Serviços Personalizados
Journal
Artigo
Indicadores
-
 Citado por SciELO
Citado por SciELO -
 Acessos
Acessos
Links relacionados
-
 Similares em
SciELO
Similares em
SciELO
Compartilhar
GE-Portuguese Journal of Gastroenterology
versão impressa ISSN 2341-4545
GE Port J Gastroenterol vol.27 no.5 Lisboa out. 2020
https://doi.org/10.1159/000507375
GUIDELINES
Small Bowel Enteroscopy – A Joint Clinical Guideline from the Spanish and Portuguese Small Bowel Study Groups
Enteroscopia flexível – Guideline conjunta dos Grupos de estudos Espanhol e Português de Intestino Delgado
Enrique Pérez-Cuadrado-Roblesa, Rolando Pinhob, Begoña Gonzalezc, Susana Mão de Ferrod, Cristina Chagase, Pilar Esteban Delgadof, Cristina Carreterog, Pedro Figueiredoh,i, Bruno Rosaj,k, Javier García Lledól, Óscar Nogalesl, Ana Ponteb, Patrícia Andradem, Jose Francisco Juanmartiñena-Fernándezn, Mileidis San-Juan-Acostao, Sandra Lopesh,i, César Prieto-Fríasg, Juan Egea-Valenzuelap, Noemí Caballeroq, Eduardo Valdivieso-Cortazarr, Hélder Cardosom, Consuelo Gálvezs, Nuno Almeidah,i, Pilar Borque Barrerao, Blas José Gómez-Rodríguezt, Francisco Sánchez Ceballosu, Carlos Bernardesv, Pedro Alonsor, Federico Argüelles-Ariast, Miguel Mascarenhas Saraivaw, Enrique Pérez-Cuadrado-Martínezf; Capsule Endoscopy and Enteroscopy Group of the Spanish, Society of Gastrointestinal Endoscopy (SEED), Portuguese Small Bowel Study Group (GEPID)
aDepartment of Gastroenterology, Hôpital Européen Georges-Pompidou, Paris, France; bDepartment of Gastroenterology, Centro Hospitalar de Vila Nova de Gaia e Espinho, Vila Nova de Gaia, Portugal; cDepartment of Gastroenterology. Endoscopy Unit, ICMDiM, Hospital Clínic, Barcelona, Spain; dDepartment of Gastroenterology, Instituto Português de Oncologia de Lisboa Francisco Gentil EPE, Lisbon, Portugal; eDepartment of Gastroenterology, Hospital de Egas Moniz, Centro Hospitalar de Lisboa Ocidental, Lisbon, Portugal; fSmall Bowel Unit, Hospital Universitario Morales Meseguer, Murcia, Spain; gDepartment of Gastroenterology, University of Navarra Clinic, Pamplona, Spain; hGastroenterology Unit, Centro Hospitalar e Universitário de Coimbra, Coimbra, Portugal; iFaculty of Medicine, University of Coimbra, Coimbra, Portugal; jDepartment of Gastroenterology, Hospital da Senhora da Oliveira, Guimarães, Portugal; kLife and Health Sciences Research Institute, School of Medicine, University of Minho, ICVS/3B’s, PT Government Associate Laboratory, Braga/Guimarães, Portugal; lDepartment of Gastroenterology, Endoscopy Unit, Hospital General Universitario Gregorio Marañón, Madrid, Spain; mDepartment of Gastroenterology, Centro Hospitalar São João, Porto, Portugal; nDepartment of Gastroenterology, Complejo Hospitalario de Navarra, Navarra, Spain; oDepartment of Gastroenterology, Gastrointestinal Endoscopy Unit, Hospital Universitario Nuestra Señora de Candelaria, Candelaria, Tenerife, Spain; pUnit of Gastrointestinal Endoscopy, Department of Digestive Disease, Hospital Clínico Universitario Virgen de la Arrixaca, Murcia, Spain; qDepartment of Gastrointestinal Endoscopy, Hospital Universitario Germans Trias i Pujol, Badalona, Spain; rDepartment of Gastroenterology, Complejo Hospitalario Universitario de A Coruña, A Coruña, Spain; sDepartment of Gastroenterology. Hospital Clínico Universitario de Valencia, Valencia, Spain; tDepartment of Gastroenterology, Hospital Universitario Virgen Macarena, Universidad de Sevilla, Sevilla, Spain; uDepartment of Gastroenterology, Hospital Clínico San Carlos, Madrid, Spain; vDepartment of Gastroenterology, Hospital de Santo António dos Capuchos, Centro Hospitalar Universitário de Lisboa Central, Lisbon, Portugal; wLaboratório de endoscopia e motilidade digestiva, ManopH, Porto, Portugal
* Corresponding author.
ABSTRACT
The present evidence-based guidelines are focused on the use of device-assisted enteroscopy in the management of small-bowel diseases. A panel of experts selected by the Spanish and Portuguese small bowel study groups reviewed the available evidence focusing on the main indications of this technique, its role in the management algorithm of each indication and on its diagnostic and therapeutic yields. A set of recommendations were issued accordingly.
Keywords: Small bowel, Enteroscopy, Angioectasia, Guidelines, Capsule endoscopy, Device-assisted enteroscopy
RESUMO
Estas recomendações baseadas na evidência detalham o uso da enteroscopia assistida por dispositivo no manejo clínico das doenças do intestino delgado. Um conjunto de Gastrenterologistas diferenciados em patologia do intestino delgado foi selecionado pelos grupos de estudos Espanhol e Português de intestino delgado para rever a evidência disponível sobre as principais indicações desta técnica, o seu papel nos algoritmos de manejo de cada indicação e sobre o seu rendimento diagnóstico e terapêutico. Foi gerado um conjunto de recomendações pelos autores.
Palavras-Chave: Intestino delgado, Enteroscopia, Angiectasia, Guidelines, Endoscopia por cápsula, Enteroscopia assistida por dispositivo
Introduction
Device-assisted enteroscopy (DAE) has shown high diagnostic yields in small bowel (SB) diseases [1]. Furthermore, this technique is the first-line therapeutic procedure for several small bowel pathologies. However, its role, place in the management algorithm and yields are dependent on numerous factors namely the indication and previous examinations such as capsule endoscopy (CE). The aim of the present guidelines is to provide evidence-based recommendations on the clinical indications and diagnostic and therapeutic yields of DAE in SB diseases.
Methods
The present guidelines were promoted and supported by the Capsule Endoscopy and Enteroscopy Group of the Spanish Society of Gastrointestinal Endoscopy (SEED) and the Portuguese Small Bowel Study Group (GEPID) – a section of the Portuguese Gastroenterology Society (SPG). Two guideline coordinators (EPCR, RP) were designated, who invited members from both societies and selected experts in the field from Spain and Portugal.
Seven task force groups comprising 3–5 persons coordinated by a group leader were created for the following subjects: overt SB bleeding, occult SB bleeding, Crohn’s disease (CD), tumors, polyposis syndromes, celiac disease and miscellaneous indications. Each group contained members from both societies, with variable levels of expertise and from different institutions (Table 1).
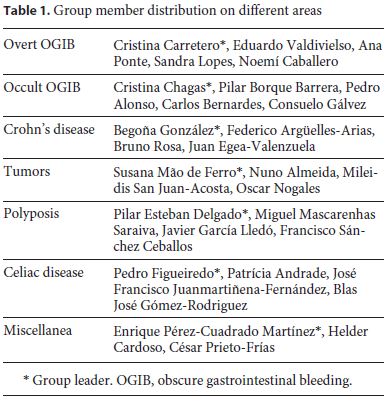
The key questions to be addressed were decided by each group coordinator but included indications, diagnostic yield, and therapeutic yield or impact in the patient clinical course. Technical aspects and the use of DAE in specific settings outside the SB, such as difficult colonoscopy and enteroscopy-assisted endoscopic retrograde cholangiopancreatography, were not included in this guideline.
A systematic review of the literature was performed. The literature search was carried out in PubMed, the Cochrane Library and Scopus until November 2019 combining the following common and specific terms:
− Common terms for all task forces: enteroscopy OR double-balloon OR DBE OR single-balloon OR SBE.
− Specific terms depending on the topic: obscure gastrointestinal bleeding OR OGIB OR small-bowel bleeding OR anemia OR anaemia; Crohn OR inflammatory bowel disease OR IBD; tumor OR tumors OR tumour OR tumours OR neoplasia OR cancer; polyp OR polyps OR polyposis OR Peutz-Jeghers OR Familial adenomatous polyposis; celiac disease OR coeliac disease.
