Introduction
Hyperplastic polyps represent 30-93% of all gastric epithelial polyps [1, 2]. They are characterized by hyperplastic foveola with an inflamed stroma, generally arising in response to a chronic inflammatory environment. Their pathogenesis is unclear. The accepted hypothesis states that gastric hyperplastic polyps (GHP) develop as the result of a hyperplastic mucosal healing regenerative response secondary to an inflammatory process [3]. They are generally detected as innocuous incidental findings in 1.2-8% of patients undergoing esophago-gastro-duodenoscopy (EGD). However, they can be present as anaemia due to occult bleeding or gastric outlet obstruction [1, 4]. Several risk factors for GHP have been identified, such as Helicobacter pylori infection [5], chronic atrophic gastritis [6], portal hypertension [5, 7], autoimmune gastritis [8], gastric surgery [9], and Ménétrier disease. Hypergastrinemia, induced by hypochlorhydria caused by chronic atrophic gastritis, has also been implicated in the pathogenesis of GHP [3]. EGD with white light shows sessile or pedunculated reddish lesions, generally < 20 mm in diameter and usually occurring as single lesions in the antrum (Fig. 1a). Narrow-band imaging typically shows a large regular mucosal pattern (Fig. 1b) [2]. Upon magnification of narrow-band imaging, GHP present a uniformly long, narrow, and elliptical marginal crypt epithelium with an unclear microvascular pattern [10].
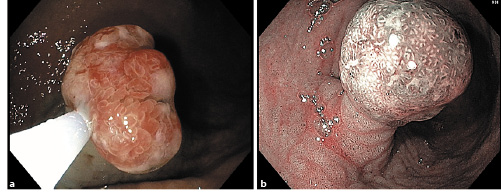
Fig. 1 Endoscopic typical appearance of gastric hyperplastic polyps (GHP). Esophagogastroduodenoscopy showing a typical reddish aspect on white-light imaging (a) and a large regular mucosal pattern on narrow-band imaging (b).
Their potential for neoplastic change has been demonstrated for lesions with a diameter > 10 mm, with a prevalence of dysplasia (low- and high-grade) ranging from 1.5 to 4.4%, and a prevalence of carcinoma between 1.1 and 2.1%. Horiuchi et al. [11] reported that combining a polyp size > 20 mm and features of magnification endoscopy with narrow-band imaging demonstrating an abnormal microvascular pattern and size reduction of the fine mucosal structure has nearly 100% sensitivity and 58% specificity for a diagnosis of cancer or dysplasia. However, the mechanisms responsible for the neoplastic change remain unknown [8]. Forté et al. [12] reported local recurrence in more than half of GHP > 10 mm in size after endoscopic resection. Unlike gastric adenomas and early gastric cancer, en bloc complete resection with free lateral and deep margins does not prevent local recurrence. Risk factors for local recurrence are not well described.
Current guidelines recommend endoscopic resection of all GHP > 5-10 mm and biopsy of the remaining tissue. Surrounding gastric mucosa should also be biopsied according to the management of epithelial pre-cancerous conditions and lesions in the stomach (MAPS II) guideline [13]. Additionally, H. pylori infection should be assessed and treated if present. Endoscopic follow-up is required 1 year after biopsy or endoscopic resection. If no residual polyp is found on surveillance endoscopy, further follow-up is according to the MAPS II guideline for the findings in the surrounding mucosa [1, 2].
Recent knowledge concerning recurrence and neoplastic transformation rates as well as their predictors are in line with changes in current management of this type of polyp. The identification of high-risk factors for recurrence and neoplastic change will allow a better and selective management of these patients.
This study intended to characterize GHP population in a Western country and evaluate the proportion and risk factors for GHP recurrence and neoplastic transformation.
Materials and Methods
Study Design
This was a single-centre, retrospective, cohort study performed from January 2009 to June 2020.
Inclusion and Exclusion Criteria
Patients aged ≥18 years who underwent endoscopic resection of GHP were consecutively included. Patients were excluded if the GHP was already a recurrence at the time of the first endoscopy, or if they underwent surgical resection of GHP, or had a hereditary gastric polyposis syndrome.
