Introduction
Surgical treatment of esophagogastric cancer is associated with significant mortality and morbidity rates. Esophagectomy’s mortality and morbidity are reported to be as high as 3.8-4.5% and 24.0-44.9%, respectively [1-3]. Gastrectomy for gastric cancer carries a mortality of 4.1-4.7% and a morbidity of 23.6-36.0% [4, 5].
Anastomotic leakage (AL) is one of the most feared postoperative complications of gastroesophageal surgery owing to its association with prolonged hospital stay, increased mortality, and reduced quality of life [6-9]. In recent decades, improving of surgical techniques and better management of postoperative complications led to a decrease of those outcomes [10, 11], although this adverse event is still frequent, with AL incidence rates ranging from 0 to 49% following esophagectomy [12] and from 2.1 to 14.6% after gastrectomy [13].
AL can be managed by conservative (which includes fasting, nutritional support, antibiotic therapy, and wound drainage), endoscopic (clips, stents, tissue adhesives, or endoscopic vacuum therapy [EVT]), or surgical methods (primary closure of the leak, reanastomosis, or resection of the conduit). Currently, treatment decision is usually based on the characteristics of the leakage and the patient’s clinical condition, but optimal treatment remains controversial [13-15]. In the past, surgery was the treatment of choice, although it carries a higher mortality rate and nowadays is mostly used in cases of severe sepsis, large defects, or when other treatments failed or are not available/indicated.
Conservative treatment can be an option in clinically stable patients with small leakages [13, 14]. More recently, endoscopic techniques for AL were developed and appear to be safer than surgical reintervention [13, 14]. Recent systematic reviews comparing stenting with EVT found that the latter was associated with higher rate of AL closure and lower mortality [16-19]. Other endoscopic methods have also been reported as safe and effective, but most of this evidence results from small case series [13, 16]. Thus, it is unclear which is the optimal strategy for endoscopic treatment of AL after oncological gastric or esophageal surgery. Moreover, the comparison of endoscopic and surgical treatments for AL is important to confirm if endoscopic treatment should be the first-line strategy. The aims of this meta-analysis were to compare the outcomes of endoscopic and surgical treatments for AL following surgery for both esophageal and gastric cancer and to compare the outcomes of the different endoscopic methods.
Methods
This meta-analysis was performed according to the guidelines of the Preferred Reporting Items for Systematic Reviews and Meta-analyses checklist [20]. Since we performed a systematic review, no institutional board review approval or written consent was obtained.
Search Strategy
To identify published literature, a systematic search strategy was performed using 3 electronic databases (MEDLINE through PubMed, ISI Web of Knowledge, and Scopus), with last search performed on 2nd September 2020. No language or publication date restrictions were imposed. The search query for PubMed was (“anastomotic leak” OR “anastomotic dehiscence” OR “anastomosis dehiscence” OR “anastomotic fistula”) AND (gastric OR stomach OR esophag* OR oesophag* OR gastroesophageal OR “upper gastrointestinal tract”) AND (endoscopy OR “endoscopic management” OR “OTSC” OR stent OR sponge OR esophagectomy OR gastrectomy).
In addition, reference lists of review articles on the topic were searched to identify additional studies. We contacted all authors of studies that did not present the data as per inclusion criteria. Studies from authors that did not answer were not included in the quantitative analysis.
Study Selection
Studies were reviewed initially based on title and abstract by two independent investigators (I.A. and R.O). The full text of the included studies was then independently screened by the same two investigators according to the criteria below. A third author (D.L.) intervened in case of disagreement. The reasons for excluding studies were recorded. This phase was performed with Rayyan on-line platform. We included (1) randomized controlled trials, case-control or cohort studies (prospective or retrospective), and case series; (2) including patients who underwent endoscopic or surgical interventions as the first treatment for an AL following a gastroesophageal cancer surgery; (3) and evaluating the success of the endoscopic and surgical interventions in terms of at least one of the primary or secondary outcomes mentioned below. Studies were excluded if they were (1) review articles, editorials, comments, letters, and surveys; (2) case reports; (3) animal studies; (4) if they included fewer than 10 patients who met the eligibility criteria; or (5) if there was population overlap between studies. In this last case, only the study with the largest sample or study period was included.