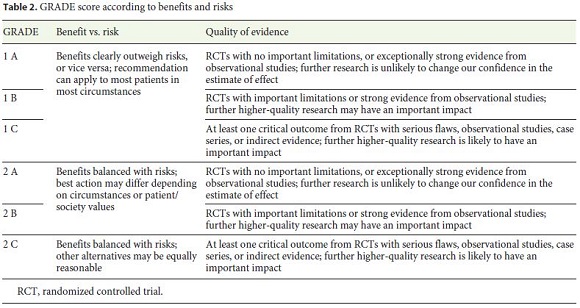
The Grading of Recommendations Assessment, Development, and Evaluation (GRADE) was used for assessing evidence levels and recommendation strengths (Table 2). The guideline development process included online discussions and one meeting during the Iberian Meeting of Enteroscopy in January 2020 to discuss draft proposals and the main recommendations of all topics. Finally, a panel of experts was decided by the Editorial Committee of the Revista Española de Enfermedades Digestivas (REED) to perform an external review of the manuscript.
The present document is intended to be an evidencebased state of the art guide for endoscopists dedicated to SB diseases. This Guideline will be considered for review in 2025, but an update will be added sooner if relevant evidence becomes available meanwhile.
Small Bowel Bleeding
Obscure gastrointestinal bleeding (OGIB) accounts for approximately 5% of all cases of gastrointestinal bleeding and is usually due to a SB lesion presenting with SB bleeding (SBB) [1]. Several meta-analyses have shown comparable diagnostic yields for CE and DAE [2]. CE is recommended as the first line examination due to its non-invasiveness and favorable safety profile. DAE should be performed after the detection of the bleeding source or in the emergency setting in patients with massive bleeding [1, 3]. The diagnostic yield of DAE ranges from 47% to 75% [2, 4–8], depending on the type of lesion and indication [9, 10]. Xin et al showed a pooled overall diagnostic yield for double balloon enteroscopy (DBE) of 68% in a meta-analysis [11]. In addition, this rate is significantly higher when DAE is performed following a positive-CE (75% vs. 27.5%) [2].
Overt Small Bowel Bleeding
Data on the diagnostic yield of DAE focused in overt-OGIB is limited to small case series. In this setting, DAE proved to have a higher diagnostic yield when performed in the urgent setting (< 72 h), averaging 70–90% [12]. This compares favorably with the diagnostic yield of DAE in the non-urgent setting, which averages 30–50% [13–15].
The therapeutic yield of DAE performed in the overt-OGIB setting ranges from 41.4 to 88.9% [14, 16–21]. Therapeutic procedures vary according to the etiology and include clipping, argon plasma coagulation, epinephrine injection, heater probe coagulation and polypectomy [14, 18, 21–24]. Additionally, endoscopic tattooing can be performed for a subsequent surgical approach [20]. The timing in which DAE is performed can also influence the therapeutic yields. Recent studies reported that emergent DAE (< 24–72 h) resulted in endoscopic therapy in 28.6–57.5%, while non-emergent DAE resulted in endoscopic therapy in 13–50% [14, 24–26]. Concerning rebleeding, repeating DAE therapy after an initial therapeutic DAE may decrease the number of episodes of overt rebleeding in patients with recurrent OGIB [27]. Moreover, Aniwan S, et al concluded that the rebleeding rate was lower after emergent DBE compared to non-emergent DBE, although not significant (10% vs. 29%, p = 0.08) [26].
The oral route is usually selected first [12, 14, 16, 28, 29]. Previous findings from imaging studies or CE have a major role guiding further approach [5, 12, 14, 28, 30–33]. If a lesion detected on CE is within the first 75% of SB transit time, an anterograde approach should be chosen [14, 17, 20, 22, 24, 25, 34]. Pérez-Cuadrado Robles E et al. concluded that real-time CE findings can also be useful to decide the initial route on emergent SBB [17]. When a prior study hasn’t revealed a potential bleeding source, the oral approach should be chosen or the insertion route should be selected according to the type of bleeding [5, 12, 16, 19, 22, 25–28, 30, 31, 33–37]. The presence of melena prompts to anterograde DAE and hematochezia to retrograde DAE [16, 26, 36]. In fact, Zhu CN et al. showed that the presence of melena doubles the odds of finding a bleeding site within the proximal SB (OR 1.97, 95% CI 1.17–3.33, p = 0.01), prompting for oral DAE in these patients [38]. If the clinical suspicion of bleeding is high despite negative findings on the initial insertion route, the limit of insertion should be marked by clipping or tattooing, and DAE using the opposite route of insertion should be performed [12, 14, 19–22, 25, 26, 28, 33, 35–37]. When a bleeding source is identified, total enteroscopy is not required [27, 30, 35].
Major adverse events associated with DAE procedures in overt-OGIB (such as perforation or pancreatitis) are very rare and occur in about 0.5% of cases [8, 16, 20, 29, 31, 37, 39]. Minor complications occur in about 11% of patients, and include abdominal discomfort, abdominal pain for < 48 h, sore throat and minimal SB mucosal trauma. Patients can usually be discharged on the same day of the procedure [19, 31, 34, 37, 40]. Mortality related to DAE or endoscopic therapy is extremely rare [41].
Occult Small Bowel Bleeding and Iron Deficiency Anemia
DAE has a high diagnostic yield in the setting of occult-OGIB or iron deficiency anemia (IDA) ranging from 52.4% to 75.6% [2, 25, 36, 40, 42–46], which increases when DAE is performed after a positive CE [20]. The most frequently identified lesions are angiodysplasias, erosions, ulcers and tumors [1, 2, 44]. The yields and findings appear to be similar to those achieved with CE, especially when a complete DAE is achieved [47–49]. CE prior DAE is useful to identify potential lesions and to select the most convenient route for the procedure, as described above for overt-OGIB [2, 47].
There are no randomized controlled trials evaluating the efficacy of DAE in patients with occult-OGIB/IDA. Moreover, most cohorts don’t divide their therapeutic results between patients with overt and occult-OGIB/IDA. However, several retrospective and prospective observational studies reported a high therapeutic yield – usually defined as the ability to successfully perform therapeutic endoscopic procedures or improvement in hemoglobin levels/decrease in transfusion requirements [19, 20, 39, 50–55]. In spite of achieving a high endoscopic therapeutic success and a reduction in the need for transfusion support, two systematic reviews concluded that the rate of recurrence/rebleeding is substantial [56, 57]. Given the fact that the major cause of occult-OGIB/IDA are SB angiodysplasias, which tend to be multiple, additional studies to verify the role of periodic enteroscopic interventions and/or its association with medical treatment in the prevention of rebleeding are needed.
The outcomes of occult-OGIB/IDA after DAE therapy evaluated in different publications are numerous: resolution of anemia (or improvement in hemoglobin levels), need for endoscopic procedures, number of hospital admissions, hospitalization time, transfusion requirements, mortality etc. Most retrospective studies and case series report high diagnostic and therapeutic yields, resulting in reduced transfusion requirements, iron supplementation or the need for subsequent endoscopic treatment [58]. Rebleeding after an initial DAE hemostasis ranges from 20% to 52.6% [18, 19, 21, 23, 27, 55, 59–61]. Female sex [OR: 1.96, 95% CI: 1.1–3.3], Osler-Weber syndrome [OR: 4.35, 95% CI: 1.2–15.4] and cardiac disease [OR: 1.89, 95% CI: 1.1–2.9] were associated with rebleeding in a recent meta-analysis [62]. Williamson et al demonstrated a significant decrease in blood transfusion requirements, need for iron supplementation and additional procedures after a first therapeutic DAE [55]. Repeating therapeutic DAE has proven to be useful in rebleeding patients [53]. Furthermore, patients with treatable lesions have better clinical outcomes [26, 35, 63]. When multiple SB vascular lesions are identified, treatment efficacy can be limited, but a reduction in the number of transfusions may be achieved. In the case of erosions or ulcers, patients with potentially treatable lesions, such as NSAIDs use, or inflammatory bowel disease (IBD), have better long-term outcomes than those in which the cause remains unknown [35].
Crohn’s Disease
DAE has a limited role in the initial evaluation of patients with suspected IBD. However, when SB lesions are identified by cross-sectional imaging modalities or CE, histologic evaluation may be necessary to rule out other diseases [64, 65]. If the location of these lesions lies outside the reach of standard endoscopy, DAE is the preferred technique. In this setting, it has a higher diagnostic yield than radiologic techniques (SB barium contrast, CT enterography and magnetic resonance enterography, MRE) [66].
Regarding established CD, DAE may be useful in patients with unclear symptoms and for therapy of SB strictures or bleeding lesions [67–70]. This technique can confirm suspected CD with a diagnostic yield of 22–80% [65, 71–73], and can also change the initial diagnosis in up to 12% of cases when the lesions that are observed or the histological findings are different from those reported by CE or radiology [73–75].