Definitions
The histological diagnosis of GHP was based on the following criteria: the presence of foveolar hyperplasia with long, deep, and hypersecreting crypts, or an inflammatory and abundant chorion. Neoplastic transformation was defined histologically by the presence of dysplasia or adenocarcinoma within GHP. Recurrence was defined by the presence of a histologically confirmed GHP at the previous complete resection of GHP at least 3 months after the initial resection.
Demographic, Clinical, Endoscopic, and Histologic Variables
Patients’ characteristics, i.e., age, gender, proton-pump inhibitor intake, a history of gastric surgery, and the haemoglobin value were retrieved from electronic reports. Endoscopic data, i.e., GHP size, location, and morphology according to the Paris classification [14], endoscopic resection procedure, and adverse events were collected from EGD report. Histologic data, i.e., H. pylori infection, atrophic gastritis, intestinal metaplasia, neoplastic transformation and type, and complete resection with free lateral and deep margins were retrieved from the pathology report.
Data and Statistical Analysis
Continuous variables are reported as mean and SD or median and range if they had a normal or skewed distribution, respectively. Categorical variables are reported as absolute and relative frequencies. Continuous variables were compared between 2 groups using Student’s t test if they had normal distribution and homogeneity of variance, or the Mann-Whitney U test if these conditions were not met. Categorical variables were compared using Pearson’s χ2 test or Fisher’s exact test. To determine factors associated with recurrence and neoplastic transformation, univariate and multivariate analysis using a logistic regression model was performed. Covariates with a p value < 0.05 in the univariate analysis were included in the multivariate analysis. The effect of factors on the risk of recurrence and neoplastic transformation were quantified using odds ratio (OR) with 95% confidence interval (CI) in both analyses. Cumulative probabilities of recurrence were estimated by the Kaplan-Meier method. All hypotheses are 2-tailed and a p value < 0.05 was considered statistically significant. Statistical analysis was performed using SPSS v25 (SPSS Inc., Chicago, IL, USA).
Results
In our study, from 364 epithelial gastric polyps submitted for endoscopic resection, 53.5% (n = 195) were GHP. Of the 195 patients included, 56% (n = 109) were female and the median age was 67 (35-87) years. The indications for performing the EGD were screening for early gastric cancer (47%, n = 91), anaemia (24%, n = 46), dyspepsia (11%, n = 21), and epigastric pain (9%, n = 18). The median size of GHP was 10 (3-50) mm and the majority were located in the antrum (61%, n = 119) and had a sessile morphology (62%, n = 120). Synchronous lesions, with inferior size, were present in 36% (n = 71) of patients. Histologic findings of surrounding mucosa showed atrophic gastritis in 87% (n = 169), intestinal metaplasia in 19% (n = 37), and dysplasia in 3.6% (n = 7). H. pylori infection was detected in 29% (n = 57) of the patients without any particular location; 41% were H. pylori-positive and 59% were H. pylori-negative (p = 0.8). Gastric surgery was described in 5.6% (n = 11) patients. Proton-pump inhibitor intake was reported in 40% (n = 78).
Hot-snare polypectomy was the technique for endoscopic resection performed in 38% (n = 75) of the lesions, corresponding to pedunculated or sub-pedunculated lesions. Endoscopic mucosal resection was performed in the remaining 62% (n = 120), corresponding to sessile lesions. No cold-snare polypectomies were performed. There were 16.7% (n = 20) piecemeal resections. Post-polypectomy bleeding was recorded in 4% (n = 8) of the cases, corresponding to 5 cases of intra-procedural bleeding managed with clips and/or snare-tip soft coagulation, and 3 cases of post-procedural bleeding. Neither perforation- nor sedation-related adverse events were reported (Table 1).
