastrinoma]. Presse Med. 2016 Nov;45(11):986–91.
9 Krausz Y, Bar-Ziv J, de Jong RB, Ish-Shalom S, Chisin R, Shibley N, et al. Somatostatin-receptor scintigraphy in the management of gastroenteropancreatic tumors. Am J Gastroenterol. 1998 Jan;93(1):66–70.
Statement of Ethics
This study did not require review or approval by the appropriate ethics committee.
Disclosure Statement
The authors have no conflicts of interest to declare.
* Corresponding author.
Arsénio Barbosa
Internal Medicine Department, Centro Hospitalar São João
Alameda Professor Hernâni Monteiro
PT–4200-319 Porto (Portugal)
E-Mail arseniomiguelbarbosa@gmail.com
Received: February 3, 2019; Accepted after revision: August 31, 2019