Therapeutic DAE in CD is mainly indicated to perform balloon dilation of SB strictures to prevent or delay surgical interventions. In experienced hands the technical feasibility is over 90%, which is equivalent to conventional endoscopic balloon dilation for colon and ileocolonic anastomoses [69]. Dilation of anastomotic or primary strictures, shorter than 5 cm, non-angulated and without significant inflammatory activity namely deep ulcers or fistulae, proved to be safe and associated with better longterm outcomes [69, 70, 76–78]. Data on adjunctive therapies for refractory strictures, such as triamcinolone or infliximab injection, stent placement or cutting techniques are scarce and mixed, currently not supporting their routine use. Approximately 80% of CD patients submitted to DAE balloon dilation remain symptom-free after 3 years, although nearly half of these cases will require at least one re-dilation procedure. The mean diameter of dilation reported is 12–15 mm, with an overall complication rate of 4.8% per patient and 2.6% per dilation [76, 77]. The presence of large and deep ulcers, fistulas and/or abdominal or pelvic abscesses are contraindications for balloon dilation.
Small Bowel Tumors
SB tumors (SBT) comprise less than 5% of gastrointestinal cancers. The incidence of primary SBT is increasing, mainly due to the rise of neuroendocrine tumors (NETs). The most common SBTs are gastrointestinal stromal tumor (GIST), adenocarcinoma, NET, and lymphoma [79]. Overall, SBTs are more frequent in the jejunum, followed by the duodenum and ileum, and most present with SBB [80–83].
CE and DAE are complementary, with high diagnostic concordance for the detection of SBT and polyps [84]. However, DAE may have a higher diagnostic yield than CE [85]. Both endoscopic techniques performed better in SBT detection than contrast-enhanced computed tomography [85–87]. DBE is an useful procedure to determine the extent, location and endoscopic characteristics of SBT, allowing biopsy examination and tattoo injection to guide a possible surgery. It also provides additional information to other procedures, that may be decisive in the clinical course of these patients [81–83, 87–90].
If there are no doubts about the diagnosis of SBT at CE, direct surgery is acceptable if cross-sectional imaging excludes inoperability [1]. In all other cases, histological confirmation is crucial to make treatment decisions [83, 90, 91]. Biopsies have a high diagnostic value, especially for adenocarcinoma and lymphoma (71.4 and 60%, respectively) [87].The need for histology of GISTs must be thoroughly balanced, since the rate of a positive histological diagnosis by DAE does not exceed 46.7% and the risk of bleeding is not negligible [92, 93].
Concerning NETs, a retrospective study demonstrated that bidirectional DAE performed in patients with previously known tumors, to exclude multifocality, revealed additional neuroendocrine tumors in 51.1% [94].
DAE can modify the clinical course in 25–65% of patients by delaying or avoiding emergent surgery or by modifying the surgical approach [83, 86].
DAE allows therapeutic interventions, primarily hemostasis of bleeding SBT (using argon plasma coagulation, hemostatic powder, clipping, and epinephrine or sclerosant injections). Some case series proposed DAE for stenting in SB obstruction secondary to SBT (as emergency treatment and in palliative patients) [95–98]. The evidence is scarce given the limited working channel of the enteroscope and the high degree of technical skills required.
After surgical resection of SBT in patients without polyposis or other inherited syndromes there are no clear recommendations for follow-up. However, some authors recommend DAE [99].
Small Bowel Polyposis
Small bowel polyps occur in 90% of Peutz-Jeghers syndrome (PJS) patients and in more than 75% of those with familial adenomatous polyposis, being distal small bowel polyps more frequent in patients who also have duodenal polyps [100]. A retrospective study showed that CE identified the number, location and size of polyps and could indicate the route (oral or anal) for DAE and predict the difficulty of polypectomy during deep enteroscopy [101]. However, DAE was better than CE to define the size and location of polyps.
A prospective study showed that MRE and DAE have similar diagnostic yield for detecting clinically relevant SB polyps (≥ 15 mm in diameter) in patients with PJS [102], but the most important advantage of DAE compared with MRE is its ability for immediate polypectomy, tattooing and biopsy.
An initial CE/MRE may be preferred to select for enteroscopy only those patients needing therapy [1] although the diagnostic rate is similar to DAE [103]. Polyp size is the most important risk factor for SB intussusception, being intussusception generally due to polyps ≥15 mm in diameter. Consequently, large polyps (10–15 mm) or symptomatic or rapidly growing polyps should be removed in PJS [104, 105]. DAE proved to be useful in the resection of SB polyps resulting in a decrease in the average number and size of lesions in periodic enteroscopies [106–108]. Additionally, absence of intussusception or complications requiring surgery after a follow-up of up to 56.5 months [107] and an adequate safety profile was also verified. There are series in which complications are not reported [109], but other studies report complications between 4–6% including bleeding, pancreatitis, perforation and post-polypectomy abscesses, treated with conservative management in most cases [106–108, 110]. The effect on cancer reduction after enteroscopy resections remains unknown.
There is little evidence in determining the indication for DAE in familial adenomatous polyposis patients. Screening and surveillance with DAE of SB adenomas could be useful in patients with Spigelman scores III or IV [111], even though the malignant potential of these lesions is unknown [112–114].
Celiac Disease
There is a little role for DAE in the diagnosis of celiac disease (CeD) as most patients are diagnosed based on endoscopic and histologic findings of upper GI endoscopy. Some studies reported a patchy distribution of histologic abnormalities [115], thus DAE may be indicated in cases of strong clinical suspicion with positive specific serology and negative duodenal biopsies at upper-GI endoscopy.
The role of DAE is mainly for the diagnosis of CeD complications [116]. Patients uncompliant or unresponsive to a gluten-free diet, with alarm symptoms or iron deficiency anemia, have an increased risk of developing SB malignancies [117]; in this case, SB evaluation by CE, upper endoscopy and imaging tests followed by DAE in order to obtain mucosal samples for histological and/or molecular analysis is recommended. Currently, it is difficult to know the diagnostic yield of DAE as only a few retrospective papers and a single meta-analysis [117] can be found. Among these studies, the overall diagnostic yield approaches up to 20%, although this value decreases when evaluating SB malignant and premalignant lesions separately. The diagnostic yield for malignant lesions ranges between 16.7–24%, whereas for premalignant lesions it ranges from 9 to 16% [118, 119].
Miscelaneous
There are other clinical conditions in which DAE is useful. Due to its ability to perform biopsies of the entire SB, DAE has been reported in the diagnosis of malabsorption syndromes and SB chronic infections, mainly tuberculosis and Whipple’s disease [120, 121]. It also enables the characterization of other diseases such as NSAID enteropathy [122], ischemic enteritis and radiation-induced enteritis, graft versus host disease with SB involvement, and other lesions such as inflammatory fibroid polyps and SB diverticula, including Meckel’s diverticulum [123]. In altered anatomy, such as Roux-en-Y anastomosis and gastrojejunostomy, DAE enables the study of intestinal segments that are inaccessible to regular endoscopes, including CE [124]. Endoscopic enteroclysis can be useful in selected cases such as stenosis. DAE has also been used for therapy: retrieval of potentially harmful foreign bodies, especially retained CE [125], but also bezoars [126], needles [127], coins [128], gastric bands [129], dentures [130] and migrated stents [131]. Many SB strictures secondaries to NSAIDs, radiation, surgical anastomosis, or malignancy have been treated with balloon dilation [76, 132, 133] or stenting, using both over-the-wire or through-the-scope techniques [134–136]. DAE can also be used for percutaneous endoscopic jejunostomy [137] and SB intussusception [138].
Conclusions
Since its introduction in routine clinical practice almost two decades ago, the diagnostic and therapeutic capabilities of DAE have continuously evolved [139]. The evidence published in the last years helped to better define the role of DAE in its various indications, as well as its advantages and disadvantages over other endoscopic and radiologic procedures. As the technology continues to evolve and mature, further refinements in its capabilities and use are anticipated.