Outcomes
The primary outcomes were (a) clinical success (defined as a complete closure of the AL, confirmed by upper endoscopy or imaging exam, with no need for reintervention and no death occurring as a consequence of the AL or its treatment, during follow-up); (b) in-hospital mortality (overall and treatment-related mortality). Secondary outcomes were rate of technical success (defined as a successful application of the chosen therapy), rate of endoscopic and surgical reintervention, rate of complications, hospital, and intensive care unit (ICU) length of stay, time until AL resolution and time until oral intake.
Data Collection
Data were extracted and recorded on an electronic data extraction sheet by two independent investigators (I.A. and R.O). Disagreements were solved by consensus. We retrieved information about: (1) study (title, first author, year of publication, country of origin, study period, study design, number of participants, number of patients with AL, and risk of bias); (2) participants (age, gender, and comorbidities); (3) tumor characteristics (location, staging, neoadjuvant therapy and type of resection, and reconstruction); (4) AL characteristics (time between surgery and AL diagnosis, modality of diagnosis, location, and dimensions of AL); (5) interventions (number of patients treated with each endoscopic and surgical method, time between cancer surgery or AL diagnosis and treatment, characteristics of each treatment), and (6) the afore-mentioned outcomes.
Assessment of Methodological Quality
The risk of bias within studies was evaluated by I.A. using the Newcastle-Ottawa Scale for cohort studies and independently checked by R.O. Disagreements were solved by consensus. We also assessed the existence of publication bias by visual inspection of funnel plots and using the Egger’s test for primary outcomes.
Statistical Analysis
We performed a meta-analysis including all studies (single-arm or double-arm) presenting data allowing the calculation of pooled prevalence (for categorical variables) and weighted mean (for continuous variables), using random-effects model with MetaXL 5.3. Double-arm comparative studies were analyzed through calculation of odds ratio (OR) and weighted mean differences (WMD). Heterogeneity between studies was tested using I2 statistic and Cochran’s Q test. Significant heterogeneity was defined as I2 >40% and/or p < 0.05. Subgroup analysis was conducted to explore potential sources of heterogeneity according to (a) tumor location (esophageal vs. gastric) and (b) mortality definition (overall mortality vs. treatment-related mortality). Sensitivity analyses were also conducted in case of important definition and/or technical differences between studies and presence of outliers.
Results
Study Selection, Study Characteristics, and Quality Evaluation
After removing 2,382 duplicates, 2,733 titles and abstracts were screened, and 126 articles underwent full-text assessment, of which 32 were included in the systematic review (Fig. 1) [21-52]. We also checked the reference list of previous systematic reviews on the topic but found no further relevant studies.
