Introduction
Patients with liver cirrhosis have several alterations on the gastroduodenal mucosa that may impair the repair processes following an injury [1, 2]. Indeed, these patients are at higher risk of developing peptic ulcer, despite H. pylori prevalence - namely the main factor for peptic ulcer development - is similar to that of controls [2, 3]. Some evidences also suggest that the incidence of gastric cancer is increased in cirrhotic patients [4]. In detail, the risk of developing gastric cancer was fund to be increased by 1.7- and 5-fold in patients <60 years and >75 years old, respectively, and by 2.6-fold cumulatively as compared to the general population [4]. In the large majority of cases, gastric cancer is preceded by precancerous lesions, including ex-tensive atrophy or metaplasia involving both antrum and gastric body mucosa [5], and the intestinal metaplasia is frequently detected on gastric mucosa of cirrhotic patients [6]. Therefore, to search for H. pylori infection and pre-cancerous lesions by taking standard biopsies on gastric mucosa during upper endoscopy is worthwhile in these patients. However, clotting impairment due to both platelets and coagulation factors reduction due to liver cirrhosis may potentially increase the risk of bleeding following gastric biopsies [7, 8]. For instance, bleeding complications during percutaneous liver biopsy, central venous cannulation, paracentesis and thoracocentesis procedures, as well as surgical interventions, were reported to occur more frequently in cirrhotic patients than controls [7]. However, taking into account data of 9 studies with 587 cirrhotic patients who underwent gastric biopsies, no major bleeding was observed when platelets count was >45,000/mm3 and prothrombin activity >45% [8]. Therefore, a biopsy-free test able to suspect or rule out H. pylori infection and extensive precancerous lesions in the stomach could be advantageous. By performing a real-time analysis of gastric juice, the current version of EndoFaster® - adevice firstly developed on 2005 [9] - is able to accurately discard the presence of H. pylori infection and extensive precancerous lesions through measure of ammonium concentration and pH levels, respectively [10]. In detail, a systematic review of several studies showed that when the results of EndoFaster® testing are negative, the negative predictive value (NPV) for excluding H. pylori infection and extensive precancerous lesions on gastric mucosa is approaching 100% [10]. In detail, by considering data of 11 studies, the NPV to rule out either H. pylori infection or extensive precancerous in the stomach
was >96% in all, but two studies showing values of 84.3%-85.4% [10]. However, to our knowledge, no data on the use of EndoFaster® in cirrhotic patients are available. Indeed, the modifications of ammonia content in the gastric juice of cirrhotic patients [11], from one side, and the alterations of gastric acid output [12], in the other, may potentially affect the accuracy of gastric juice analysis with such adevice. We therefore designed this study to evaluate the accuracy of gastric juice analysis to rule out H. pylori infection and extensive precancerous lesions in patients with liver cirrhosis, aiming to avoid clinically useless and potentially unsafe gastric biopsies in these patients.
Materials and Methods
Study Design and Patients
In this prospective study, consecutive cirrhotic patients who underwent upper endoscopy for any indication in our endoscopy unit were considered for the enrolment. Inclusion criteria were the presence of a documented liver cirrhosis on the basis of clinical findings, abdominal sonogram, laboratory parameters, or liver biopsy and age >18 years. Patients with marked clotting impairment (platelet counts <50,000/mm3 and/or international normalized ratio (INR) > 1.5) were excluded [8]. Informed consent was obtained for all procedures. In detail, following explanation on the clinical research, patients were informed and signed the consent for both procedures (endoscopy with biopsies and real-time gastric juice analysis) and anonymous use of their data for scientific purposes. The study was performed in accordance with the ethical standards as laid down in the 1964 Declaration of Helsinki and its later amendments. Since no experimental drugs were administered, no additional costs or procedures for the patients were required, no identification of patients was allowed, and no funds were received, a formal approval by Investigational Review Boards could be waived.
Endoscopic Procedures
All patients underwent endoscopy and standard biopsy sampling (2 antrum, 1 angulus, and 2 gastric body) of gastric mucosa were performed with standard 5-mm biopsy forceps. After removing the biopsy specimens, we waited for haemostasis to take place. Biopsies were used for histological assessment and to search for H. pylori. The infection was considered present when histological assessment revealed the presence of bacteria together with a feature of chronic active gastritis. Extensive atrophy or intestinal metaplasia were considered present when these histological findings involved both antral and gastric body mucosa, as reported in the RE.GA.IN system and graded according to the OLGA-OLGIM systems [13, 14].