Appendix: Statements
Small bowel bleeding
In OGIB, the route of insertion of DAE should be based on pre-DAE investigations, such as CE. GRADE 2B (weak recommendation, moderate-quality evidence)
DAE should be the first-line therapeutic procedure following CE in both patients with overt and occult OGIB. GRADE 2A (weak recommendation, high-quality evidence)
There are no serious complications regarding the examination, or the endoscopic treatment. The procedure is generally well tolerated. GRADE 2B (weak recommendation, moderate-quality evidence)
1. Overt OGIB
DAE has a high therapeutic yield in overt-OGIB. The therapeutic technique should be selected according to the bleeding source. GRADE 1B (strong recommendation, moderate-quality evidence)
In patients with recurrent overt-OGIB, a second therapeutic DAE is recommended. GRADE 2C (weak recommendation, low-quality evidence)
In overt-OGIB, DAE should be performed whenever possible in the first 72h, as this leads to higher diagnostic and therapeutic yields and lower rebleeding rates. GRADE 2C (weak recommendation, low-quality evidence)
2. Occult OGIB
DAE has a high therapeutic yield in occult-OGIB. The recommended treatment options are: argon plasma coagulation for angioectasia coagulation; hemostatic clipping for Dieulafoy lesions or bleeding ulcers; polypectomy for bleeding small bowel polyps; and/or tattooing lesions for further surveillance or treatment. GRADE 2B (weak recommendation, moderate-quality evidence)
Follow-up of patients with vascular lesions is recommended due to their high rebleeding rate. Repeated treatment with DAE might improve management of refractory OGIB. Grade 1C (strong recommendation, lowquality evidence)
Crohn’s disease
DAE is indicated in suspected CD in patients with SB lesions identified by other non-invasive techniques (CE or CT/MR enterography) and non-accessible to conventional endoscopy. In these cases, histological diagnosis is recommended. GRADE 1B (strong recommendation, moderate-quality evidence)
In established CD, DAE can be useful for diagnosis and therapy in selected cases (dilatation of strictures and retrieval of impacted capsules). GRADE 1B (strong recommendation, moderate-quality evidence)
DAE dilatation of primary or anastomotic strictures which are shorter than 5cm, non-angulated and without significant inflammatory activity, is safe and effective, with a low rate of complications. GRADE 1B (strong recommendation, moderate-quality evidence)
Tumors
DAE should be used in a combined approach with CE and radiological examinations for the diagnosis of SB tumors. GRADE 1C (strong recommendation, low-quality evidence)
DAE should be considered in patients with a high clinical suspicion of SB tumors despite negative CE and crosssectional studies. GRADE 2C (weak recommendation, low-quality evidence)
In patients with suspected SB tumors, if there is no undeniable indication for surgery, DAE should be performed to confirm diagnosis, obtain biopsies for histological documentation, establish the precise location of the lesion and mark it for further surgical treatment. GRADE 1C (strong recommendation, low-quality evidence)
Polyposis
DAE therapy is recommended for polyps > 10-15mm to prevent polyp-related complications GRADE 1B (strong recommendation, moderate-quality evidence)
DAE may also be used in symptomatic patients with polyps causing intussusception symptoms and hemorrhage / anemia in intestinal polyposis syndromes (FAP and PJS) GRADE 1C (strong recommendation, low-quality evidence)
DAE may be used in FAP patients with Spigelman stages III/IV for screening and surveillance of intestinal adenomas and endoscopic treatment. GRADE 2C (weak recommendation, low-quality evidence)
Celiac disease
There is a little role for enteroscopy in the diagnosis of celiac disease. Enteroscopy may be indicated in cases of strong clinical suspicion with positive specific serology and negative duodenal biopsies at upper GI endoscopy. GRADE 2C (weak recommendation, low-quality evidence)
The role of enteroscopy is mainly for the diagnosis of celiac disease complications. In patients uncompliant or unresponsive to gluten-free diet, with alarm symptoms or iron deficiency anemia, enteroscopy is recommended after SB evaluation by CE, upper-GI endoscopy and imaging tests, in order to obtain mucosal samples for histologic and/or molecular analysis. GRADE 2B (weak recommendation, moderate-quality evidence)
References
1 Pennazio M, Spada C, Eliakim R, Keuchel M, May A, Mulder CJ, et al. Small-bowel capsule endoscopy and device-assisted enteroscopy for diagnosis and treatment of small-bowel disorders: European Society of Gastrointestinal Endoscopy (ESGE) Clinical Guideline. Endoscopy. 2015 Apr;47(4):352–76.
2 Teshima CW, Kuipers EJ, van Zanten SV, Mensink PB. Double balloon enteroscopy and capsule endoscopy for obscure gastrointestinal bleeding: an updated meta-analysis. J Gastroenterol Hepatol. 2011 May;26(5):796–801.
3 Pérez-Cuadrado-Robles E, Pérez-Cuadrado-Martínez E. The Role of Emergency Endoscopy in Small Bowel Bleeding: A Review. GE Port J Gastroenterol. 2015 Dec;23(2):84–90.
4 Pattni V, Tate DJ, Terlevich A, Marden P, Hughes S. Device-assisted enteroscopy in the UK: description of a large tertiary case series under conscious sedation. Frontline Gastroenterol. 2018 Apr;9(2):122–8.
5 Hashimoto R, Matsuda T, Nakahori M. Falsenegative double-balloon enteroscopy in overt small bowel bleeding: long-term follow-up after negative results. Surg Endosc. 2019 Aug;33(8):2635–41.
6 Akarsu M, Akkaya Özdinç S, Celtik A, Akpınar H. Diagnostic and therapeutic efficacy of double-balloon endoscopy in patients with small intestinal diseases: single-center experience in 513 procedures. Turk J Gastroenterol. 2014 Aug;25(4):374–80.
7 Khashab MA, Lennon AM, Dunbar KB, Singh VK, Chandrasekhara V, Giday S, et al. A comparative evaluation of single-balloon enteroscopy and spiral enteroscopy for patients with mid-gut disorders. Gastrointest Endosc. 2010 Oct;72(4):766–72.
8 Arakawa D, Ohmiya N, Nakamura M, Honda W, Shirai O, Itoh A, et al. Outcome after enteroscopy for patients with obscure GI bleeding: diagnostic comparison between double-balloon endoscopy and videocapsule endoscopy. Gastrointest Endosc. 2009 Apr;69(4):866–74.
9 Perez-Cuadrado-Robles E, Esteban-Delgado P, Martinez-Andres B et al. [Diagnosis agreement between capsule endoscopy and doubleballoon enteroscopy in obscure gastrointestinal bleeding at a referral center]. Rev Esp Enferm Dig. 2015;107:495–500.
10 Ohmiya N, Yano T, Yamamoto H, Arakawa D, Nakamura M, Honda W, et al. Diagnosis and treatment of obscure GI bleeding at double balloon endoscopy. Gastrointest Endosc. 2007 Sep;66(3 Suppl):S72–7.
11 Xin L, Liao Z, Jiang YP, Li ZS. Indications, detectability, positive findings, total enteroscopy, and complications of diagnostic double-balloon endoscopy: a systematic review of data over the first decade of use. Gastrointest Endosc. 2011 Sep;74(3):563–70.
12 Liu Y, Jiang W, Chen G, Li Y. Diagnostic Value and Safety of Emergency Single-Balloon Enteroscopy for Obscure Gastrointestinal Bleeding. Gastroenterol Res Pract. 2019 Aug;2019:9026278. [ Links ]
13 Mönkemüller K, Neumann H, Meyer F, Kuhn R, Malfertheiner P, Fry LC. A retrospective analysis of emergency double-balloon enteroscopy for small-bowel bleeding. Endoscopy. 2009 Aug;41(8):715–7.
14 Rodrigues JP, Pinho R, Rodrigues A, Sousa M, Silva JC, Gomes C, et al. Diagnostic and therapeutic yields of urgent balloon-assisted enteroscopy in overt obscure gastrointestinal bleeding. Eur J Gastroenterol Hepatol. 2018 Nov;30(11):1304–8.
15 Aniwan S, Viriyautsahakul V, Rerknimitr R, Angsuwatcharakon P, Kongkam P, Treeprasertsuk S, et al. Urgent double balloon endoscopy provides higher yields than non-urgent double balloon endoscopy in overt obscure gastrointestinal bleeding. Endosc Int Open. 2014 Jun;2(2):E90–5.
16 Tu CH, Kao JY, Tseng PH, Lee YC, Chiang TH, Chen CC, et al. Early timing of single balloon enteroscopy is associated with increased diagnostic yield in patients with overt small bowel bleeding. J Formos Med Assoc. 2019 Dec;118(12):1644–51.
17 Perez-Cuadrado Robles E, Bebia Conesa P, Esteban Delgado P et al. Emergency doubleballoon enteroscopy combined with realtime viewing of capsule endoscopy: a feasible combined approach in acute overt-obscure gastrointestinal bleeding? Dig Endosc. 2015;27:338–44. https://doi.org/10.1111/den.12384.
18 Edwards AL, Mönkemüller K, Pamboukian SV, George JF, Wilcox CM, Peter S. Utility of double-balloon enteroscopy in patients with left ventricular assist devices and obscure overt gastrointestinal bleeding. Endoscopy. 2014 Nov;46(11):986–91.