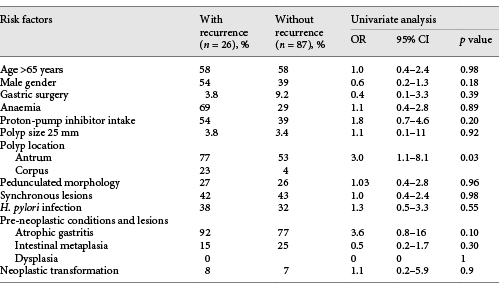
Of the study population, 58% (n = 113) patients had at least 1 EGD follow-up. The median time for the first EGD was 1 (1-5) years, with 94% (n = 106) performed at 1 year. After the initial resection, 23% (n = 26) recurrences were identified, 69% (n = 18) of which were detected at the first EGD follow-up. Median size of GHP with recurrence was 10 (6-40) mm, 80% (n = 20) were in the antrum, and 43% (n = 11) had synchronic lesions. H. pylori infection was reported in 39% (n = 10) cases. Regarding pre-malignant conditions, in patients with recurrence, atrophic gastritis was present in 92% (n = 24) and intestinal metaplasia in 16% (n = 4). Dysplasia was not reported. Anaemia was present in 69% (n = 18) of the patients. Recurrence was significantly higher in polyps located in the antrum than in the corpus/fundus (80 vs. 53%, OR 3.0; 95% CI 1.1-8.1). Gender, age, polyp size and morphology, H. pylori status, pre-malignant gastric conditions in the surrounding mucosa, and haemoglobin value were not associated with recurrence (Table 2). Of the 26 recurrent GHP, 15 were submitted for a new endoscopic resection. In the remaining patients, the biopsy confirmed hyperplastic origin and no further treatment or surveillance was performed. After the first re-treatment, 4 were lost to follow-up. A recurrence frequency of 36% (4/11) was reported after endoscopic resection of the first recurrence. The median time to recurrence was estimated at 7.5 (95% CI 6.7-8.4) years (Fig. 2).
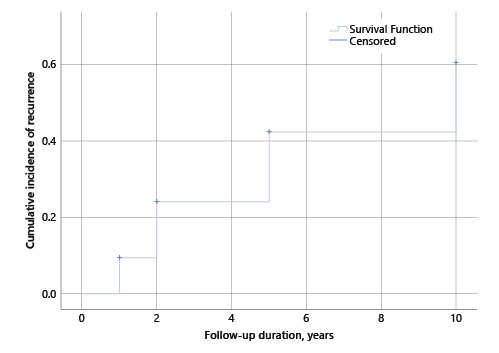
Fig. 2 Estimation of cumulative incidence of recurrence after first endoscopic resection of gastric hyperplastic polyps (GHP), 95% CI.
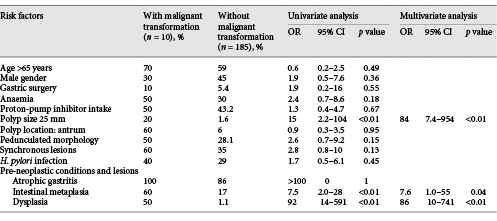
Neoplastic transformation was identified in 10 GHP, 5 with low-grade dysplasia, 4 with high-grade dysplasia, and 1 with an intra-mucosal carcinoma. Median size of GHP with neoplastic transformation was 17 (10-50) mm, 60% (n = 6) of GHP were located in the antrum, and 60% (n = 6) had synchronic lesions. H. pylori infection was reported in 40% (n = 4). In these patients with a GHP with neoplastic transformation, pre-malignant conditions in adjacent mucosa were present in 100% (n = 10), 100% (n = 10) had chronic gastritis, 60% (n = 6) had intestinal metaplasia, and 50% (n = 5) had dysplasia (Table 3).
Univariate analysis found that a GHP size > 25 mm, intestinal metaplasia, and/or dysplasia in surrounding mucosa were associated with neoplastic transformation. Neoplastic transformation was significantly higher in GHP with a diameter > 25 mm (80 vs. 20%; OR 15; 95% CI 2.2-104), and with intestinal metaplasia (60 vs. 17%; OR 7.5; 95% CI 2.0-28), or dysplasia (50 vs. 1.0%; OR 92; 95% CI 14-591) in adjacent mucosa. Multivariate analysis after backward selection found that a size > 25 mm (OR 84; 95% CI 7.4-954), intestinal metaplasia (OR 7.6; 95% CI 1.0-55), and dysplasia (OR 86; 95% CI 10-741) remained significantly associated with neoplastic transformation (Table 3).