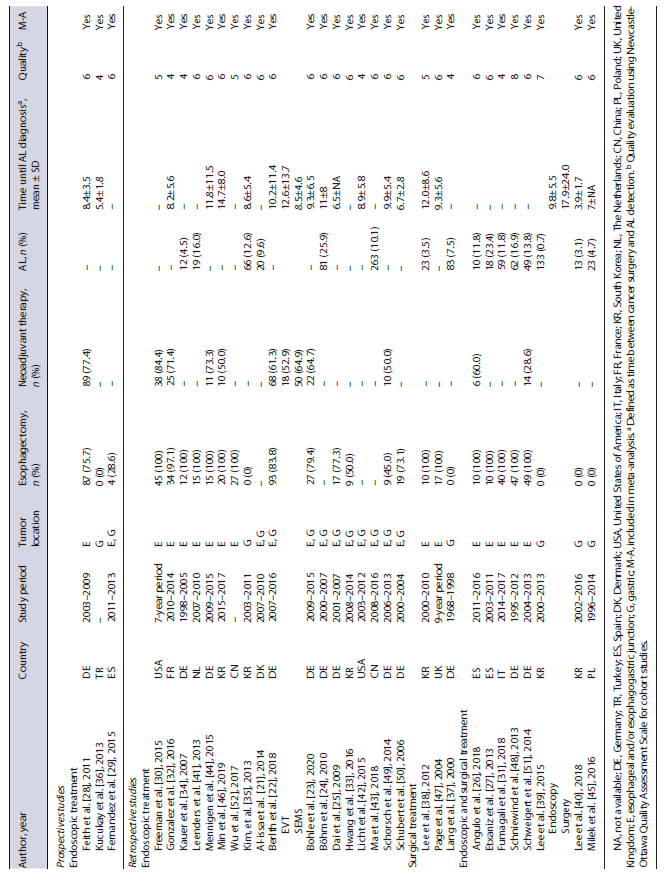
General characteristics of the included studies (29 retrospective and 3 prospective) are shown in Table 1. Details regarding demographic and clinical characteristics of the patients are presented in online supplementary Table 1 (see www.karger.com/doi/10.1159/000527769 for all online suppl. material). Twenty-one of the included studies (65.6%) evaluated endoscopic treatment [21-25, 28-30, 32-36, 41-44, 46, 49, 50, 52], 3 (9.4%) focused on surgical intervention [37, 38, 47], and 8 (25.0%) evaluated both types of intervention [26, 27, 31, 39, 40, 45, 48, 51].
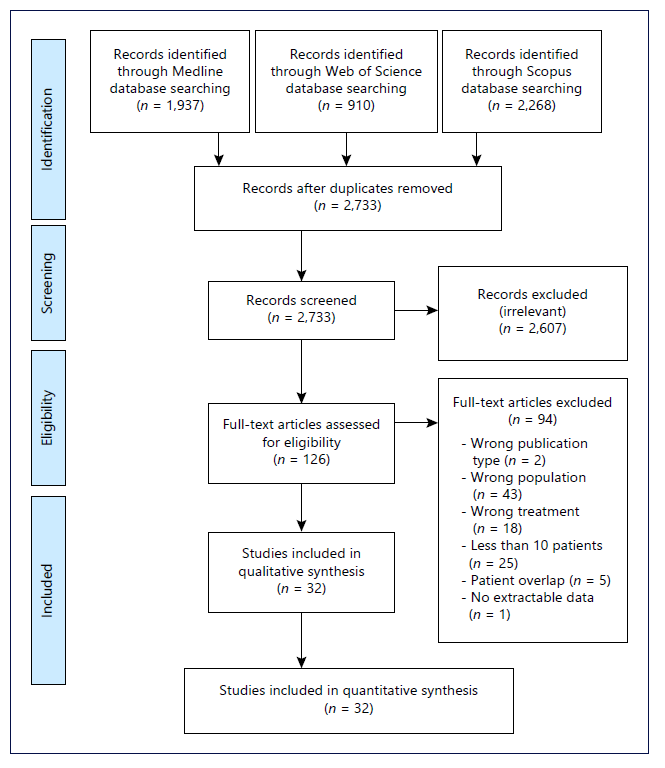
Fig. 1 Flow diagram of study selection according to the Preferred Reporting Items for Systematic Reviews and Meta-analyses.
Overall, 936 patients were treated with endoscopic methods, including 533 with stent placement (22 studies) [21-23, 25-31, 33, 34, 36, 40-43, 45, 48, 50-52], 133 with EVT (6 studies) [22, 33, 44, 46, 48, 49], 70 with clips (3 studies) [21, 43, 45, 50], 14 with fibrin glue (2 studies) [34, 50], 86 with argon plasma coagulation (1 study) [43], and 45 with multimodal interventions (3 studies) [24, 39, 41]. In 75 patients across 4 studies, the outcomes of different endoscopic treatments were evaluated together [31, 32, 35, 50]. Regarding stent placement, most studies used self-expanded metal stents, which were fully covered in the majority of the patients. Meta-analysis comparing partially and fully covered metal stents was not performed due to the low number of studies evaluating outcomes separately. Details about the endoscopic treatment are summarized in online supplementary Table 2.