19 Lin WP, Chiu CT, Su MY, Hsu CM, Sung CM, Chen PC. Treatment decision for potential bleeders in obscure gastrointestinal bleeding during double-balloon enteroscopy. Dig Dis Sci. 2009 Oct;54(10):2192–7.
20 Kaffes AJ, Siah C, Koo JH. Clinical outcomes after double-balloon enteroscopy in patients with obscure GI bleeding and a positive capsule endoscopy. Gastrointest Endosc. 2007 Aug;66(2):304–9.
21 Hsu CM, Chiu CT, Su MY, Lin WP, Chen PC, Chen CH. The outcome assessment of double-balloon enteroscopy for diagnosing and managing patients with obscure gastrointestinal bleeding. Dig Dis Sci. 2007 Jan;52(1):162–6.
22 Lipka S, Rabbanifard R, Kumar A, Brady P. A single-center United States experience with bleeding Dieulafoy lesions of the small bowel: diagnosis and treatment with single-balloon enteroscopy. Endosc Int Open. 2015 Aug;3(4):E339–45.
23 Chen YY, Chiu CT, Hsu CM, Chen TH, Chiu YC, Chu YC, et al. Enteroscopic Diagnosis and Management of Small Bowel Diverticular Hemorrhage: A Multicenter Report from the Taiwan Association for the Study of Small Intestinal Diseases. Gastroenterol Res Pract. 2015;2015:564536. [ Links ]
24 Pinto-Pais T, Pinho R, Rodrigues A, Fernandes C, Ribeiro I, Fraga J, et al. Emergency single-balloon enteroscopy in overt obscure gastrointestinal bleeding: efficacy and safety. United European Gastroenterol J. 2014 Dec;2(6):490–6.
25 Nelson KK, Lipka S, Davis-Yadley AH, Rodriguez AC, Doraiswamy V, Rabbanifard R, et al. Timing of single balloon enteroscopy: significant or not? Endosc Int Open. 2016 Jul;4(7):E761–6.
26 Aniwan S, Viriyautsahakul V, Angsuwatcharakon P, Kongkam P, Treeprasertsuk S, Rerknimitr R, et al. Comparison of urgent video capsule endoscopy and urgent doubleballoon endoscopy in massive obscure gastrointestinal bleeding. Hepatogastroenterology. 2014 Oct;61(135):1990–4.
27 Shinozaki S, Yamamoto H, Yano T, Sunada K, Hayashi Y, Shinhata H, et al. Favorable longterm outcomes of repeat endotherapy for small-intestine vascular lesions by doubleballoon endoscopy. Gastrointest Endosc. 2014 Jul;80(1):112–7.
28 Madisch A, Schmolders J, Brückner S, Aust D, Miehlke S. Less favorable clinical outcome after diagnostic and interventional double balloon enteroscopy in patients with suspected small-bowel bleeding? Endoscopy. 2008 Sep;40(9):731–4.
29 Tanaka S, Mitsui K, Tatsuguchi A, Kobayashi T, Ehara A, Gudis K, et al. Current status of double balloon endoscopyindications, insertion route, sedation, complications, technical matters. Gastrointest Endosc. 2007 Sep;66(3 Suppl):S30–3.
30 Hashimoto R, Nakahori M, Matsuda T. Impact of Urgent Double-Balloon Enteroscopy on the Short-Term and Long-Term Outcomes in Overt Small Bowel Bleeding. Dig Dis Sci. 2019 Oct;64(10):2933–8.
31 Hermans C, Stronkhorst A, Tjhie-Wensing A, Kamphuis J, Balkom BV, Dahlmans R, et al. Double-Balloon Endoscopy in Overt and Occult Small Bowel Bleeding: Results, Complications, and Correlation with Prior Videocapsule Endoscopy in a Tertiary Referral Center. Clin Endosc. 2017 Jan;50(1):69–75.
32 Nakamura M, Ohmiya N, Shirai O, Takenaka H, Morishima K, Miyahara R, et al. Route selection for double-balloon endoscopy, based on capsule transit time, in obscure gastrointestinal bleeding. J Gastroenterol. 2010 Jun;45(6):592–9.
33 Gerson LB, Batenic MA, Newsom SL et al. Long-term outcomes after double-balloon enteroscopy for obscure gastrointestinal bleeding. Clin Gastroenterol Hepatol. 2009;7:664–9. https://doi.org/10.1016/j.cgh.2009.01.021.
34 Moran RA, Barola S, Law JK, Amateau SK, Rolshud D, Corless E, et al. A Randomized Controlled Trial Comparing the Depth of Maximal Insertion Between Anterograde Single- Balloon Versus Spiral Enteroscopy. Clin Med Insights Gastroenterol. 2018 Jan;11:1179552218754881. [ Links ]
35 Shinozaki S, Yano T, Sakamoto H, Sunada K, Hayashi Y, Sato H, et al. Long-Term Outcomes in Patients with Overt Obscure Gastrointestinal Bleeding After Negative Double-Balloon Endoscopy. Dig Dis Sci. 2015 Dec;60(12):3691–6.
36 Sun B, Rajan E, Cheng S, Shen R, Zhang C, Zhang S, et al. Diagnostic yield and therapeutic impact of double-balloon enteroscopy in a large cohort of patients with obscure gastrointestinal bleeding. Am J Gastroenterol. 2006 Sep;101(9):2011–5.
37 Kaffes AJ, Koo JH, Meredith C. Double-balloon enteroscopy in the diagnosis and the management of small-bowel diseases: an initial experience in 40 patients. Gastrointest Endosc. 2006 Jan;63(1):81–6.
38 Zhu CN, Friedland J, Yan B, Wilson A, Gregor J, Jairath V, et al. Presence of Melena in Obscure Gastrointestinal Bleeding Predicts Bleeding in the Proximal Small Intestine. Dig Dis Sci. 2018 May;63(5):1280–5.
39 Shelnut DJ, Sims OT, Zaibaq JN, Oh H, Venkata KV, Peter S. Predictors for outcomes and readmission rates following double balloon enteroscopy: a tertiary care experience. Endosc Int Open. 2018 Jun;6(6):E751–7.
40 Prachayakul V, Deesomsak M, Aswakul P, Leelakusolvong S. The utility of single-balloon enteroscopy for the diagnosis and management of small bowel disorders according to their clinical manifestations: a retrospective review. BMC Gastroenterol. 2013 Jun;13(1):103. [ Links ]
41 Pinho R, Ponte A, Rodrigues A, Pinto-Pais T, Fernandes C, Ribeiro I, et al. Long-term rebleeding risk following endoscopic therapy of small-bowel vascular lesions with device-assisted enteroscopy. Eur J Gastroenterol Hepatol. 2016 Apr;28(4):479–85.
42 Santhakumar C, Liu K. Evaluation and outcomes of patients with obscure gastrointestinal bleeding. World J Gastrointest Pathophysiol. 2014 Nov;5(4):479–86.
43 Sidhu R, McAlindon ME, Drew K, Hardcastle S, Cameron IC, Sanders DS. Evaluating the role of small-bowel endoscopy in clinical practice: the largest single-centre experience. Eur J Gastroenterol Hepatol. 2012 May;24(5):513–9.
44 Möschler O, May A, Müller MK, Ell C; German DBE Study Group. Complications in and performance of double-balloon enteroscopy (DBE): results from a large prospective DBE database in Germany. Endoscopy. 2011 Jun;43(6):484–9.
45 Barreto-Zuñiga R, Tellez-Avila FI, Chavez- Tapia NC, Ramirez-Luna MA, Sanchez-Cortes E, Valdovinos-Andraca F, et al. Diagnostic yield, therapeutic impact, and complications of double-balloon enteroscopy in patients with small-bowel pathology. Surg Endosc. 2008 May;22(5):1223–6.
46 Manabe N, Tanaka S, Fukumoto A, Nakao M, Kamino D, Chayama K. Double-balloon enteroscopy in patients with GI bleeding of obscure origin. Gastrointest Endosc. 2006 Jul;64(1):135–40.
47 Hadithi M, Heine GD, Jacobs MA, van Bodegraven AA, Mulder CJ. A prospective study comparing video capsule endoscopy with double-balloon enteroscopy in patients with obscure gastrointestinal bleeding. Am J Gastroenterol. 2006 Jan;101(1):52–7.
48 Pasha SF, Leighton JA, Das A et al. Doubleballoon enteroscopy and capsule endoscopy have comparable diagnostic yield in smallbowel disease: a meta-analysis. Clin Gastroenterol Hepatol. 2008;6:671–6. https://doi.org/10.1016/j.cgh.2008.01.005.