Discussion
In our population, the frequency of GHP was higher than reported in studies from the USA and from other European countries, but similar to that reported in a Brazilian study [15-17]. This data is in line with the high prevalence of H. pylori infection in Portugal compared with other Western countries [18]. As reported in previous studies, GHP were more frequent in females [15]. Endoscopic findings are in accordance with previous studies regarding median size, sessile morphology, and antrum location [1]. The proportion of H. pylori infection at the time of endoscopic resection (29%) was comparable with previous data descriptions of percentages between 15 and 37% [12, 19]. However, this proportion was lower than expected, taking into account the prevalence of H. pylori infection in Portugal [18]; it can be explained by previous eradication and false-negatives introduced by proton-pump inhibitor intake in a considerable percentage of the patients (40%). Furthermore, Giemsa coloration of histopathologic specimens was the diagnostic test used for H. pylori detection. This method has a lower sensitivity (83%) than immunohistochemistry and fluorescence in situ hybridization; this, together with the high frequency of atrophic gastritis (87%), may have led to an underestimation of the real prevalence of H. pylori infection [20]. Concerning the advanced stages of gastritis, gastric atrophy was present in 87%, intestinal metaplasia in 19%, and dysplasia in 3.6% of our patients. Previous data also demonstrated the higher prevalence of epithelial pre-cancerous conditions in patients with GHP [3, 8]. In our study, the updated Sydney system was not used, defaulting further conclusions [21].
The study showed local recurrence in 23% of the patients. It revealed that GHP recurrence was more frequent for polyps located in the antrum, in agreement with a previous study conducted by Forté et al. [12]. One of the possible hypotheses for taking into account GHP location is that the mucosal trauma induced by contractions of the antrum or duodeno-gastric reflux could promote the hyperplastic wound-healing process [12, 22]. The mechanism of recurrence remains unclear. However, it seems that it can be explained, at least in part, by the iteration of the wound-healing process. Recurrent lesions can be treated by endoscopic resection, as described in most cases, despite the presence of sub-mucosal fibrosis.
Similar to the findings of Forté et al. [12], in our cohort, GHP > 25 mm had a higher neoplastic potential as well as advanced gastritis stages such as intestinal metaplasia and dysplasia. Although intestinal metaplasia and atrophic gastritis have been reported as risk factors in some studies [3, 19], others found no correlation between these factors and the occurrence of neoplasia [23]. A Japanese study suggested that GHP with neoplastic changes develop via a hyperplasia-dysplasia-carcinoma sequence rather than an intestinal metaplasia-dysplasia-carcinoma sequence [24]. Given the controversial data, whether therapy should be directed toward the underlying pathology rather than the polyp itself remains a relevant question under investigation [25]. Hypergastrinemia, not evaluated in our cohort, has also been implicated in neoplastic transformation, namely in the autoimmune gastritis setting [8].
In this study, recurrence was not a risk factor for neoplastic change. No previous study had demonstrated a positive association between recurrence and neoplastic change.
This study has some limitations. First, it was performed in a single tertiary centre and was designed retrospectively. Therefore, some data is missing, namely H. pylori eradication, the haemoglobin value, and current medication. Additionally, an adequate staging and grading of intestinal metaplasia and gastric atrophy, with biopsies of at least 2 topographic sites (from both the antrum and the corpus, at the lesser and greater curvature) taken and clearly labelled in to 2 separate vials [13], was performed only in 21% of patients. Although recurrence was defined as GHP developing at the resection scar, it is possible that some of the new GHP were mistakenly considered as recurrent lesions, owing to close proximity, a faded scar, or evaluation by a different endoscopist. Finally, endoscopic surveillance intervals were based on the clinician’s decision and did not follow a uniform protocol. A uniform endoscopic follow-up protocol and a prospective cohort study seem warranted to confirm our conclusions and better define recurrence time, patterns, and management as well as risk factors for neoplastic transformation.
In conclusion, this study characterized population with GHP polyps in a Western country and evaluated the risk factors for recurrence and neoplastic transformation. Lesions > 25 mm with advanced stages of atrophic gastritis require endoscopic resection, and gastric mucosa needs endoscopic surveillance. However, in the absence of other high-risk features, particularly intestinal metaplasia and dysplasia in the surrounding mucosa, endoscopic resection and follow-up of smaller lesions (10-25 mm) should be more selectively performed. Recurrence, frequent in GHP, is not a predictor of neoplastic transformation. In summary, a tailored endoscopic approach for GHP could reduce costs and morbidity as well as optimize the use of human and material resources, in line with the recently published guidelines of the European Society of Gastrointestinal Endoscopy [26].