A total of 144 patients were treated with surgical interventions, including 13 with anastomosis disassembly and ostomy (3 studies) [27, 31, 47], 17 with suture of anastomosis (3 studies) [37, 38, 40], 20 with reanastomosis (3 studies) [31, 45, 47], and 19 with other surgical interventions (3 studies) [31, 38, 47]. In 75 patients across 4 studies, the outcomes of different surgical treatments were evaluated together [26, 39, 48, 51].
Methodological quality of the included studies is described in Table 1. The median Newcastle-Ottawa score was 6 (IQR 5-6). Funnel plots and Egger’s test did not show evidence of publication bias when evaluating in-hospital mortality after endoscopic (p = 0.410) and surgical treatment (p = 0.169) and clinical success after endoscopic treatment (p = 0.053).
Surgical versus Endoscopic Treatment
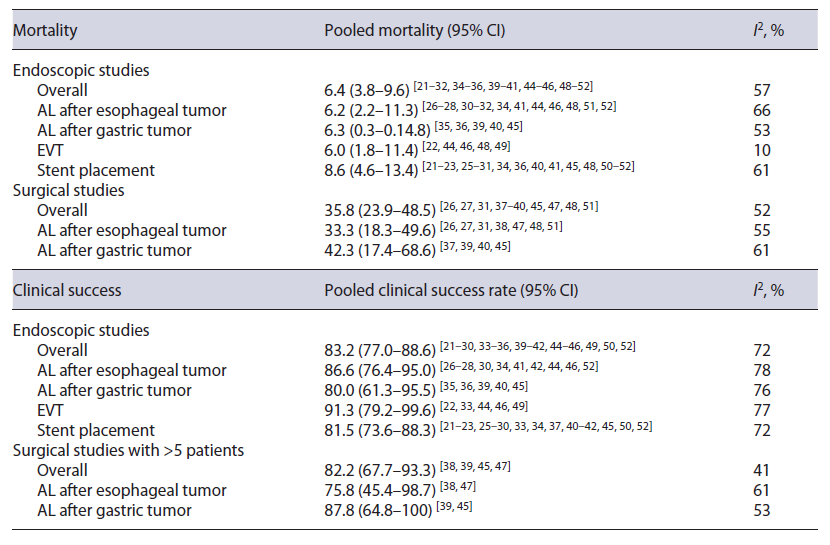
Technical success was presented in 6 endoscopic studies, with 5 of them reporting a technical success of 100% [25, 27, 28, 32, 52] and the other presenting a rate of 92.9% [29]. Clinical success (leak closure rate) was similar in endoscopic and surgical studies (83.2% [95% CI: 77.0-88.6%] vs. 82.2% [95% CI: 67.7-93.3%]) (online suppl. Fig. 1; Table 2). However, overall in-hospital mortality was significantly higher in surgical studies than in endoscopic studies (35.8% [95% CI: 23.9-48.5%) vs. 6.4% [95% CI: 3.8-9.6%]) (Fig. 2; Table 2). Death directly due to adverse events of endoscopic treatment was described in 8 endoscopic studies, and the pooled treatment-related mortality was 1.4% (95% CI: 0.0-3.8%). Clinical success and mortality were similar when stratifying by lesion location (Table 2).
After surgical treatment, there were no surgical reinterventions (0% [95% CI: 0-4.8%]) [26, 27, 31, 38-40, 45, 47]. After endoscopic treatment, the rate of surgical reintervention was 4.9% (95% CI: 2.7-7.6%) (online suppl. Fig. 2) [21-36, 39-42, 44-46, 49, 50, 52].