Gastric Juice Analysis
Gastric juice analysis was performed by using EndoFaster® (Manufacturer: NISO Biomed S.r.l, Turin; Italy; Italian distributor: Waldner Tecnologie Medicali, Trento; Italy). The device was provided for 2 months to the endoscopic unit without any adjunctive cost for both hospitals and patients. In detail, the device was interposed between the endoscope and the suction system, so that no adjunctive invasive procedure was required and without any discomfort for the patient [10]. During endoscopy, lumen washing was avoided until the stomach was reached and until 3 mL of gastric juice were aspirated. H. pylori diagnosis was based on the determination of ammonium concentration, as a conse-quence of the urease activity of the bacterium within 60-90 s that is during endoscopy. The device performs in the meantime also H+ concentration. The infection was considered to be present when the ammonium concentration in gastric juice was >62 ppm, whilst extensive atrophy/metaplasia involving gastric body mucosa was suspected when pH values of were >4.5, as reported elsewhere [15, 16].
Statistical Analysis
Frequencies, percentages and means values with standard deviations were calculated for all observations. Sensitivity, specificity, positive predictive value (PPV), NPV, the overall accuracy, and the likelihood ratios were calculated by considering the histology as gold standard for both H. pylori infection and extensive precancerous lesion on gastric mucosa. To assess the cumulative impact of gastric juice analysis for clinical practice, the accuracy values were also calculated by combining data of both results of testing, by considering eventually missed diagnosis when both tests were negative, but histology revealed at least one of the investigated conditions (H. pylori and/or pre-cancerous lesions).
Results
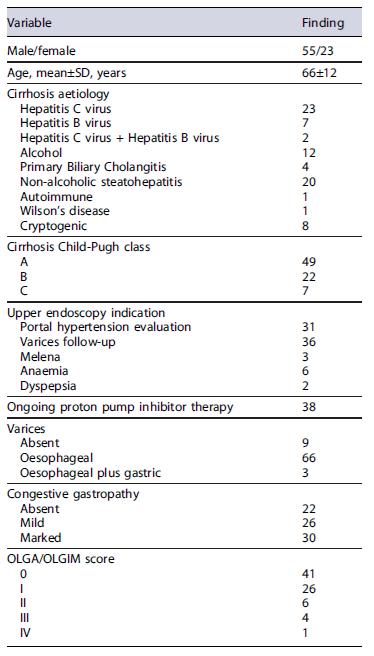
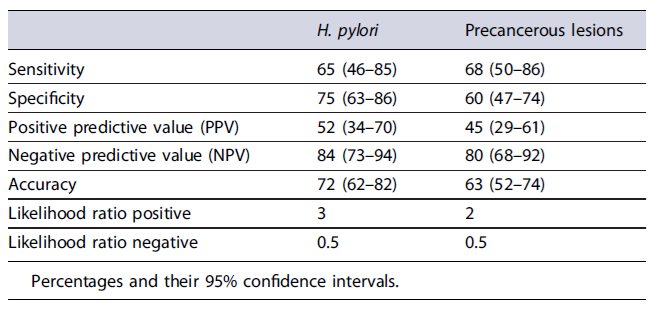
Demographic and the main clinical characteristic of the 78 cirrhotic patients enrolled in this study are provided in Table 1. Beyond varices and congestive gastropathy, at upper endoscopy 4 (5.1%) patients had erosive oesophagitis, 1 (1.3%) Barrett’soesophagus, 16 (20.5%) erosive gastritis, 4 (5.1%) gastric ulcer, 8 (8.2%) erosive duodenitis, and 1 (1.3%) duodenal ulcer. No case of bleeding following gastric biopsies occurred. At histology, H. pylori infection was diagnosed in 23 (29.5%) patients, whilst extensive atrophy/metaplasia - that is involving antral and gastric body mucosa, irrespective of grade - was present in 23 (29.5%) patients, and a grade III-IV OLGA/OLGIM was overall detected in 5 (6.4%) cases. The values of sensitivity, specificity, PPV, NPV, the overall accuracy, the likelihood ratios positive (LH+) and negative (LH−)forH. pylori infection were 65%, 75%, 52%, 84%, 72%, 3, and 0.5, and that for extensive precancerous lesions were 68%, 60%, 45%, 80%, 63%, 2, and 0.5 (Table 2). In detail, NPVs of 84% and 80% were found, respectively, in ruling out H. pylori infection and extensive atrophy/metaplasia on gastric mucosa. However, by considering as positive EndoFaster® results when at least one of two tests (ammonium and pH levels)werepositive, the NPV sincreased to 89% and 86%, respectively. These values indicate that a missed H. pylori infection occurred in only 8 cases every 100 patients with negative EndoFaster result for ammonium concentration, and a missed extensive precancerous condition in only 9 cases every 100 patients with normal pH values.