49 Tee HP, Kaffes AJ. Non-small-bowel lesions encountered during double-balloon enteroscopy performed for obscure gastrointestinal bleeding. World J Gastroenterol. 2010 Apr;16(15):1885–9.
50 Chettiar R, Selby WS, Kaffes AJ. Antegrade double balloon enteroscopy for continued obscure gastrointestinal bleeding following push enteroscopy: is there a role? Dig Dis Sci. 2010 May;55(5):1381–4.
51 Jeon SR, Byeon JS, Jang HJ, Park SJ, Im JP, Kim ER, et al.; Small Intestine Research Group of the Korean Association for the Study of Intestinal Disease (KASID). Clinical outcome after enteroscopy for small bowel angioectasia bleeding: A Korean Associateion for the Study of Intestinal Disease (KASID) multiceter study. J Gastroenterol Hepatol. 2017 Feb;32(2):388–94.
52 Lara LF, Bloomfeld RS, Pineau BC. The rate of lesions found within reach of esophagogastroduodenoscopy during push enteroscopy depends on the type of obscure gastrointestinal bleeding. Endoscopy. 2005 Aug;37(8):745–50.
53 Ponte A, Perez-Cuadrado Robles E, Pinho R et al. High short-term rebleeding rate in patients undergoing a second endoscopic therapy for small-bowel angioectasias after recurrent bleeding. Rev Esp Enferm Dig. 2018;110:88–93.
54 Vakil N, Huilgol V, Khan I. Effect of push enteroscopy on transfusion requirements and quality of life in patients with unexplained gastrointestinal bleeding. Am J Gastroenterol. 1997 Mar;92(3):425–8.
55 Williamson JB, Judah JR, Gaidos JK, Collins DP, Wagh MS, Chauhan SS, et al. Prospective evaluation of the long-term outcomes after deep small-bowel spiral enteroscopy in patients with obscure GI bleeding. Gastrointest Endosc. 2012 Oct;76(4):771–8.
56 Jackson CS, Gerson LB. Management of gastrointestinal angiodysplastic lesions (GIADs): a systematic review and meta-analysis. Am J Gastroenterol. 2014 Apr;109(4):474–83.
57 Romagnuolo J, Brock AS, Ranney N. Is Endoscopic Therapy Effective for Angioectasia in Obscure Gastrointestinal Bleeding?: A Systematic Review of the Literature. J Clin Gastroenterol. 2015 Nov-Dec;49(10):823–30.
58 Yamamoto H, Ogata H, Matsumoto T et al. Clinical Practice Guideline for Enteroscopy. Digestive endoscopy : official journal of the Japan Gastroenterological Endoscopy Society 2017;29:519-546 https://doi.org/10.1111/den.12883. [ Links ]
59 Pinho R, Mascarenhas-Saraiva M, Mão-de-Ferro S, Ferreira S, Almeida N, Figueiredo P, et al. Multicenter survey on the use of deviceassisted enteroscopy in Portugal. United European Gastroenterol J. 2016 Apr;4(2):264–74.
60 May A, Friesing-Sosnik T, Manner H, Pohl J, Ell C. Long-term outcome after argon plasma coagulation of small-bowel lesions using double-balloon enteroscopy in patients with midgastrointestinal bleeding. Endoscopy. 2011 Sep;43(9):759–65.
61 Dulic-Lakovic E, Dulic M, Hubner D, Fuchssteiner H, Pachofszky T, Stadler B, et al.; Austrian Dieulafoy-bleeding Study Group. Bleeding Dieulafoy lesions of the small bowel: a systematic study on the epidemiology and efficacy of enteroscopic treatment. Gastrointest Endosc. 2011 Sep;74(3):573–80.
62 Pérez-Cuadrado-Robles EP, Moreels TG, Zamora-Nava LE, Blanco Velasco G, Esteban Delgado P, Samaha E, et al. A comprehensive systematic review and meta-analysis of risk factors for rebleeding following device-assisted enteroscopy therapy of small-bowel vascular lesions. Rev Esp Enferm Dig. 2020;in press. https://doi.org/10.17235/reed.2020.6802/2019. [ Links ]
63 Otani K, Watanabe T, Shimada S, Hosomi S, Nagami Y, Tanaka F, et al. Clinical Utility of Capsule Endoscopy and Double-Balloon Enteroscopy in the Management of Obscure Gastrointestinal Bleeding. Digestion. 2018;97(1):52–8.
64 Holleran G, Valerii G, Tortora A, Scaldaferri F, Conti S, Amato A, et al. The use of single balloon enteroscopy in Crohn’s disease and its impact on clinical outcome. Scand J Gastroenterol. 2018 Aug;53(8):925–9.
65 Yen HH, Chang CW, Chou JW, Wei SC. Balloon-Assisted Enteroscopy and Capsule Endoscopy in Suspected Small Bowel Crohn’s Disease. Clin Endosc. 2017 Sep;50(5):417–23.
66 Takenaka K, Ohtsuka K, Kitazume Y, Nagahori M, Fujii T, Saito E, et al. Comparison of magnetic resonance and balloon enteroscopic examination of the small intestine in patients with Crohn’s disease. Gastroenterology. 2014 Aug;147(2):334–342.e3.
67 Navaneethan U, Lourdusamy V, Njei B, Shen B. Endoscopic balloon dilation in the management of strictures in Crohn’s disease: a systematic review and meta-analysis of nonrandomized trials. Surg Endosc. 2016 Dec;30(12):5434–43.
68 Lopes S, Rodrigues-Pinto E, Andrade P, Afonso J, Baron TH, Magro F, et al. Endoscopic balloon dilation of Crohn’s disease strictures-safety, efficacy and clinical impact. World J Gastroenterol. 2017 Nov;23(41):7397–406.
69 Hirai F, Andoh A, Ueno F, Watanabe K, Ohmiya N, Nakase H, et al. Efficacy of Endoscopic Balloon Dilation for Small Bowel Strictures in Patients With Crohn’s Disease: A Nationwide, Multi-centre, Open-label, Prospective Cohort Study. J Crohn’s Colitis. 2018 Mar;12(4):394–401.
70 Arulanandan A, Dulai PS, Singh S, Sandborn WJ, Kalmaz D. Systematic review: safety of balloon assisted enteroscopy in Crohn’s disease. World J Gastroenterol. 2016 Oct;22(40):8999–9011.
71 Bourreille A, Ignjatovic A, Aabakken L, Loftus EV Jr, Eliakim R, Pennazio M, et al.; World Organisation of Digestive Endoscopy (OMED) and the European Crohn’s and Colitis Organisation (ECCO). Role of small-bowel endoscopy in the management of patients with inflammatory bowel disease: an international OMED-ECCO consensus. Endoscopy. 2009 Jul;41(7):618–37.
72 Riff BP, DiMaio CJ. Exploring the Small Bowel: Update on Deep Enteroscopy. Curr Gastroenterol Rep. 2016 Jun;18(6):28. [ Links ]
73 Kim M, Jang HJ. The role of small bowel endoscopy in small bowel Crohn’s disease: when and how? Intest Res. 2016 Jul;14(3):211–7.
74 Ching HL, McAlindon ME, Sidhu R. An update on small bowel endoscopy. Curr Opin Gastroenterol. 2017 May;33(3):181–8.
75 Tun GS, Rattehalli D, Sanders DS, McAlindon ME, Drew K, Sidhu R. Clinical utility of double-balloon enteroscopy in suspected Crohn’s disease: a single-centre experience. Eur J Gastroenterol Hepatol. 2016 Jul;28(7):820–5.
76 Baars JE, Theyventhiran R, Aepli P, Saxena P, Kaffes AJ. Double-balloon enteroscopy-assisted dilatation avoids surgery for small bowel strictures: A systematic review. World J Gastroenterol. 2017 Dec;23(45):8073–81.
77 Sunada K, Shinozaki S, Nagayama M, Yano T, Takezawa T, Ino Y, et al. Long-term Outcomes in Patients with Small Intestinal Strictures Secondary to Crohn’s Disease After Double-balloon Endoscopy-assisted Balloon Dilation. Inflamm Bowel Dis. 2016 Feb;22(2):380–6.
78 Rondonotti E, Spada C, Adler S, May A, Despott EJ, Koulaouzidis A, et al. Smallbowel capsule endoscopy and device-assisted enteroscopy for diagnosis and treatment of small-bowel disorders: European Society of Gastrointestinal Endoscopy (ESGE) Technical Review. Endoscopy. 2018 Apr;50(4):423–46.