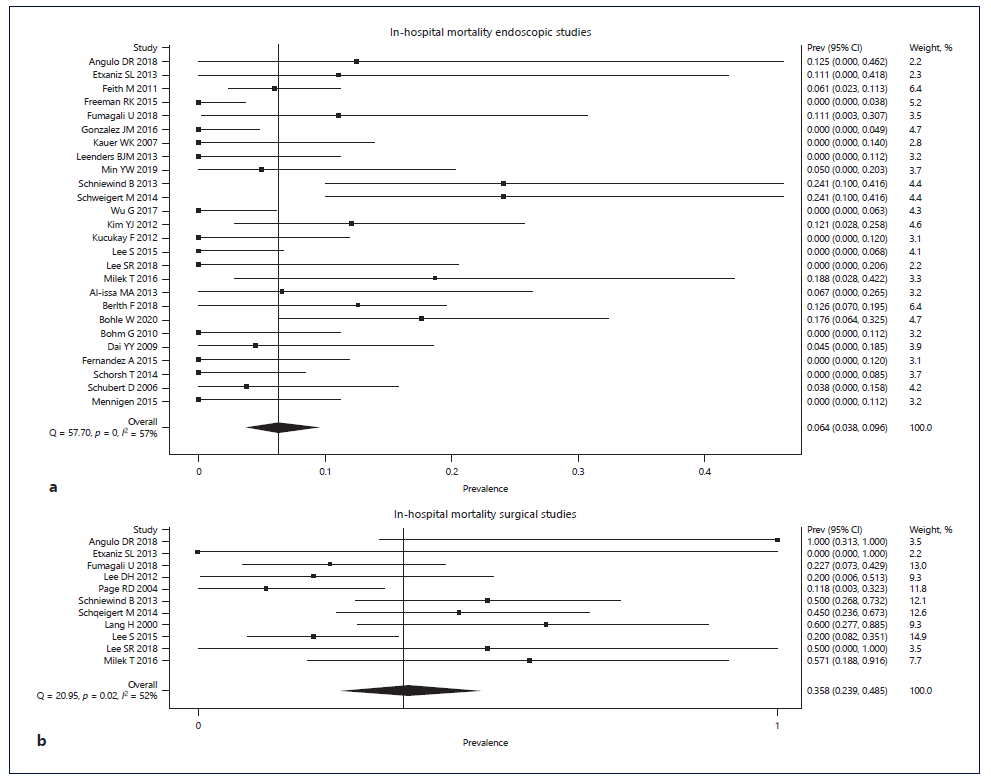
Fig. 2 Forest plot of in-hospital mortality according to treatment: (a) endoscopic treatment; (b) surgical treatment.
Surgical complications were presented in 3 studies, which reported development of stenosis, fistulae, and severe bleeding in 17.6% [47], 30.0% [38], and 2.9% [39] of the patients, respectively. Overall adverse events occurred in 26.6% of the patients treated with EVT or stenting (95% CI: 20.7-33.0%; detailed below).
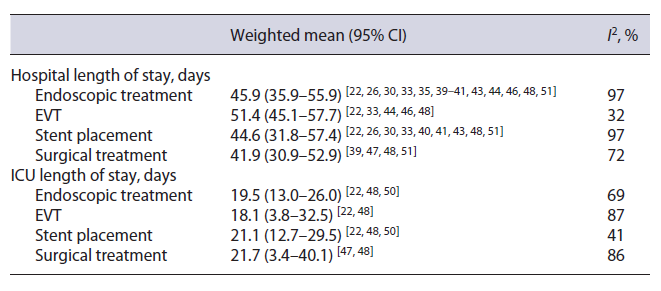
There were no significant differences in terms of hospital or ICU length of stay (Table 3). Most studies defined hospital length of stay as time between cancer surgery and discharge. Sensitivity analysis excluding two studies with slightly different definitions of this outcome [30, 39] did not significantly affect the estimates.
Time until AL resolution was only presented in 1 surgical study (50.1 ± 60.0 days) [38]. In endoscopic studies, mean time until AL resolution ranged from 12.0 to 63.4 days [22-26, 29, 30, 32, 33, 41, 42, 44, 46, 49, 50, 52].