Discussion
The prevalence of gastric and duodenal ulcers has been reported to be 10-fold higher in cirrhotic patients than in controls [17], accounting for a 7.4-16% rate of upper gastrointestinal bleeding, with a 5-fold increased mortality rate in these patients [2]. Data of a meta-analysis showed that H. pylori infection significantly increases peptic ulcer risk in these patients, with an estimated odds ratio of 2.70 (95% CI = 1.91-3.82) as compared to controls [18]. Moreover, some studies suggested a potential role of H. pylori in causing hepatic encephalopathy by ammonia production in the stomach, although data are largely controversial [19]. On the other hand, the incidence of upper gastrointestinal (oesophageal, gastric, pancreatic) cancers in patients with cirrhosis was reported to be higher as compared to controls [20]. In detail, the incidence of gastric cancer was 2.6-fold increased in these patients as compared to that expected in the general population [4]. Moreover, gastric epithelial cell proliferation is increased in patients with cirrhosis, particularly when congestive gastropathy and H. pylori infection were present [21], and intestinal metaplasia is frequently detected in these patients [6, 22]. In detail, the present study showed a prevalence of extensive precancerous lesions on gastric mucosa as high as 30%, with a stage III-IV OLGA/OLGIM present in 6.4% of cases. The latter value is in agreement data of previous Italian studies showing a frequency ranging from 2.3 to 7.8%in routine endoscopic examinations [16]. These patients deserve scheduled follow-up to detect early neoplastic lesions amenable of endoscopic removal [23]. Some evidences suggest that the endoscopic submucosal dissection is safely performed also in cirrhotic patients, allowing to avoid a more harmful surgical approach [24]. All these observations clearly suggest how it is clinically worthy to exclude the presence of both H. pylori infection and precancerous lesions at endoscopy in cirrhotic patients.
This is first study that evaluated the accuracy of real-time gastric juice analysis by EndoFaster® for ruling out both H. pylori infection and extensive precancerous lesions on gastric mucosa in patients with cirrhosis. Our data found that when the cumulative results of the test were considered, a NPV of 89% and 86% in excluding H. pylori infection and extensive atrophy/metaplasia on gastric mucosa, respectively, were found, indicating an acceptably high precision for these purposes. Indeed, the NPV for H. pylori diagnosis we observed was consistent with the 92.3% reported for the 13C-urea breath testing in these patients [25]. Therefore, gastric juice analysis during endoscopy allows to safely avoid useless biopsies on normal appearing gastric mucosa, a procedure particularly advantageous in cirrhotic patients, when considering their clotting impairment [7, 8]. However, a negative EndoFaster® result decreases the post-test probability of finding both H. pylori infection and extensive precancerous, but in those situations where the bleeding risk is not prohibitive, biopsies should still be performed. On the other hand, a positive EndoFaster® increases the post-test probability and should lead to performing biopsies, when considering that both PPV and positive likelihood ratio values are not enough elevated.
Although satisfactory, the data of this study would suggest that the EndoFaster® performs less well than what observed in patients without liver cirrhosis. Indeed, data of previous studies found NPV values as high as 96% and 97% for H. pylori and precancerous lesions, respectively, in more than 2,000 investigated patients [10]. The reasons of a lower performance of gastric juice analysis in cirrhotic patients remain unclear. The relatively low sample size (N = 78) of cirrhotic patients studied in the present study may have played a role in estimating the EndoFaster® performance. However, the a posteriori calculation of the study’s power with an alfa error of 0.05 was 91.3%, by considering a NPV of 86% we computed and the 96% found in non-cirrhotic patients [10]. On the other hand, the levels of ammonium concentrations in gastric juice - that could potentially impair ammonium testing with EndoFaster® - of patients with liver cirrhosis with or without H. pylori infection were reported to be very similar to that of matched controls [11]. On the other hand, gastric acid output - that could potentially impair pH evaluation with EndoFaster® irrespective of extensive gastric mu-cosa atrophy - has been inconsistently reported to be lower, similar or increased compared to controls [26]. Therefore, the findings of present study should be confirmed in further studies.
Conclusions
This study adds evidence that real-time gastric juice analysis might avoid clinically unrewarding and potentially unsafe gastric biopsies in a definite portion of cirrhotic patients, also reducing the environmental impact [27], but more data are needed.