79 Rondonotti E, Pennazio M, Toth E, Menchen P, Riccioni ME, De Palma GD, et al.; European Capsule Endoscopy Group; Italian Club for Capsule Endoscopy (CICE); Iberian Group for Capsule Endoscopy. Smallbowel neoplasms in patients undergoing video capsule endoscopy: a multicenter European study. Endoscopy. 2008 Jun;40(6):488–95.
80 Bilimoria KY, Bentrem DJ, Wayne JD, Ko CY, Bennett CL, Talamonti MS. Small bowel cancer in the United States: changes in epidemiology, treatment, and survival over the last 20 years. Ann Surg. 2009 Jan;249(1):63–71.
81 Zouhairi ME, Venner A, Charabaty A, Pishvaian MJ. Small bowel adenocarcinoma. Curr Treat Options Oncol. 2008 Dec;9(4-6):388–99.
82 Mitsui K, Tanaka S, Yamamoto H, Kobayashi T, Ehara A, Yano T, et al. Role of double-balloon endoscopy in the diagnosis of small-bowel tumors: the first Japanese multicenter study. Gastrointest Endosc. 2009 Sep;70(3):498–504.
83 Robles EP, Delgado PE, Conesa PB, Andrés BM, Guggiana MF, Mateos EA, et al. Role of double-balloon enteroscopy in malignant small bowel tumors. World J Gastrointest Endosc. 2015 Jun;7(6):652–8.
84 Sulbaran M, de Moura E, Bernardo W, Morais C, Oliveira J, Bustamante-Lopez L, et al. Overtube- assisted enteroscopy and capsule endoscopy for the diagnosis of small-bowel polyps and tumors: a systematic review and meta-analysis. Endosc Int Open. 2016 Feb;4(2):E151–63.
85 Honda W, Ohmiya N, Hirooka Y, Nakamura M, Miyahara R, Ohno E, et al. Enteroscopic and radiologic diagnoses, treatment, and prognoses of small-bowel tumors. Gastrointest Endosc. 2012 Aug;76(2):344–54.
86 Lee BI, Choi H, Choi KY, Byeon JS, Jang HJ, Eun CS, et al. Clinical characteristics of small bowel tumors diagnosed by double-balloon endoscopy: KASID multi-center study. Dig Dis Sci. 2011 Oct;56(10):2920–7.
87 Chen WG, Shan GD, Zhang H, Yang M, L L, Yue M, et al. Double-balloon enteroscopy in small bowel diseases: eight years single-center experience in China. Medicine (Baltimore). 2016 Oct;95(42):e5104. [ Links ]
88 He Q, Bai Y, Zhi FC, Gong W, Gu HX, Xu ZM, et al. Double-balloon enteroscopy for mesenchymal tumors of small bowel: nine years’ experience. World J Gastroenterol. 2013 Mar;19(11):1820–6.
89 Imaoka H, Higaki N, Kumagi T, Miyaike J, Ohmoto M, Yamauchi K, et al. Characteristics of small bowel tumors detected by double balloon endoscopy. Dig Dis Sci. 2011 Aug;56(8):2366–71.
90 Almeida N, Figueiredo P, Lopes S, Gouveia H, Leitão MC. Double-balloon enteroscopy and small bowel tumors: a South-European single- center experience. Dig Dis Sci. 2009 Jul;54(7):1520–4.
91 Cardoso H, Rodrigues JT, Marques M, Ribeiro A, Vilas-Boas F, Santos-Antunes J, et al. Malignant Small Bowel Tumors: Diagnosis, Management and Prognosis. Acta Med Port. 2015 Jul-Aug;28(4):448–56.
92 Nakano A, Nakamura M, Watanabe O, Yamamura T, Funasaka K, Ohno E, et al. Endoscopic Characteristics, Risk Grade, and Prognostic Prediction in Gastrointestinal Stromal Tumors of the Small Bowel. Digestion. 2017;95(2):122–31.
93 Mitsui K, Tanaka S, Yamamoto H, Kobayashi T, Ehara A, Yano T, et al. Role of double-balloon endoscopy in the diagnosis of smallbowel tumors: the first Japanese multicenter study. Gastrointest Endosc. 2009 Sep;70(3):498–504.
94 Gangi A, Siegel E, Barmparas G, Lo S, Jamil LH, Hendifar A, et al. Multifocality in Small Bowel Neuroendocrine Tumors. J Gastrointest Surg. 2018 Feb;22(2):303–9.
95 Ross AS, Semrad C, Waxman I, Dye C. Enteral stent placement by double balloon enteroscopy for palliation of malignant small bowel obstruction. Gastrointest Endosc. 2006 Nov;64(5):835–7.
96 Hayashi Y, Yamamoto H, Kita H, Sunada K, Miyata T, Yano T, et al. Education and imaging. Gastrointestinal: metallic stent for an obstructing jejunal cancer. J Gastroenterol Hepatol. 2006 Dec;21(12):1861. [ Links ]
97 Kida A, Matsuda K, Noda Y. Endoscopic metallic stenting by double-balloon enteroscopy and its overtube for malignant gastrointestinal obstruction as palliative treatment. Dig Endosc. 2013;25:552–3. https://doi.org/10.1111/den.12136.
98 Espinel J, Pinedo E. A simplified method for stent placement in the distal duodenum: enteroscopy overtube. World J Gastrointest Endosc. 2011 Nov;3(11):225–7.
99 Nakamura M, Ohmiya N, Hirooka Y, Miyahara R, Ando T, Watanabe O, et al. Endoscopic diagnosis of follicular lymphoma with small-bowel involvement using video capsule endoscopy and double-balloon endoscopy: a case series. Endoscopy. 2013;45(1):67–70.
100 Schulmann K, Hollerbach S, Kraus K, Willert J, Vogel T, Möslein G, et al. Feasibility and diagnostic utility of video capsule endoscopy for the detection of small bowel polyps in patients with hereditary polyposis syndromes. Am J Gastroenterol. 2005 Jan;100(1):27–37.
101 Rahmi G, Samaha E, Lorenceau-Savale C, Landi B, Edery J, Manière T, et al. Small bowel polypectomy by double balloon enteroscopy: correlation with prior capsule endoscopy. World J Gastrointest Endosc. 2013 May;5(5):219–25.
102 Goverde A, Korsse SE, Wagner A, van Leerdam ME, Krak NC, Stoker J, et al. Smallbowel Surveillance in Patients With Peutz- Jeghers Syndrome: Comparing Magnetic Resonance Enteroclysis and Double Balloon Enteroscopy. J Clin Gastroenterol. 2017 Apr;51(4):e27–33.
103 Ohmiya N, Nakamura M, Takenaka H, Morishima K, Yamamura T, Ishihara M, et al. Management of small-bowel polyps in Peutz-Jeghers syndrome by using enteroclysis, double-balloon enteroscopy, and videocapsule endoscopy. Gastrointest Endosc. 2010 Dec;72(6):1209–16.
104 Torroni F, Romeo E, Rea F, Angelis PD, Foschia F, Faraci S, et al. Conservative approach in Peutz-Jeghers syndrome: single-balloon enteroscopy and small bowel polypectomy. World J Gastrointest Endosc. 2014 Jul;6(7):318–23.
105 Beggs AD, Latchford AR, Vasen HF, Moslein G, Alonso A, Aretz S, et al. Peutz-Jeghers syndrome: a systematic review and recommendations for management. Gut. 2010 Jul;59(7):975–86.
106 Sakamoto H, Yamamoto H, Hayashi Y, Yano T, Miyata T, Nishimura N, et al. Nonsurgical management of small-bowel polyps in Peutz-Jeghers syndrome with extensive polypectomy by using double-balloon endoscopy. Gastrointest Endosc. 2011 Aug;74(2):328–33.
107 Serrano M, Mao-de-Ferro S, Pinho R et al. Double-balloon enteroscopy in the management of patients with Peutz-Jeghers syndrome: a retrospective cohort multicenter study. Rev Esp Enferm Dig. 2013;105:594–9. https://doi.org/10.4321/S1130-01082013001000004.
108 Wang YX, Bian J, Zhu HY, Dong YH, Fang AQ, Li ZS, et al. The role of double-balloon enteroscopy in reducing the maximum size of polyps in patients with Peutz-Jeghers syndrome: 12-year experience. J Dig Dis. 2019 Aug;20(8):415–20.
109 Gao H, van Lier MG, Poley JW, Kuipers EJ, van Leerdam ME, Mensink PB. Endoscopic therapy of small-bowel polyps by doubleballoon enteroscopy in patients with Peutz-Jeghers syndrome. Gastrointest Endosc. 2010 Apr;71(4):768–73.