EVT versus Stent
Single-Arm Meta-Analysis
EVT, in comparison to stent, was associated with a nonsignificantly higher clinical success rate (91.3% [95% CI: 79.2-99.6%] vs. 81.5% [95% CI: 73.6-88.3%]) (Table 2) and a nonsignificantly lower in-hospital mortality rate (6.0% [95% CI: 1.8-11.4%] vs. 8.6% [95% CI: 4.6-13.4%]) (Table 2).
EVT was associated with a nonsignificantly lower rate of surgical reinterventions (1.8% [95% CI: 0-5.2.0%] vs. 5.7% [95% CI: 2.6-9.6%]), but a nonsignificantly higher rate of endoscopic reinterventions (8.3% [95% CI: 0-21.4%] vs. 4.0% [95% CI: 2.2-6.4%]) [21-23, 25-30, 33, 34, 36, 40-42, 44-46, 49, 50, 52]. Moreover, EVT required 1 to 18 sponges, while the number of stents ranged from 1 to 7 (online suppl. Table 2).
EVT and stenting complications are shown in Table 4. The overall complications rate, considering the occurrence of migration of endoscopic device, stenosis, severe bleeding, perforation, or fistulization, was nonsignificantly lower in the EVT group (14.0% [95% CI: 3.2-27.7%] vs. 32.6% [95% CI: 24.0-41.9%]) [21-23, 25, 26, 28-30, 33, 34, 36, 40-46, 49-52]. Sensitivity analysis, excluding an outlier (Feith et al. [28], 67.0% of overall complications after stenting), did not significantly affect the estimates. EVT was associated with a nonsignificantly lower migration rate compared to stenting. Sensitivity analysis excluding an outlier (Feith et al. [28], 53% migration after stent placement) did not significantly affect the estimates. Stenosis rate was nonsignificantly higher in the EVT group. Sensitivity analysis excluding two outliers (Min et al. [46], 35% stenosis after EVT; Ma et al. [43], 43% stenosis after stent placement) found that stenosis rate was similar in EVT and stent studies. Other adverse events (severe bleeding, perforation, and fistulization) were infrequent (<3.5%) and were similar in EVT and stent groups.
There were no significant differences in terms of hospital or ICU length of stay (Table 3). Freeman et al. [30], which included patients who underwent stent placement before being transferred from other facilities, reported a shorter hospital length of stay (9.0 days [95% CI: 7.2-10.8]); sensitivity analysis excluding this study did not significantly affect the estimates.
EVT was associated with a significantly shorter time until AL resolution compared with stenting (17.6 days [95% CI: 14.1-21.2] vs. 39.4 days [95% CI: 27.0-51.8]) (online suppl. Fig. 3) [22, 23, 28, 29, 33, 41, 42, 44, 46, 49, 50, 52]. Sensitivity analysis excluding an outlier (Freeman et al. [30], 12 days until AL resolution after stent placement) did not significantly affect the estimates. Mean time until oral intake, only reported in 4 stent studies, ranged between 1.7 and 28.8 days [25, 36, 40, 43].
Double-Arm Meta-Analysis
Meta-analysis of the studies directly comparing EVT with stent placement revealed that EVT was associated with nonsignificantly higher clinical success (OR 1.91 [95% CI: 0.47-7.79]) [22, 33], lower in-hospital mortality (OR 0.39 [95% CI: 0.13-1.18]) [22, 48], and lower endoscopic (OR 0.21 [95% CI: 0.02-1.88]) (online suppl. Fig. 4) and surgical (OR 0.45 [95% CI: 0.04-5.61]) reintervention rates [22, 33]. There were also nonsignificantly lower rates of migration of endoscopic device (OR 0.51 [95% CI: 0.17-1.55]) and stenosis (OR 0.58 [95% CI: 0.09-3.97]), but a significantly lower rate of overall complications in the EVT group (OR 0.35 [95% CI: 0.13-0.95]) (online suppl. Fig. 5) [22, 33].
EVT was associated with nonsignificantly shorter time until AL resolution (WMD -8.67 days [95% CI: −22.54 to 5.20]) [22, 33] and shorter hospital length of stay (WMD -12.98 days [−31.27 to 7.98]) (online suppl. Fig. 6) [22, 33, 48]. There was a significantly shorter ICU length of stay in the EVT group compared to the stent group (WMD -14.77 days [95% CI: −26.57 to −2.98]) (online suppl. Fig. 7) [22, 48].
Other Endoscopic Treatments
Some of the included studies focused on other endoscopic treatments besides stents and EVT, namely clips, fibrin glue, argon plasma coagulation, and multimodal modalities that were not included in meta-analysis due to the reduced number of studies on these treatments. There were no deaths directly related to any of these treatments. Clipping and fibrin glue had clinical success in 66.7% and 78.6% of the patients, respectively. Multimodal modalities had higher rates of clinical success ranging from 80.0% to 96.0%. Details on the outcomes of these treatments are shown in online supplementary Table 3.
Discussion
This systematic review and meta-analysis evaluated the efficacy of endoscopic and surgical interventions in the management of AL after gastroesophageal cancer surgery. Even though there have been some reviews regarding the treatment of AL, to our knowledge, this is the first meta-analysis simultaneously comparing (1) endoscopic versus surgical interventions and (2) EVT versus stent placement in this specific context.
Our results demonstrated that endoscopic treatment, in comparison to surgical intervention, was associated with a significantly lower in-hospital mortality rate. However, no significant differences were found between these treatments in terms of clinical success, surgical reinterventions, hospital length of stay, and ICU length of stay. The decreased mortality found in the endoscopic therapy group may be related with the lesser invasiveness of these therapies, although it is also possible that there are differences in the clinical status and/or dehiscence characteristics of the patients between the two groups that may contribute to this difference in mortality. For instance, Schweigert et al. [51] found that patients in the surgical group were generally in worse condition, being more frequently septic. A recent cohort study also concluded that AL with a more severe initial presentation (i.e., requiring operative management) was associated with a lower rate of primary management success [53]. In our meta-analysis, however, most studies did not present data on AL size or clinical status at presentation.
The use of stents for the treatment of postoperative esophagogastric AL has already been established [54]. However, recent systematic reviews have shown that EVT and stent placement in single-arm studies, although in comparative studies EVT was associated with a significantly shorter ICU length of stay compared to stenting.
Our study has some limitations. Included studies are mostly retrospective, single-arm, and/or include a small sample size. In addition, one problem that led to limited comparability of several outcomes was the fact that their definitions were heterogenous or absent in many studies. Another limitation was the heterogeneity found on most analyses that did not decrease when stratifying by tumor location. Variables such as presence of comorbidities, dimensions and location of AL, time until diagnosis, or time until treatment have differences between studies and may also contribute for heterogeneity. Moreover, where-as stent placement is quite standardized and reproducible, EVT procedure may differ between institutions in terms of the magnitude of negative pressure, interval between sponge changes and placement of the sponge (extra- or intraluminal). A fourth limitation refers to the relatively low number of EVT studies and patients, which may have led to underpowerment to detect existent differences.
In conclusion, we found that endoscopic treatment was associated with a lower in-hospital mortality compared to surgical intervention. EVT is associated with a lower rate of overall complications and a shorter ICU length of stay compared to stenting. Other differences, although not statistically significant, seemed to point to a greater suggest similar efficacy and better safety profile of endoscopic treatment when compared to surgical intervention and of EVT compared to stenting. These findings can help in the definition of standardized treatment algorithms.
Although EVT seems like a promising treatment, the lack of comparative studies and standardization of clinical conditions poses a challenge in making definite conclusions. Therefore, it is essential to develop more robust prospective randomized comparative studies with standardized interventions and outcomes in order to com pare EVT with other modalities and define which is the best treatment in specific situations (according to patient and leak characteristics).