110 Belsha D, Urs A, Attard T, Thomson M. Effectiveness of Double-balloon Enteroscopyfacilitated Polypectomy in Pediatric Patients With Peutz-Jeghers Syndrome. J Pediatr Gastroenterol Nutr. 2017 Nov;65(5):500–2.
111 Sulbaran M, Campos FG, Ribeiro U Jr, Kishi HS, Sakai P, de Moura EG, et al. Risk factors for advanced duodenal and ampullary adenomatosis in familial adenomatous polyposis: a prospective, single-center study. Endosc Int Open. 2018 May;6(5):E531–40.
112 Tescher P, Macrae FA, Speer T, Stella D, Gibson R, Tye-Din JA, et al. Surveillance of FAP: a prospective blinded comparison of capsule endoscopy and other GI imaging to detect small bowel polyps. Hered Cancer Clin Pract. 2010 Apr;8(1):3. [ Links ]
113 Matsumoto T, Esaki M, Yanaru-Fujisawa R, Moriyama T, Yada S, Nakamura S, et al. Small-intestinal involvement in familial adenomatous polyposis: evaluation by doubleballoon endoscopy and intraoperative enteroscopy. Gastrointest Endosc. 2008 Nov;68(5):911–9.
114 Alderlieste YA, Rauws EA, Mathus-Vliegen EM, Fockens P, Dekker E. Prospective enteroscopic evaluation of jejunal polyposis in patients with familial adenomatous polyposis and advanced duodenal polyposis. Fam Cancer. 2013 Mar;12(1):51–6.
115 Kav T, Sokmensuer C, Sivri B. Enteroscopic findings of Celiac Disease and their correlation with mucosal histopathologic changes. Comput Biol Med. 2015 Oct;65:315–9.
116 Malamut G, Cellier C. Refractory coeliac disease. Curr Opin Oncol. 2013 Sep;25(5):445–51.
117 Elli L, Casazza G, Locatelli M, Branchi F, Ferretti F, Conte D, et al. Use of enteroscopy for the detection of malignant and premalignant lesions of the small bowel in complicated celiac disease: a meta-analysis. Gastrointest Endosc. 2017 Aug;86(2):264–273.e1.
118 Hadithi M, Al-toma A, Oudejans J, van Bodegraven AA, Mulder CJ, Jacobs M. The value of double-balloon enteroscopy in patients with refractory celiac disease. Am J Gastroenterol. 2007 May;102(5):987–96.
119 Tomba C, Sidhu R, Sanders DS, Mooney PD, Branchi F, Locatelli M, et al. Celiac Disease and Double-Balloon Enteroscopy: What Can We Achieve?: The Experience of 2 European Tertiary Referral Centers. J Clin Gastroenterol. 2016 Apr;50(4):313–7.
120 Perez-Cuadrado E, Mas P, Hallal H et al. Double-balloon enteroscopy: a descriptive study of 50 explorations. Rev Esp Enferm Dig. 2006;98:73–81. https://doi.org/10.4321/S1130-01082006000200002.
121 Nath NC, Dhar SC, Uddin AF, Shil BC, Banik RK, Saha SK, et al. Double balloon enteroscopy: bangladesh experience. Bangladesh Med Res Counc Bull. 2013 Dec;39(3):116–9.
122 Diaz Alcazar MDM, Martin-Lagos Maldonado A, Roa Colomo A et al. Enteroscopic diagnosis of intestinal malabsorption of a rare etiology: diaphragm disease. Rev Esp Enferm Dig. 2020;112. https://doi.org/10.17235/reed.2020.6723/2019. [ Links ]
123 Yu H, Song H, Cai J. Laparoscopic treatment of hemorrhagic Meckel diverticulum after diagnosis with wireless capsule endoscopy and double-balloon enteroscopy. Rev Esp Enferm Dig. 2017;109:315. https://doi.org/10.17235/reed.2017.4522/2016. [ Links ]
124 Keren D, Madi H, Matter I, Assalia A, Rainis T. Feasibility and Importance of Enteroscopy after Gastric Bypass. Obes Surg. 2017 May;27(5):1309–15.
125 Mitsui K, Fujimori S, Tanaka S, Ehara A, Omori J, Akimoto N, et al. Retrieval of Retained Capsule Endoscopy at Small Bowel Stricture by Double-Balloon Endoscopy Significantly Decreases Surgical Treatment. J Clin Gastroenterol. 2016 Feb;50(2):141–6.
126 Shiotsuki K IK, Hotta K. Small bowel obstruction by massive impacted diospyrobezoars relieved by endoscopic lithotripsy with a polypectomy snare using double balloon endoscopy. Dig Endosc. 2019;31. [ Links ]
127 Mönkemüller K, Zabielski M, Poppen D, Fry LC. Endoscopic removal of an impacted root canal needle in the jejunum using doubleballoon enteroscopy. Gastrointest Endosc. 2011 Apr;73(4):844–6.
128 Neumann H, Fry LC, Rickes S, Jurczok C, Malfertheiner P, Mönkemüller K. A ‘double- balloon enteroscopy worth the money’: endoscopic removal of a coin lodged in the small bowel. Dig Dis. 2008;26(4):388–9.
129 Jeon SR, Kim JO, Kim HG, Lee TH, Kim WJ, Cho JY, et al. Migrated anchoring gastric band removed by double-balloon enteroscopy. Gastrointest Endosc. 2011 Jul;74(1):225–7.
130 Murino A, Koukias N, Gaeta L, Despott EJ. Emergency double-balloon enteroscopy for retrieval of an ingested dental-bridge, partially embedded within the wall of the jejunum. Dig Liver Dis. 2018 May;50(5):516. [ Links ]
131 Gutierrez JP, Wilcox CM, Mönkemüller K. Endoscopic removal of a migrated duodenal stent into the ileum using double-balloon enteroscopy. Gastrointest Endosc. 2015 Apr;81(4):1033–4.
132 Nishimura N, Yamamoto H, Yano T, Hayashi Y, Sato H, Miura Y, et al. Balloon dilation when using double-balloon enteroscopy for small-bowel strictures associated with ischemic enteritis. Gastrointest Endosc. 2011 Nov;74(5):1157–61.
133 Hayashi Y, Yamamoto H, Taguchi H, Sunada K, Miyata T, Yano T, et al. Nonsteroidal anti-inflammatory drug-induced smallbowel lesions identified by double-balloon endoscopy: endoscopic features of the lesions and endoscopic treatments for diaphragm disease. J Gastroenterol. 2009;44(S19 Suppl 19):57–63.
134 Nakahara K, Okuse C, Matsumoto N, Suetani K, Morita R, Michikawa Y, et al. Enteral metallic stenting by balloon enteroscopy for obstruction of surgically reconstructed intestine. World J Gastroenterol. 2015 Jun;21(24):7589–93.
135 Popa D, Ramesh J, Peter S, Wilcox CM, Mönkemüller K. Small Bowel Stent-in-Stent Placement for Malignant Small Bowel Obstruction Using a Balloon-Assisted Overtube Technique. Clin Endosc. 2014 Jan;47(1):108–11.
136 Perez-Cuadrado E, Carballo F, Latorre R et al. An endoscopic technique for treating symptomatic distal jejunum obstruction by leaving the overtube in place. Rev Esp Enferm Dig. 2013;105:107–9. https://doi.org/10.4321/S1130-01082013000200009.
137 Bernardes C, Pinho R, Rodrigues A et al. Direct percutaneous endoscopic jejunostomy using single-balloon enteroscopy without fluoroscopy: a case series. Rev Esp Enferm Dig. 2017;109:679–83. https://doi.org/10.17235/reed.2017.4717/2016.
138 Miura Y, Yamamoto H, Sunada K, Yano T, Arashiro M, Miyata T, et al. Reduction of ileoileal intussusception by using double-balloon endoscopy in Peutz-Jeghers syndrome (with video). Gastrointest Endosc. 2010 Sep;72(3):658–9.
139 Pinho R. The Vanishing Frontiers of Therapeutic Enteroscopy. GE Port J Gastroenterol. 2015 Jul;22(4):133–4.
Disclosure Statement
The authors have nothing to disclose.
* Corresponding author.
Enrique Pérez-Cuadrado-Robles
Department of Gastroenterology, Georges-Pompidou European Hospital
20 Rue Leblanc
FR–75015 Paris (France)
Received: March 1, 2020; Accepted: March 19, 2020
Enrique Pérez-Cuadrado-Robles and Rolando Pinho are first co-authors and contributed equally to the present guidelines. Note: This article is published simultaneously in GE – Portuguese Journal of Gastroenterology, DOI: 10.1159/000507375, and The Spanish Journal of Gastroenterology, DOI: 10.17235/reed.2020.7020/2020, with the consent of the authors and editors.














